Abstract
Natural rubber latex (NRL) from Hevea brasiliensis has shown great potential for dermal applications due to its angiogenesis capacity and biocompatibility. Metronidazole (MET) is a synthetic antibiotic used to treat various infections. However, this drug reports dangerous effects when high concentrations are administered. In this study, we used silver nanoparticles (AgNPs) in order to minimize these toxicological effects. For this, the membranes were characterized by physicochemical, molecular modeling, in vitro release and hemocompatibility assays. In the release assays, 14.25% of the MET and 27.28% of the AgNP + MET complex were released simultaneously, indicating that these nanoparticles work as drug carriers. Molecular modeling simulation helped to explain the in vitro release results, showing that AgNPs can interact more efficiently with MET molecules. Moreover, similarities in reactivity between NRL and MET suggested that some drug molecules may remain in the matrix during the release process. The membranes did not present significant hemolytic activity after 24 h of incubation. These results demonstrated that the NRL + AgNP + MET membrane can be used as a dressing in the treatment of infectious processes.






Similar content being viewed by others
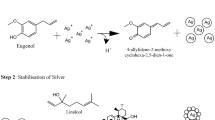
Data availability
The data that support the findings of this research are available from the corresponding author upon reasonable request.
References
Li ZZ, Tangadanchu VKR, Battini N et al (2019) Indole-nitroimidazole conjugates as efficient manipulators to decrease the genes expression of methicillin-resistant Staphylococcus aureus. Eur J Med Chem 179:723–735. https://doi.org/10.1016/j.ejmech.2019.06.093
Rocha-Garduño G, Hernández-Martínez NA, Colín-Lozano B et al (2020) Metronidazole and secnidazole carbamates: synthesis, antiprotozoal activity, and molecular dynamics studies. Molecules 25:793. https://doi.org/10.3390/molecules25040793
Cutinho PF, Roy J, Anand A et al (2019) Design of metronidazole derivatives and flavonoids as potential non-nucleoside reverse transcriptase inhibitors using combined ligand- and structure-based approaches. J Biomol Struct Dyn. https://doi.org/10.1080/07391102.2019.1614094
Upadhyay A, Chandrakar P, Gupta S et al (2019) Synthesis, Biological evaluation, structure-activity relationship, and mechanism of action studies of quinoline-metronidazole derivatives against experimental visceral leishmaniasis. J Med Chem 62:5655–5671. https://doi.org/10.1021/acs.jmedchem.9b00628
Riches A, Hart CJS, Trenholme KR, Skinner-Adams TS (2020) Anti- Giardia drug discovery: current status and gut feelings. J Med Chem 63:13330–13354. https://doi.org/10.1021/acs.jmedchem.0c00910
Olender D, Żwawiak J, Lukianchuk V et al (2009) Synthesis of some N-substituted nitroimidazole derivatives as potential antioxidant and antifungal agents. Eur J Med Chem 44:645–652. https://doi.org/10.1016/j.ejmech.2008.05.016
Naumov RN, Panda SS, Girgis AS et al (2015) Synthesis and QSAR study of novel anti-inflammatory active mesalazine–metronidazole conjugates. Bioorg Med Chem Lett 25:2314–2320. https://doi.org/10.1016/j.bmcl.2015.04.023
Tukulula M, Sharma R-K, Meurillon M et al (2013) Synthesis and antiplasmodial and antimycobacterial evaluation of new nitroimidazole and nitroimidazooxazine derivatives. ACS Med Chem Lett 4:128–131. https://doi.org/10.1021/ml300362a
Patel OPS, Jesumoroti OJ, Legoabe LJ, Beteck RM (2021) Metronidazole-conjugates: a comprehensive review of recent developments towards synthesis and medicinal perspective. Eur J Med Chem 210:112994. https://doi.org/10.1016/j.ejmech.2020.112994
