Abstract
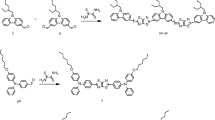
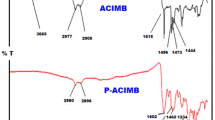
Poly(p-phenylene-1-(2,5-dimethylphenyl)-5-phenyl-1H-pyrazole-3,4-dicarboxy amide) (poly(PDPPD)) was synthesized from reactions of p-phenylene-diamine and 1-(2,5-dimethylphenyl)-5-phenyl-1H-pyrazole-3,4-dicarbonyl dichloride (monomer) by heating under solvent. This newly synthesized poly(PDPPD) was characterized by 1H, 13C-NMR, FT-IR, gel permeation chromatography (GPC). In addition, we investigated optical properties of the poly(PDPPD) containing the substituted pyrazole ring at different molarities. The average transmittance (T avg) values of the poly(PDPPD) in the visible (V) region were higher than the T avg values in the near-ultraviolet (NU) region. The T avg values of the poly(PDPPD) in the NU and visible (V) region decreased with increasing molarity. The absorption band edge (Absbe) value of the poly(PDPPD) decreased with increasing molarity. It was observed that the optical band gap (E g) of the poly(PDPPD) value decreased more with increasing molarity. The refractive index of the poly(PDPPD) decreased with increasing wavelength and decreasing molarity. The structure of the poly(PDPPD) in the lowest energy was optimized by DFT calculation and its HOMO–LUMO orbitals were plotted.








Similar content being viewed by others
References
Kamal A, Shaik AB, Jain N, Kishor C, Nagabhushana A, Supriya B, Chourasiya SS, Suresh Y, Mishra KR, Addlaganatta A (2015) Design and synthesis of pyrazole–oxindole conjugates targeting tubulin polymerization as new anticancer agents. Eur J Med Chem 92:501–513
Kendre BV, Landge MG, Jadhav WN, Bhusare SR (2013) Synthesis and bioactivities of some new 1H-pyrazole derivatives containing an aryl sulfonate moiety. Chin Chem Lett 24:325–328
Mert S, Kasımoğulları R, Iça T, Çolak F, Altun A, Ok S (2014) Synthesis, structure–activity relationships, and in vitro antibacterial and antifungal activity evaluations of novel pyrazole carboxylic and dicarboxylic acid derivatives. Eur J Med Chem 78:86–96
El-Sayed AAM, Abdel-Aziz IN, Abdel-Aziz MAA, El-Azab SA, ElTahir EHK (2012) Synthesis, biological evaluation and molecular modelling study of pyrazole and pyrazoline derivatives as selective COX-2 inhibitors and anti-inflammatory agents. Part 2. Bioorg Med Chem 20:3306–3316
Shen SL, Zhu J, Li M, Zhao BX, Miao JY (2012) Synthesis of ferrocenyl pyrazole-containing chiral aminoethanol derivatives and their inhibition against A549 and H322 lung cancer cells. Eur J Med Chem 54:287–294
Chambers LJ, Stevens AJ, Moses AP, Michel AD, Walter SD, Davies DJ, Livermore DG, Fonfria E, Demont EH, Vimal M, Theobald PJ, Beswick PJ, Gleave RJ, Roman SA, Stefan S (2010) Synthesis and structure–activity relationships of a series of (1H-pyrazol-4-yl) acetamide antagonists of the P2X7 receptor. Bioorg Med Chem Lett 20:3161–3164
Kaya E, Gündüz B, Çetin A (2016) Synthesis and characterization of conjugated polymers containing phenyl and bithiophene: controlling of optical properties with molarity. Colloid Polym Sci 294:339–345
Chang EM, Lee CT, Chen CY, Wong FF, Yeh MY (2008) Synthesis, optical properties, and characterization of new soluble conjugated poly (p-phenylenevinylene) derivatives constituted of alternating pyrazole and 1, 3, 4-oxadiazole moieties. Aust J Chem 61:342–349
Lv HS, Zhao BX, Li JK, Xia Y, Lian S, Liu WY, Gong ZL (2010) The synthesis, characterization and optical properties of novel, substituted, pyrazoly1,3,4-oxadiazole derivatives. Dyes Pigments 86:25–31
Bailey DM, Hansen PE, Hlavac AG, Baizman ER, Pear J, DeFelice AF, Feigensonf ME (1985) 3,4-Diphenyl-1H-pyrazole-1-propanamine antidepressants. J Med Chem 28:256–260
Siddall TL, Ouse DG, Benko ZL, Garvin GM, Jackson JL, McQuiston JM, Ricks MJ, Thibault TD, Turner JA, VanHeertum JC, Weimer MR (2002) Synthesis and herbicidal activity of phenyl-substituted benzoylpyrazoles. Pest Manag Sci 58:1175–1186
Tahermansouri H, Biazar E (2013) Functionalization of carboxylated multi-wall carbon nanotubes with 3,5-diphenyl pyrazole and an investigation of their toxicity. New Carbon Mater 28:199–207
Terrett NK, Bell AS, Brown D, Ellis P (1996) Sildenafil (Viagra TM), a potent and selective inhibitor of type 5 cGMP phosphodiesterase with utility for the treatment of male erectile dysfunction. Bioorg Med Chem Lett 6:1819–1824
Ramkumar V, Kannan P (2015) Novel heterocyclic based blue and green emissive materials for opto-electronics. Opt Mater 46:314–323
Oh CS, Lee JY (2014) High efficiency blue phosphorescent organic light-emitting diodes using 2-(1H-pyrazol-1-yl) pyridin-3-ol ligand based Be compound. Dyes Pigments 101:25–29
