Abstract
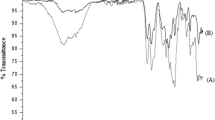
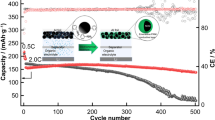
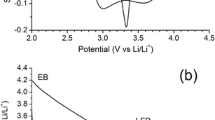
Poly(2-chloroaniline-co-2-ethylaniline)(poly(OCA-co-OEA)) was synthesized by in situ chemical oxidative method using ammonium per sulfate as an initiator and HCl as dopant. The copolymer was characterized by FTIR, UV-Visible, NMR spectroscopic techniques and XRD. FESEM and EDAX were taken to study the surface morphology and elemental composition. Thermal stability of the copolymer was confirmed by thermogravimetric analysis. The conduction was examined in terms of the dielectric constant and the electrical conductivity of 8.083 × 10–6 S cm−1 measured shows semi conducting nature of the emeraldine salt form of the copolymer. FESEM images show spongy and porous surface. The oxidation/reduction potentials and electrochemical reaction of Li/copolymer cells were tested by cyclic voltammetric technique. The discharge capacity of 63.4 mAh/g and charge capacity 62.8 mAh/g at 7–8 cycles show good reversibility. The practical specific charge capacity was 58.2% of the theoretical specific charge capacity which is 107.76 mAh/g. Hence it can efficiently be used as an anodic electrode material in lithium ion secondary batteries.




















Similar content being viewed by others
References
Vivekanandan J, Ponnusamy V, Mahudeswaran A, Vijayanand PS (2011) Synthesis, characterization and conductivity study of polyaniline prepared by chemical oxidative and electrochemical methods. Arch Appl Sci Res 3(6):147–153
Palaniappan S (2000) Chemical copolymerization of aniline witho-chloroaniline: thermal stability by spectral studies. Polym Int 49:659–662. https://doi.org/10.1002/1097-0126(200007)49:7%3c659
Vijayanand PS, Vivekanandan J, Mahudeswaran A, Ravi Kumar G, Anandarasu R (2015) Synthesis and characterization of poly (m-toluidine)-silver halide nanocomposites: thermal properties and its conducting behavior. Des Monomers Polym 18:12–17. https://doi.org/10.1080/15685551.2014.947548
Ravi Kumar G, Vivekanandan J, Mahudeswaran A, Vijayanand PS (2013) Synthesis and characterization of novel poly(aniline-co-m-aminoacetophenone) copolymer nanocomposites using dodecylbenzene sulfonic acid as a soft template. Iran Polym J 22:923–929. https://doi.org/10.1007/s13726-013-0191-x
Sharma AL, Saxena V, Annapoorni S, Malhotra BD (2001) Synthesis and characterization of a copolymer: poly(aniline-co-fluoroaniline). J Appl Polym Sci 81:1460–1466. https://doi.org/10.1002/app.1572
Schemia AL, Susana I, de Torresi C, Bassetto AN, Carlos IA (2000) Structural, morphological and spectroelectrochemical characterization of poly (2-ethyl aniline). J Bra Chem Soc 11(3):317–323
Colbow KM, Dahn JR, Haering RR (1989) Structure and electrochemistry of the spinel oxides LiTi2O4 and Li43Ti53O4. Power Sources 26:397–402
Zaghib K, Armand M, Gauthier M (1998) Electrochemistry of anodes in solid‐state li‐ion polymer batteries. J Electrochem Soc 145:3135–3140
Ohzuku T, Iwakoshi Y, Sawai K (1993) Formation of lithium‐graphite intercalation compounds in nonaqueous electrolytes and their application as a negative electrode for a lithium ion (shuttlecock) cell. J Electrochem Soc 140:2490
Nishisawa M, Hashitani R, Itoh T, Matsue T, Uchida I (1998) Physics of electron and lithium-ion transport in electrode materials for Li-ion batteries Solid-State. Met 1:10–12
Zaghib K, Simoneau M, Armand M, Gauthier M (1999) Electrochemical study of Li4Ti5O12 as negative electrode for Li-ion polymer rechargeable batteries. J Power Sources 81–82:300–305
Linganathan P, Samuel JM (2014) Effect of Dodecyl Benzene Sulphonic Acid on the Electrical Conductivity Behaviour of Poly(2-chloroaniline) and Poly(2-chloroaniline)/Silk Blends. American J Polymer Sci 4(4):107–116. https://doi.org/10.5923/j.ajps.20140404.02
Haba Y, Segal E, Narkis M, Titelman GI, Siegmann A (2000) Polyaniline–DBSA/polymer blends prepared via aqueous dispersions. Synth Met 110:189–193