Hernández Ceruelos A, Romero-Quezada LC, RuvalcabaLedezma JC, López Contreras L (2019) Therapeutic uses of metronidazole and its side effects: an update. Eur Rev Med Pharmacol Sci 23:397–401. https://doi.org/10.26355/eurrev_201901_16788
Mudry MD, Martínez-Flores I, Palermo AM et al (2001) Embryolethality induced by metronidazole (MTZ) in Rattus norvegicus: MTZ: embryolethality in Rattus norvegicus. Teratog Carcinog Mutagen 21:197–205. https://doi.org/10.1002/tcm.1008
Rodriguez Ferreiro G, Cancino Badı́asLopez-Nigro LM et al (2002) DNA single strand breaks in peripheral blood lymphocytes induced by three nitroimidazole derivatives. Toxicol Lett 132:109–115. https://doi.org/10.1016/S0378-4274(02)00039-5
Talapatra SN, Dasgupta S, Guha G et al (2010) Therapeutic efficacies of Coriandrum sativum aqueous extract against metronidazole-induced genotoxicity in Channa punctatus peripheral erythrocytes. Food Chem Toxicol 48:3458–3461. https://doi.org/10.1016/j.fct.2010.09.021
Das Roy L, Giri S, Singh S, Giri A (2013) Effects of radiation and vitamin C treatment on metronidazole genotoxicity in mice. Mutat Res/Gen Toxicol Environ Mutagen 753:65–71. https://doi.org/10.1016/j.mrgentox.2013.02.001
Menéndez D, Bendesky A, Rojas E et al (2002) Role of P53 functionality in the genotoxicity of metronidazole and its hydroxy metabolite. Mutat Res/Fundam Mol Mech Mutagen 501:57–67. https://doi.org/10.1016/S0027-5107(02)00012-X
Al-Nahi A, Abood AH, Al-Khafaji KHA (2020) Chromosomal aberration and histopathological effect of metronidazole-induced toxicity in male rat. Med Legal Update 20:607–613. https://doi.org/10.37506/mlu.v20i2.1177
Liu D, Yang F, Xiong F, Gu N (2016) The smart drug delivery system and its clinical potential. Theranostics 6:1306–1323. https://doi.org/10.7150/thno.14858
Ivanova N, Gugleva V, Dobreva M et al (2019) Silver nanoparticles as multi-functional drug delivery systems. In: Farrukh MA (ed) Nanomedicines, 5th edn. IntechOpen, London, pp 71–92
Burdușel A-C, Gherasim O, Grumezescu AM et al (2018) Biomedical applications of silver nanoparticles: an up-to-date overview. Nanomater 8:681. https://doi.org/10.3390/nano8090681
Neri F, Scala A, Grimato S et al (2016) Biocompatible silver nanoparticles embedded in a PEG–PLA polymeric matrix for stimulated laser light drug release. J Nanopart Res 18:153. https://doi.org/10.1007/s11051-016-3467-1
Guidelli ÉJ, Kinoshita A, Ramos AP, Baffa O (2013) Silver nanoparticles delivery system based on natural rubber latex membranes. J Nanopart Res 15:1536. https://doi.org/10.1007/s11051-013-1536-2
Chaiyasat P, Suksawad C, Nuruk T, Chaiyasat A (2012) Preparation and characterization of nanocomposites of natural rubber with polystyrene and styrene-methacrylic acid copolymer nanoparticles. Express Polym Lett 6:511–518. https://doi.org/10.3144/expresspolymlett.2012.54
Chen J, Chen S, Gao T et al (2019) A novel approach in blending natural rubber latex with siliceous earth nanoparticles. Iran Polym J 28:759–768. https://doi.org/10.1007/s13726-019-00740-4
Herculano RD, Silva CP, Ereno C et al (2009) Natural rubber latex used as drug delivery system in guided bone regeneration (GBR). Mater Res 12:253–256. https://doi.org/10.1590/S1516-14392009000200023
Hati S, Sakure A, Mandal S (2017) Impact of proteolytic Lactobacillus helveticus MTCC5463 on production of bioactive peptides derived from honey based fermented milk. Int J Pept Res Ther 23:297–303. https://doi.org/10.1007/s10989-016-9561-5
Garms BC, Borges FA, de Barros NR et al (2019) Novel polymeric dressing to the treatment of infected chronic wound. Appl Microbiol Biotechnol 103:4767–4778. https://doi.org/10.1007/s00253-019-09699-x
