Abstract
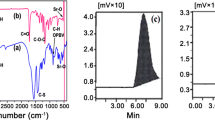
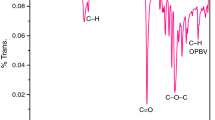
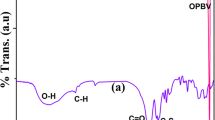
The calcium mercaptosuccinate (Ca-MS) functionalized poly(ε-caprolactone) (PCL) was successfully synthesized by ring-opening polymerization (ROP) method at 160 °C for 2 h under nitrogen atmosphere with mild stirring in the presence of stannous octoate (SO) as ROP catalyst. The synthesized Ca-MS functionalized PCL was characterized by FTIR spectroscopy, NMR spectroscopy, atomic force microscopy, HRTEM and polarized optical microscopy (POM) like analytical techniques. The microstructure of Ca-MS nucleated PCL crystals was analysed by POM. The thermal properties of Ca-MS functionalized PCL were examined using differential scanning calorimetry and thermogravimetric analysis techniques. The non-isothermal crystallization and degradation kinetic studies were carried out to assess the crystallinity as well as the energy of activation (Ea) of Ca-MS functionalized PCL. The FTIR spectrum showed a peak at 1720 cm−1 corresponding to the carbonyl stretching of PCL. The 1H-NMR spectrum showed an alkoxy proton signal at 4.1 ppm. The non-isothermal crystallization kinetic study showed the 3D crystal growth with the Ea value of 142.4 kJ/mol.













Similar content being viewed by others
References
Sung GA, Chang GC (2004) Synthesis and characterization of amphiphilic poly(caprolactone) star block copolymers. Macromol Rapid Commun 25:618–622
Vroman I, Tighzert L (2009) Bio-degradable polymers. Materials 2:307–344
Ayala GG, Pace ED, Laurienzo P, Pantalena D, Somma E, Nobila MR (2009) Poly(ε-caprolactone) modified by functional groups: preparation and chemical-physical investigation. Eur Polym J 45:3217–3229
Ludueña LN, Alvarez VA, Vazquez A (2007) Processing and microstructure of PCL/clay nanocomposites. Mater Sci Eng A 461:121–129
Liao L, Zhang C, Gong S (2007) Preparation of poly(ε-caprolactone)/clay nanocomposites by microwave assisted in situ ring opening polymerization. Macromol Rapid Commun 28:1148–1154
Bryan GO, Wong BM, Mcelhanon JR (2010) Stress sensing in polycaprolactone films via an embedded photochromic compound. Appl Mater Interfaces 2:1594–1600
Sivabalan A, Meenarathi B, Palanikumar S, Anbarasan R (2014) Synthesis and characterization of poly(caprolactone): a comparative study. Int J Sci Res Eng Tech 1:9–14
Gosur M, Yilmaz F, Kilic A, Demirei A, Kosemen A, San SE (2010) Synthesis, characterization, electrochromic properties, and electrochromic device application of a novel star polymer consisting of thiophene end-capped poly(ε-caprolactone) arms emanating from a hexafunctional cyclotriphosphazene core. J Polym Sci A Polym Chem 48:3668–3682
Meenarathi B, Siva P, Palanikumar S, Kannammal L, Anbarasan R (2016) Synthesis, characterization and drug release activity of poly(caprolactone)/Fe3O4 nanocomposites. Nanocomposites 2:98–107
Pappalardo D, Ahnumziata L, Pellecchia C (2009) Synthesis and spectroscopic studies of macrocyclic polystyrene containing two fluorene units and single 9,10-anthracenylidene group. Macromolecules 42:6015–6062
Sivabalan B, Meenarathi S, Palanikumar R (2014) Synthesis, characterization, application and band gap study of calcium mercaptosuccinate. J Sci Res Eng Tech 1:9–14
Jiang L, Lou W, Sun L, Xu Z (2005) Ring opening polymerization of caprolactone with a divalent samarium bis(phosphido) complex. J Appl Polym Sci 98:1558–1564
Chen HH, Anbarasan R, Kuo LS, Chen PH (2011) A novel report on Eosin Y functionalized MWCNT as an initiator for the ring opening polymerization of ε-caprolactone. Mater Chem Phys 126:584–590
Contreras JM, Medina D, López-Carrasquero F, Contreras RR (2013) Ring opening polymerization of ε-caprolactone initiated by samarium acetate. J Polym Res 20:244–249
Monelave M, Contreras JM, Laredo E, Carrasquero FL (2010) Ring opening polymerization of (R, S)-β-butyrolactone and caprolactone using sodium hydride as initiator. Exp Polym Lett 7:431–441
Persenaire O, Alexandre M, Degée P, Dubois P (2001) Mechanisms and kinetics of thermal degradation of poly(ε-caprolactone). Biomacromolecules 2:288–294
