Abstract
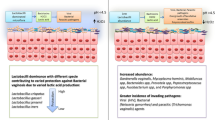
The cervicovaginal microbiota is an essential aspect of women’s reproductive and overall health. In this study, we aimed to evaluate the probiotic properties of a cervicovaginal isolate, obtained from a gynecologically healthy woman and assess its antagonistic effect against various microorganisms isolated from the vagina. Cytological examination was performed using Papanicolaou staining, and the isolated microorganism was identified via 16S Ribosomal RNA Gene Sequence Analysis. Probiotic characteristics were evaluated by determining the tolerance of the isolate to low pH, different NaCl concentrations, and bile salts. Bacterial adherence to stainless steel sheets, antibiotic susceptibility, and antimicrobial activity tests were also conducted and analyzed. Antimicrobial tests and antagonistic activities were assessed through disc diffusion assays. The cervicovaginal isolate was identified as B. velezensis ON116948 and was found to be tolerant to low pH, high NaCl and 0.3% bile salts. Additionally, it exhibited adherence. With the exception of amoxicillin/clavulanic acid (AMC) (30 μg) and oxacillin (OX) (1 μg), this isolate was susceptible to all the antibiotics tested. Candida species did not grow on B. velezensis spread media, while B. velezensis was able to grow on C. albicans, C. glabrata, C. tropicalis, S. condimenti and S. epidermidis spread media with growth zones of 13.7 ± 0.6, 13.3 ± 0.6, 14.2 ± 4.4, 10.5 ± 0.5 and 16.0 ± 1.0 (around discs), respectively. Our findings suggest that the cervicovaginal B. velezensis ON116948 isolate exhibits probiotic properties and antagonistic activity. These results provide important insights into the potential use of this isolate as a probiotic for the prevention of vaginal infections.




Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Code Availability
Not applicable.
References
Anahtar MN, Gootenberg DB, Mitchell CM, Kwon DS (2018) Cervicovaginal microbiota and reproductive health: the virtue of simplicity. Cell Host Microbe 23(2):159–168. https://doi.org/10.1016/j.chom.2018.01.013
Gaspar C, Donders GG, Palmeira-de-Oliveira R, Queiroz JA, Tomaz C, Martinez-de-Oliveira J, Palmeira-de-Oliveira A (2018) Bacteriocin production of the probiotic Lactobacillus acidophilus KS400. AMB Expr. https://doi.org/10.1186/s13568-018-0679-z
Barrientos-Durán A, Fuentes-López A, de Salazar A, Plaza-Díaz J, García F (2020) Reviewing the composition of vaginal microbiota: inclusion of nutrition and probiotic factors in the maintenance of eubiosis. Nutrients 12(2):1–30. https://doi.org/10.3390/nu12020419
Chen X, Huang H, Zhang S, Zhang Y, Jiang J, Qiu Y, Liu J, Wang A (2021) Bacillus velezensis wz-37, a new broad-spectrum biocontrol strain, promotes the growth of tomato seedlings. Agriculture 11(7):1–14. https://doi.org/10.3390/agriculture11070581
Kumherová M, Veselá K, Kosová M, Mašata J, Horáčková Š, Šmidrkal J (2021) Novel potential probiotic Lactobacilli for prevention and treatment of vulvovaginal infections. Probiotics Antimicrob Prot 13(1):163–172. https://doi.org/10.1007/s12602-020-09675-2
Donmez HG, Cagan M, Fadiloglu E, Unal C, Onder SC, Beksac MS (2020) Is bacterial vaginosis associated with autoimmune antibody positivity? Cytopathology 4:298–302. https://doi.org/10.1111/cyt.12846
Donmez HG, Sahal G, Akgor U, Cagan M, Ozgul N, Beksac MS (2020) The relationship between the presence of HPV infection and biofilm formation in cervicovaginal smears. Infection 48(5):735–740. https://doi.org/10.1007/s15010-020-01478-5