Gemeinder JLP, de Barros NR, Pegorin GS et al (2021) Gentamicin encapsulated within a biopolymer for the treatment of Staphylococcus aureus and Escherichia coli infected skin ulcers. J Biomater Sci, Polym Ed 32:93–111. https://doi.org/10.1080/09205063.2020.1817667
da Silva TV, de Barros NR, Costa-Orlandi CB et al (2020) Voriconazole-natural latex dressings for treating infected Candida spp. skin ulcers. Future Microbiol 15:1439–1452. https://doi.org/10.2217/fmb-2020-0122
Neves-Junior WFP, Ferreira M, Alves MCO et al (2006) Influence of fabrication process on the final properties of natural-rubber latex tubes for vascular prosthesis. Braz J Phys 36:586–591. https://doi.org/10.1590/S0103-97332006000400021
Herculano RD, Guimarães SAC, Belmonte GC et al (2010) Metronidazole release using natural rubber latex as matrix. Mater Res 13:57–61. https://doi.org/10.1590/S1516-14392010000100013
Herculano RD, Alencar de Queiroz AA, Kinoshita A et al (2011) On the release of metronidazole from natural rubber latex membranes. Mater Sci Eng C 31:272–275. https://doi.org/10.1016/j.msec.2010.09.007
Marques L, Martinez G, Guidelli É et al (2020) Performance on bone regeneration of a silver nanoparticle delivery system based on natural rubber membrane NRL-AgNP. Coatings 10:323. https://doi.org/10.3390/coatings10040323
Ereno C, Guimarães SAC, Pasetto S et al (2010) Latex use as an occlusive membrane for guided bone regeneration. J Biomed Mater Res 95A:932–939. https://doi.org/10.1002/jbm.a.32919
Borges FA, de Barros NR, Garms BC et al (2017) Application of natural rubber latex as scaffold for osteoblast to guided bone regeneration. J Appl Polym Sci 134:45321. https://doi.org/10.1002/app.45321
Carlos BL, Yamanaka JS, Yanagihara GR et al (2019) Effects of latex membrane on guided regeneration of long bones. J Biomater Sci, Polym Ed 30:1291–1307. https://doi.org/10.1080/09205063.2019.1627653
de Barros NR, Heredia-Vieira SC, Borges FA et al (2018) Natural rubber latex biodevice as controlled release system for chronic wounds healing. Biomed Phys Eng Express 4:035026. https://doi.org/10.1088/2057-1976/aab33a
Rosa SSRF, Rosa MFF, Marques MP et al (2019) Regeneration of diabetic foot ulcers based on therapy with red LED light and a natural latex biomembrane. Ann Biomed Eng 47:1153–1164. https://doi.org/10.1007/s10439-019-02220-5
Araujo MM, Massuda ET, Hyppolito MA (2012) Anatomical and functional evaluation of tympanoplasty using a transitory natural latex biomembrane implant from the rubber tree Hevea brasiliensis. Acta Cir Bras 27:566–571. https://doi.org/10.1590/S0102-86502012000800009
Zimmermann M, Raiser AG, Braga FVA et al (2008) Membranas de látex natural na herniorrafia diafragmática experimental em cães. Arq Bras Med Vet Zootec 60:1476–1483. https://doi.org/10.1590/S0102-09352008000600026
Wang J, Wolf RM, Caldwell JW et al (2004) Development and testing of a general amber force field. J Comput Chem 25:1157–1174. https://doi.org/10.1002/jcc.20035
Allouche A-R (2011) Gabedit-A graphical user interface for computational chemistry softwares. J Comput Chem 32:174–182. https://doi.org/10.1002/jcc.21600
Stewart JJP (2007) Optimization of parameters for semiempirical methods V: modification of NDDO approximations and application to 70 elements. J Mol Model 13:1173–1213. https://doi.org/10.1007/s00894-007-0233-4
Geerlings P, De Proft F, Langenaeker W (2003) Conceptual density functional theory. Chem Rev 103:1793–1874. https://doi.org/10.1021/cr990029p
Domingo L, Ríos-Gutiérrez M, Pérez P (2016) Applications of the conceptual density functional theory indices to organic chemistry reactivity. Molecules 21:748. https://doi.org/10.3390/molecules21060748