Annette C, Renouf G, John R, David FF, Ruth EC (2005) The effect of crystallinity on the deformation mechanism and bulk mechanical properties of PLLA. Biomaterials 26:5771–5782
Tsuji H, Ikada Y (1995) Synthesis and spectroscopic studies of macrocyclic polystyrene containing two fluorene units and single 9,10-anthracenylidene group. Polymer 6:2709–2716
Yaming W, Rodriguez-Perez A, Reis L, Mano F (2005) Thermal and thermomechanical behaviour of poly(caprolactone) and poly(caprolactone)/starch blends for biomedical applications. Macromol Mater Eng 290:792–801
Stavroula G, George Z, Papageorgiou DN (2012) Crystallization of novel poly(ε-caprolactone)-block-poly(propyleneadipate) copolymers. J Therm Anal Calorim 108:633–645
Achla S, Miti N, Joesmon J (2017) Non-isothermal crystallization and microstructural behaviour of poly(ε-caprolactone) and granular tapioca starch-based biocomposite. Int J Polym Anal Charact 22:222–236
Vackova T, Ostafinska A, Kova SK, Nevoralova M, Slouf M (2017) Impact of particle morphology on structure, crystallization kinetics, and properties of PCL composites with TiO2 based particles. Polym Bull 74:445–464
Nanaki SN, Papageorgion GZ, Bikiaris DN (2012) Crystallization of novel poly(caprolactone-block-propyladipate) copolymers. J Therm Anal Calorim 108:633–645
Su TT, Jiang H, Gong H (2008) Thermal stabilities and the thermal degradation kinetics of poly(ε-caprolactone). Polym Plast Technol Eng 47:398–403
Wang XL, Huang FY, Zhou Y, Wang YZ (2009) Non-isothermal crystallization kinetics of poly(ε-caprolactone)/montmorillonite nanocomposites. J Macromol Sci Part B Phys 48:710–722
Chrissafis K, Antoniadis G, Paraskevopoulos KM, Vassiliou A, Bikiaris DN (2007) Comparative study of the effect of different nanoparticles on the mechanical properties and thermal degradation mechanism of in situ prepared poly(e-caprolactone) nanocomposites. Compos Sci Technol 67:2165–2174
Vyazovkin S, Sbirrazzuoli N (2003) Estimating the activation energy for non-isothermal crystallization of polymer melts. J Therm Anal Calorim 72:681–686
Vyazovkin S, Burnham AK, Criado JM, Perez-Maqueda LA, Popescu C, Sbirrazzuoli N (2011) ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta 520:1–19
Demir P (2017) Investigation of thermal degradation kinetics of poly(ε-caprolactone) grafted onto PEMA-co-PHEMA. J Sci Eng 17:73–85
Chrissafis K (2009) Kinetics of thermal degradation of polymers: complementary use of isoconversional and model-fitting methods. J Therm Anal Calorim 95:273–283
Meenarathi B, Siva P, Palanikumar S, Kannammal L, Anbarasan R (2016) Synthesis, characterization and drug release activity of poly(ε-caprolactone)/Fe3O4–alizarinred nanocomposites. Nanocomposites 2:98–107
Wang L, Sheng J (2014) Non-isothermal crystallization kinetics of polypropylene/attapulgite nanocomposites. J Macromol Sci B Phys 44:31–42
Ma J, Zhang S, Qi Z, Li G, Hu Y (2002) Viscoelastic properties of young and old human dermis: a proposed molecular mechanism for elastic energy storage in collagen and elastin. J Appl Polym Sci 83:1978–1985
Kawazu K, Nakagawa S, Ishizone T, Nojima S, Arai D, Yamaguchi K, Nakahama S (2017) Effects of bulky end groups on the crystallization kinetics of poly(ε-caprolactone) homopolymers confined in a cylindrical nanodomain. Macromolecules 50:7202–7210
Wu TM, Chen EC (2006) Isothermal and non-isothermal crystallization kinetics of poly(ε-caprolactone)/multi-walled carbon nanotube composites. Polym Eng Sci 46:1309–1317
Jancirani A, Kohila V, Meenarathi B, Yellilarasi A, Anbarasan R (2016) Synthesis, characterization and non-isothermal degradation kinetics of novel poly(mono ethylene glycol dimethacrylate-co-4-aminobenzoate). Bull Mater Sci 39:1725–1733
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mahalakshmi, S., Alagesan, T., Parthasarathy, V. et al. Thermal degradation and crystallization kinetics studies on synthesized calcium mercaptosuccinate end-capped poly(ε-caprolactone) nanocomposite. Polym. Bull. 76, 4991–5009 (2019). https://doi.org/10.1007/s00289-018-2614-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-018-2614-5