Abstract.
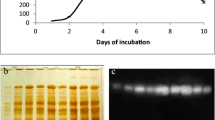
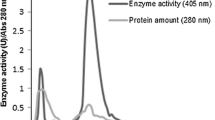
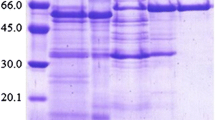
Xylaria regalis, a wood-grown ascomycete isolated in Taiwan, produces β-glucosidase (EC 3.2.1.21) extracellularly. The β-glucosidase was purified to homogeneity by ammonium sulfate precipitation, ion-exchange, and gel filtration chromatography. The molecular mass of the purified enzyme was estimated to be 85 kDa by sodium dodecyl sulfate–polyacrylamide gel electrophoresis. With p-nitrophenyl β-D-glucopyranoside (PNPG) as the substrate at pH 5.0 and 50°C, the K m was 1.72 mM and V max was 326 μmol/min/mg. Optimal activity with PNPG as the substrate was at pH 5.0 and 50°C. The enzyme was stable at pH 5.0 at temperatures up to 50°C. The purified β-glucosidase was active against PNPG, cellobiose, sophorose, and gentiobiose, but did not hydrolyze lactose, sucrose, Avicel, and o-nitrophenyl β-D-galactopyranoside. The activity of β-glucosidase was stimulated by Ca2+, Mg2+, Mn2+, Cd2+ and β-mercaptoethanol, and inhibited by Ag+, Hg2+, SDS, and p-chloromercuribenzoate (PCMB).
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 30 March 1996 / Accepted: 3 May 1996
Rights and permissions
About this article
Cite this article
Wei, DL., Kirimura, K., Usami, S. et al. Purification and Characterization of an Extracellular β-Glucosidase from the Wood-Grown Fungus Xylaria regalis . Curr Microbiol 33, 297–301 (1996). https://doi.org/10.1007/s002849900117
Issue Date:
DOI: https://doi.org/10.1007/s002849900117