Cherpak V, Stakhira P, Khomyak S, Volynyuk D, Hotra Z, Voznyak L, Oliynyk O (2011) Properties of 2, 6-di-tert-butyl-4-(2, 5-diphenyl-3, 4-dihydro-2H-pyrazol-3-yl)-phenol as hole-transport material for life extension of organic light emitting diodes. Opt Mater 33:1727–1731
Duan Z, Hu D, Ohuchi H, Zhao M, Zhao G, Nishioka Y (2012) Organic field-effect transistors based on two phenylene–thiophene oligomer derivatives with a biphenyl or fluorene core. Synth Metals 162:1292–1298
Back JY, Kim Y, An TK, Kang MS, Kwon SK, Park CE, Kim YH (2015) Synthesis and electrical properties of novel oligomer semiconductors for organic field-effect transistors (OFETs): asymmetrically end-capped acene-heteroacene conjugated oligomers. Dyes Pigments 112:220–226
Kandasamy K, Ganesabaskaran S, Pachamuthu MP, Ramanathan AA (2015) A novel pyrazole biscoumarin based chemosensors for the selective detection of Cu2+ and Zn2+ ions. Spectrochim Acta A 148:184–188
Sengupta S, Goswami A, Mondal R (2014) Silver-promoted gelation studies of an unorthodox chelating tripodal pyridine–pyrazole-based ligand: templated growth of catalytic silver nanoparticles, gas and dye adsorption. New J Chem 38:2470–2479
Gondek E (2013) Photovoltaic solar cells based on pyrazole derivative. Mater Lett 112:94–96
Gómez-Iglesias P, Guyon F, Khatyr A, Ulrich G, Knorr M, Martín-Alvarez JM, Villafañe F (2015) Luminescent rhenium (I) tricarbonyl complexes with pyrazolylamidino ligands: photophysical, electrochemical, and computational studies. Dalton Trans 44:17516–17528
Ma CQ, Zhang LQ, Zhou JH, Wang XS, Zhang BW, Cao Y, Qiu Y (2002) 1,3-Diphenyl-5-(9-phenanthryl)-4, 5-dihydro-1H-pyrazole (DPPhP): structure, properties, and application in organic light-emitting diodes. J Mater Chem 12:3481–3486
Deng L, Li J, Li W (2014) Solution-processible small-molecular host materials for high-performance phosphorescent organic light-emitting diodes. Dyes Pigments 102:150–158
Singh M, Haverinen HM, Dhagat P, Jabbour GE (2010) Inkjet printing—process and its applications. Adv Mater 22:673–685
Sakthinathan SP, Vanangamudi G, Thirunarayanan G (2012) Synthesis, spectral studies and antimicrobial activities of some 2-naphthyl pyrazoline derivatives. Spectrochim Acta A 95:693–700
Yakimansky AV, Menshikova AY, Shevchenko NN, Shabsels BM, Bazhenova AG, Sel’kin AV, Alfimov MV (2009) From polymeric nanoparticles to dye-containing photonic crystals: synthesis, self-assembling, optical features, and possible applications. Polym Adv Tech 20:581–588
Lan L, Zhang G, Dong Y, Ying L, Huang F, Cao Y (2015) Novel medium band gap conjugated polymers based on naphtho [1, 2-c: 5, 6-c] bis [1–3] triazole for polymer solar cells. Polymer 67:40–46
Çetin A, Bildirici İ (2016) A study on synthesis and antimicrobial activity of 4-acyl-pyrazoles. J Saudi Chem Soc. doi:10.1016/j.jscs.2016.05.008
Sener A, Tozlu I, Genc H, Bildirici I, Arisoy K (2007) Synthesis and some reactions of 4-(ethoxycarbonyl)-1,5-diphenyl-1H-pyrazole-3-carboxylic acid. J Het Chem 44:1077–1081
Valero R, Gomes JR, Truhlar DG, Illas F (2010) Density functional study of CO and NO adsorption on Ni-doped MgO (100). J Chem Phys 132:104701–104709
Raczynska E, Hallmann M, Duczmal K (2011) Quantum-chemical studies of amide–iminol tautomerism for inhibitor of lactate dehydrogenase: oxamic acid. Comput Theor Chem 964:310–317
Tauc J, Menth A (1972) States in the gap. J Non-Crystal Solids 10:569–585
Gündüz B (2015) Optical properties of poly [2-methoxy-5-(3′,7′-dimethyloctyloxy)-1,4-phenylenevinylene] light-emitting polymer solutions: effects of molarities and solvents. Polym Bull 72:3241–3267
Gündüz B (2015) Surface morphology properties of the 5,5′-Di (4-biphenylyl)-2,2′-bithiophene (PPTTPP) nanofiber film and calculations of the optical parameters of the PPTTPP nanofiber optoelectronic devices. J Nanoelectron Optoelectron 10:1–8
Ho WF, Uddin MA, Chan HP (2009) The stability of high refractive index polymer materials for high-density planar optical circuits. Polym Degrad Stab 94:158–161
Ticha H, Tichy L (2002) Semiempirical relation between non-linear susceptibility (refractive index), linear refractive index and optical gap and its application to amorphous chalcogenides. J Optoelectron Adv Mater 4:381–386
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors confirm that this article content has no conflict of interest. There is no financial funding and support for our manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cetin, A., Gündüz, B., Menges, N. et al. Unsymmetrical pyrazole-based new semiconductor oligomer: synthesis and optical properties. Polym. Bull. 74, 2593–2604 (2017). https://doi.org/10.1007/s00289-016-1846-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-016-1846-5