Yang W, Mortier WJ (1986) The use of global and local molecular parameters for the analysis of the gas-phase basicity of amines. J Am Chem Soc 108:5708–5711. https://doi.org/10.1021/ja00279a008
Maia RA, Ventorim G, Batagin-Neto A (2019) Reactivity of lignin subunits: the influence of dehydrogenation and formation of dimeric structures. J Mol Model 25:228. https://doi.org/10.1007/s00894-019-4130-4
Mandú LO, Batagin-Neto A (2018) Chemical sensors based on N-substituted polyaniline derivatives: reactivity and adsorption studies via electronic structure calculations. J Mol Model 24:157. https://doi.org/10.1007/s00894-018-3660-5
De Proft F, Van Alsenoy C, Peeters A et al (2002) Atomic charges, dipole moments, and Fukui functions using the Hirshfeld partitioning of the electron density. J Comput Chem 23:1198–1209. https://doi.org/10.1002/jcc.10067
Roy RK, Pal S, Hirao K (1999) On non-negativity of Fukui function indices. J Chem Phys 110:8236–8245. https://doi.org/10.1063/1.478792
Estrada-Salas RE, Barrón H, Valladares AA, José-Yacamán M (2012) Exploring the surface reactivity of Ag nanoparticles with antimicrobial activity: a DFT study. Int J Quantum Chem 112:3033–3038. https://doi.org/10.1002/qua.24207
Lee L-H (1991) Relevance of the hard-soft acid-base (HSAB) principle to solid adhesion. J Adhes 36:39–54. https://doi.org/10.1080/00218469108026522
Onuma Y, Satake M, Ukena T et al (1999) Identification of putative palytoxin as the cause of clupeotoxism. Toxicon 37:55–65. https://doi.org/10.1016/S0041-0101(98)00133-0
Hensens OD, Goldberg IH (1989) Mechanism of activation of the antitumor antibiotic neocarzinostatin by mercaptan and sodium borohydride. J Antibiot 42:761–768. https://doi.org/10.7164/antibiotics.42.761
Furini LN, Constantino CJL, Sanchez-Cortes S et al (2016) Adsorption of carbendazim pesticide on plasmonic nanoparticles studied by surface-enhanced Raman scattering. J Colloid Interface Sci 465:183–189. https://doi.org/10.1016/j.jcis.2015.11.045
Danna CS, Cavalcante DGSM, Gomes AS et al (2016) Silver nanoparticles embedded in natural rubber films: synthesis, characterization, and evaluation of in vitro toxicity. J Nanomater 2016:1–10. https://doi.org/10.1155/2016/2368630
Megalai SM, Manjula P, Manonmani KN et al (2012) Metronidazole: a corrosion inhibitor for mild steel in aqueous environment. Electrochim Acta 30:395–403. https://doi.org/10.4152/pea.201206395
Honary S, Barabadi H, Gharaei-Fathabad E, Naghibi F (2013) Green synthesis of silver nanoparticles induced by the fungus Penicillium citrinum. Trop J Pharm Res 12:7–11. https://doi.org/10.4314/tjpr.v12i1.2
Oliveira MJS, Rubira RJG, Furini LN et al (2020) Detection of thiabendazole fungicide/parasiticide by SERS: quantitative analysis and adsorption mechanism. Appl Surf Sci 517:145786. https://doi.org/10.1016/j.apsusc.2020.145786
Vassallo A, Silletti MF, Faraone I, Milella L (2020) Nanoparticulate antibiotic systems as antibacterial agents and antibiotic delivery platforms to fight infections. J Nanomater 2020:1–31. https://doi.org/10.1155/2020/6905631
Li P, Li J, Wu C et al (2005) Synergistic antibacterial effects of β-lactam antibiotic combined with silver nanoparticles. Nanotechnology 16:1912–1917. https://doi.org/10.1088/0957-4484/16/9/082
Alfarra A, Frackowiak E, Béguin F (2004) The HSAB concept as a means to interpret the adsorption of metal ions onto activated carbons. Appl Surf Sci 228:84–92. https://doi.org/10.1016/j.apsusc.2003.12.033
Lewars E (2003) Computational chemistry: introduction to the theory and applications of molecular and quantum mechanics, 1st edn. Kluwer Academic Publishers, Dordrecht
Schaechter M, Engleberg NC, DiRita VJ, Dermody T (2013) Schaechter’s mechanisms of microbial disease, 5th edn. Wolters Kluwer Health/Lippincott Williams & Wilkins, Philadelphia
