Abstract
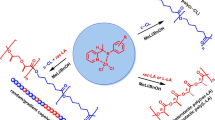
Dioxomolybdenum complexes were examined as catalysts for the copolymerization of ε-caprolactone (ε-CL) and l-lactide (l-LA). The bis-[(5-OMe)salicylaldehydato]dioxomolybdenum complex completed the copolymerization after 20 h at 110 °C with 0.05 mol% of the catalyst to produce a copolymer in high yield. The microstructure of the copolymer was analyzed using 1H and 13C NMR spectroscopy and was determined to have a random structure. The r values calculated from the heterodiad analysis of the 1H NMR data were r LA = 0.91 and r CL = 0.93, and the L values calculated from the triad analysis of the 13C NMR data were L LA = 1.58 and L CL = 1.81. Other dioxomolybdenum complexes, such as cis-α MoO2[(3-MeO)DiMeSaltn], MoO2(acac)2 and (NH4)8[Mo10O34] exhibited comparable or slightly lower reactivity for the copolymerization. Consecutive polymerization of ε-CL followed by l-LA afforded a block copolymer without trans-esterification.




Similar content being viewed by others
Notes
Although the initiator moiety could not be analyzed in the 1H NMR spectra of the resulting polymer due to overlapping, the degree of polymerization was calculated based on the integration of the peaks of the alcohol terminal.
Copolymerization with other lactones: δ-Valerolactone (δ-VL) afforded copolymers with slightly reduced contents of δ-VL with nearly equal efficiency as ε-CL. However, substituted lactones such as β-BL and HL, only reacted slightly under the reaction conditions to afford copolymers with fairly reduced contents of the substituted lactones. See supporting information.
Higher solubility of the copolymer in organic solvents resulted in low recovery of the products.
References
Penczek S, Cypryk M, Duda A, Kubisa P, Słomkowski S (2007) Living ring-opening polymerizations of heterocyclic monomers. Prog Polym Sci 32:247–282
Albertsson AC, Varma IK (2003) Recent developments in ring opening polymerization of lactones for biomedical applications. Biomacromolecules 4:1466–1486
Grijpma DW, Pennings AJ (1991) Polymerization temperature effects on the properties of l-lactide and ε-caprolactone copolymers. Polym Bull 25:335–341
Vanhoorne P, Dubois P, Jerome R, Teyssie P (1992) Macromolecular engineering of polylactones and polylactides. 7. structural analysis of copolyesters of ε-caprolactone and l- or d, l-lactide initiated by Al(OiPr)3. Macromolecules 25:37–44
Bero M, Kasperczyk J, Adamus G (1993) Coordination polymerization of lactides. 3. Copolymerization of l, l-lactide and ε-caprolactone in the presence of initiators containing Zn and Al. Makromol Chem 194:907–912
Kasperczyk J, Bero M (1993) Coordination polymerization of lactides. 4. The role of transesterification in the copolymerization of l, l-lactide and ε-caprolactone. Makromol Chem 194:913–925
Shen Y, Zhiquan KJ, Yao K-M (1996) Synthesis and characterization of highly random copolymer of ε-caprolactone and d, l-lactide using rare earth catalyst. J Polym Sci Part A Polym Chem 34:1799–1805
Baimark Y, Molloy R (2004) Synthesis and characterization of poly(l-lactide-co-ε-lactone) copolymers: effects of stannous octoate initiator and diethylene glycol coinitiator concentrations. Sci Asia 30:327–334
Yu TL, Wu CC, Chen CC, Huang BH, Wu J, Lin CC (2005) Catalysts for the ring-opening polymerization of ε-caprolactone and l-lactide and the mechanistic study. Polymer 46:5909–5917
Fernández J, Etxeberria A, Sarasua J-R (2012) Synthesis, structure and properties of poly(l-lactide-co-ε-caprolactone) statistical copolymers. J Mech Behav Biomed Mater 9:100–112
Darensbourg DJ, Karroonnirun O (2010) Ring-opening polymerization of l-lactide and ε-caprolactone utilizing biocompatible zinc catalysts. Random copolymerization of l-lactide and ε-caprolactone. Macromolecules 43:8880–8886
Chen YJ, Fang HJ, Hsu SCN, Jheng NY, Chang HC, Ou SW, Peng WT, Lai YC, Chen JY, Chen PL, Kao CH, Zeng ZX, Chen JL, Chen HY (2013) Improving the ring-opening polymerization of ε-caprolactone and l-lactide using stannous octanoate. Polym Bull 70:993–1001
Florczak M, Duda A (2008) Effect of the configuration of the active center on comonomer reactivities: the case of ε-caprolactone/l, l- lactide copolymerization. Angew Chem Int Ed 47:9088–9091
Nomura N, Akita A, Ishii R, Mizuno M (2010) Random copolymerization of ε-caprolactone with lactide using a homosalen—Al complex. J Am Chem Soc 132:1750–1751
Pappalardo D, Annunziata L, Pellecchia C (2009) Living ring-opening homo- and copolymerization of ε-caprolactone and l- and d, l-lactides by dimethyl(salicylaldiminato)aluminum compounds. Macromolecules 42:6056–6062
Li G, Lamberti M, Pappalardo D, Pellecchia C (2012) Random copolymerization of ε-caprolactone and lactides promoted by pyrrolylpyridylamido aluminum complexes. Macromolecules 45:8614–8620
Maruta Y, Abiko A (2014) unpublished results
Maruta Y, Abiko A (2014) submitted for publication
Kubo M, Nakanishi M, Kimura M (1980) Lactone polyesters. DE Patent 2,947,978, 12 Jun 1980
Báez JE, Martínez-Richa A (2005) Synthesis and characterization of poly(ε-caprolactone) and copolyesters by catalysis with molybdenum compounds: polymers with acid-functional asymmetric telechelic architecture. Polymer 46:12118–12129
Dakshinamoorthy D, Peruch F (2012) Block and random copolymerization of ε-caprolactone, l-, and rac-lactide using titanium complex derived from aminodiol ligand. J Polym Sci Part A Polym Chem 50:2161–2171
Kasperczyk J, Bero M (1991) Coordination polymerization of lactides, 2. Microstructure determination of poly[(l, l-lactide)-co-(ε-caprolactone)] with 13C nuclear magnetic resonance spectroscopy. Makromol Chem 192:1777–1787
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Maruta, Y., Abiko, A. Random copolymerization of ε-caprolactone and l-lactide with molybdenum complexes. Polym. Bull. 71, 989–999 (2014). https://doi.org/10.1007/s00289-014-1106-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-014-1106-5