Wei G, Zhengwei L, Nigel S (2011) An Introduction to Electronic Materials for Engineers. In: Zhengwei L, Nigel S (ed) Ionic conductivity and dielectric studies of LiClO4 doped poly(vinylalcohol)(PVA)/chitosan(CS) composites, 2nd edn. Journal of Advanced Dielectrics, World Scientific Publishing Company, pp 345–440
Kim YH, Foster C, Chiang J, Heeger AJ (1988) Photoinduced localized charged excitations in polyaniline. Synth Met 26(1):49–59
Savitha P, Sathyanarayana DN (2004) Poly (o-toluidine-co-o-chloroaniline) copolymers: effect of polymerization conditions on the properties of the copolymers. Indian J Chem 43A:487–493
Oh M, Kim S (2012) Effect of dodecyl benzene sulfonic acid on the preparation of polyaniline/activated carbon composites by in situ emulsion polymerization. Electrochim Acta 59:196–201
Lee D, Char K, Lee SW, Park YW (2003) Structural changes of polyaniline/montmorillonite nanocomposites and their effects on physical properties. J Mater Chem 13:2942–2947
Bhadra S, Khastgir D, Singha NK (2009) Progress in preparation, processing and applications of polyaniline. Prog Polym Sci 34(8):783–810
Sergeyeva TA, Lavrik NV, Piletsky SA, Rachkov AE, El`skaya AV (1996) Polyaniline label-based conductometric sensor for IgG detection. Sensor Actuat B-Chem 34(1–3):283–288
Nguyen VH, Shim JJ (2011) Facile synthesis and characterization of carbon nanotubes/silver nanohybrids coated with polyaniline. Synth Met 161:2078–2082
Nguyen VH, Shim JJ (2013) Supercritical fluid-assisted synthesis of a carbon nanotubes-grafted biocompatible polymer composite. Compos Interfaces 20:155–162
Shamsudin MS, Suriani AB, Abdullah S, Yahya SYS, Rusop M (2013) Impact of thermal annealing under nitrogen ambient on structural micro-Raman, and thermogravimetric analyses of camphoric-CNT. J Spectrosc Article. https://doi.org/10.1155/2013/167357
Oueiny C, Berlioz S, Perrin FX (2014) Carbon nanotube–polyaniline composites. Prog in Poly Sci 39(4):707–748
Gajendran P, Saraswathi R (2008) Polyaniline-carbon nanotube composites. Pure Appl Chem 80(11):2377–2395
Gok A, Seri B, Talu M (2004) Synthesis and characterization of conducting substituted polyanilines. Synth Met 142:41–48
Saroj AL, Singh RK (2012) Thermal, dielectric and conductivity studies on PVA/Ionic liquid [EMIM][EtSO4] based polymer electrolytes. J Phys Chem Solids 73(2):162–168
Tang Z, Qi L, Gao G (2009) Polymer electrolytes based on copolymer of poly(ethylene glycol) dimethacrylate and imidazolium ionic liquid. Solid State Ion 180:226–230
Jeevanandam P, Vasudevan S (1998) Arrhenius and non-Arrhenius conductivities in intercalated polymer electrolytes. J Chem Phys 109:8109
Kadir MFZ, Majid SR, Arof AK (2010) Plasticized chitosan–PVA blend polymer electrolyte based proton battery. Electrochim Acta 55(4):1475–1482
Suthanthiraraj SA, KumaraVadivel SM (2012) Electrical and structural properties of poly (ethylene oxide)/silver triflate polymer electrolyte system dispersed with MgO nanofillers. Ionics 18:385–394
Genies EM, Hany P, Santier C (1988) A rechargeable battery of the type polyaniline/propylene carbonate-LiClO4/Li-Al. J Appl Electrochem 18:751–756
MacDiarmid AG, Chiang JC, Richter AR, Epstein AJ (1987) Polyaniline: a new concept in conducting polymers. Synth Met 18:285–290
Ryu KS, Kim KM, Kang SG, Lee GJ, Joo SH (2000) Electrochemical and physical characterization of lithium ionic salt doped polyaniline as a polymer electrode of lithium secondary battery. Synth Met 110(3):213–217
Ryu KS, Kim KM, Kang SG, Lee GJ, Chang SH (2000) The charge/discharge mechanism of polyaniline films doped with LiBF4 as a polymer electrode in a Li secondary battery. Solid State Ion 135(1–4):229–234
Sertkol SB, Sinirlioglu D, Esat B, Muftuoglu AE (2015) A novel cathode material based on polystyrene with pendant TEMPO moieties obtained via click reaction and its use in rechargeable batteries. J Res Polym. https://doi.org/10.1007/s10965-015-0777-4
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Padmaja, S., Samuel, J.M. Chemically copolymerized poly(2-chloroaniline-co-2-ethylaniline) as an anodic material in Li-ion batteries. Polym. Bull. 78, 7129–7146 (2021). https://doi.org/10.1007/s00289-020-03478-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-020-03478-9