Borges FA, FilhoMiranda EDMCR et al (2015) Natural rubber latex coated with calcium phosphate for biomedical application. J Biomater Sci, Polym Ed 26:1256–1268. https://doi.org/10.1080/09205063.2015.1086945
Fischer D, Li Y, Ahlemeyer B et al (2003) In vitro cytotoxicity testing of polycations: influence of polymer structure on cell viability and hemolysis. Biomaterials 24:1121–1131. https://doi.org/10.1016/S0142-9612(02)00445-3
de Barros NR, Miranda MCR, Borges FA et al (2016) Oxytocin sustained release using natural rubber latex membranes. Int J Pept Res Ther 22:435–444. https://doi.org/10.1007/s10989-016-9523-y
Morise BT, Chagas ALD, Barros NR et al (2019) Scopolamine loaded in natural rubber latex as a future transdermal patch for sialorrhea treatment. Int J Polym Mater Polym Biomater 68:788–795. https://doi.org/10.1080/00914037.2018.1506984
Cesar MB, Borges FA, Bilck AP et al (2020) Development and characterization of natural rubber latex and polylactic acid membranes for biomedical application. J Polym Environ 28:220–230. https://doi.org/10.1007/s10924-019-01596-8
Fayaz AM, Balaji K, Girilal M et al (2010) Biogenic synthesis of silver nanoparticles and their synergistic effect with antibiotics: a study against gram-positive and gram-negative bacteria. Nanomed: Nanotechnol Biol Med 6:103–109. https://doi.org/10.1016/j.nano.2009.04.006
Marcelino MY, Borges FA, Scorzoni L et al (2021) Synthesis and characterization of gold nanoparticles and their toxicity in alternative methods to the use of mammals. J Environ Chem Eng 9:106779–106790. https://doi.org/10.1016/j.jece.2021.106779
Acknowledgements
This work was financially supported by Higher Education Improvement Coordination (CAPES), National Council for Scientific and Technological Development (CNPq) (grants numbers 470261/2012-9 and 420449/2018-3) and the São Paulo Research Foundation (FAPESP) (grants numbers 2011/17411-8 and 2014/17526-8). This research was also supported by resources supplied by the Center for Scientific Computing (NCC/Grid UNESP) of the São Paulo State University (UNESP).
Funding
Fundação de Amparo à Pesquisa do Estado de São Paulo,2011/17411-8,Rondinelli Donizetti Herculano,2014/17526-8,Rondinelli Donizetti Herculano,Conselho Nacional de Desenvolvimento Científico e Tecnológico,470261/2012-9,Rondinelli Donizetti Herculano,420449/2018-3,Augusto Batagin-Neto
Author information
Authors and Affiliations
Contributions
MCRM: Methodology, Software, Formal analysis, Resources, Data Curation, Writing—Original Draft; NCS: Conceptualization, Methodology, Software, Formal analysis, Investigation; GSPB: Formal analysis, Writing—Original Draft, Writing—Review & Editing, Visualization; RDP: Methodology, Resources; MJ: Methodology, Resources; NRB: Software, Validation, Formal analysis, Resources; FAB: Software, Validation, Formal analysis, Resources; ABN: Investigation, Resources, Data Curation, Writing—Original Draft, Writing—Review & Editing, Visualization, Funding acquisition; WMS: Resources, Writing—Original Draft, Writing—Review & Editing, Visualization; RDH: Conceptualization, Resources, Writing—Original Draft, Writing—Review & Editing, Visualization, Supervision, Project administration, Funding acquisition; NBG: Data Curation, Writing—Original Draft, Writing—Review & Editing, Visualization.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Miranda, M.C.R., Sato, N.C., Brasil, G.S.P. et al. Silver nanoparticles effect on drug release of metronidazole in natural rubber latex dressing. Polym. Bull. 79, 9957–9973 (2022). https://doi.org/10.1007/s00289-021-03983-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-021-03983-5