Miller EA, Beasley DAE, Dunn RR, Archie EA (2016) Lactobacilli dominance and vaginal pH: why is the human vaginal microbiome unique? Front Microbiol 7:1–13. https://doi.org/10.3389/fmicb.2016.01936
Ravel J, Gajer P, Abdo Z, Schneider GM, Koenig SS, McCulle SL, Karlebach S, Gorle R, Russell J, Tacket CO, Brotman RM, Davis CC, Ault K, Peralta L, Forney LJ (2011) Vaginal microbiome of reproductive-age women. Proc Natl Acad Sci USA 108(1):4680–4687. https://doi.org/10.1073/pnas.1002611107
Jang SJ, Lee K, Kwon B, You HJ, Ko GP (2019) Vaginal lactobacilli inhibit growth and hyphae formation of Candida albicans. Sci Rep 9(1):1–9. https://doi.org/10.1038/s41598-019-44579-4
Rabbee MF, Sarafat Ali M, Choi J, Hwang BS, Jeong SC, Baek K (2019) Bacillus velezensis: a valuable member of bioactive molecules within plant microbiomes. Molecules 24(6):1–13. https://doi.org/10.3390/molecules24061046
Khalid F, Khalid A, Fu Y, Hu Q, Zheng Y, Khan S, Wang Z (2021) Potential of Bacillus velezensis as a probiotic in animal feed: a review. J Microbiol 59(7):627–633. https://doi.org/10.1007/s12275-021-1161-1
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10(3):512–526. https://doi.org/10.1093/oxfordjournals.molbev.a040023
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Mulaw G, Sisay Tessema T, Muleta D, Tesfaye A (2019) In vitro evaluation of probiotic properties of lactic acid bacteria isolated from some traditionally fermented Ethiopian food products. Int J Microbiol 2019:7179514. https://doi.org/10.1155/2019/7179514
Nath S, Sikidar J, Roy M, Deb B (2020) In vitro screening of probiotic properties of Lactobacillus plantarum isolated from fermented milk product. Food Qual Saf 4(4):213–223. https://doi.org/10.1093/fqsafe/fyaa026
Sanhueza E, Paredes-Osses E, González CL, García A (2015) Effect of PH in the survival of Lactobacillus salivarius strain UCO_979C wild type and the Ph acid acclimated variant. Electron J Biotechnol 18(5):343–346. https://doi.org/10.1016/j.ejbt.2015.06.005
Thakkar P, Modi HA, Prajapati JB (2015) Isolation, characterization and safety assessment of lactic acid bacterial ısolates from fermented food products. Int J Curr Microbiol Appl Sci 4(4):713–725
Kang M, Su X, Yun L, Shen Y, Feng J, Yang G, Meng X, Zhang J, Chang X (2022) Evaluation of probiotic characteristics and whole genome analysis of Bacillus velezensis R-71003 isolated from the intestine of common carp (Cyprinus carpio L.) for its use as a probiotic in aquaculture. Aquacult Rep. https://doi.org/10.1016/j.aqrep.2022.101254
Sahal G, Bilkay IS (2015) Multidrug resistance by biofilm-forming clinical strains of Proteus mirabilis. Asian Biomed 9(4):535–554. https://doi.org/10.5372/1905-7415.0904.424
Sahal G, Bilkay IS (2018) Distribution of clinical isolates of Candida spp. and antifungal susceptibility of high biofilm-forming Candida isolates. Rev Soc Bras Med Trop 51(5):644–650. https://doi.org/10.1590/0037-8682-0136-2018
Sahal G, Bilkay IS (2014) Multi drug resistance in strong biofilm forming clinical isolates of Staphylococcus epidermidis. Braz J Microbiol 45(2):539–544. https://doi.org/10.1590/s1517-83822014005000042
Handalishy II, Behery MA, Elkhouly M (2014) Comparative study between probiotic vaginal tampons and oral metronidazole in treatment of bacterial vaginosis. Al-Azhar Assiut Med j 12:185–203
Lewis FMT, Bernstein KT, Aral SO (2017) Vaginal microbiome and its relationship to behavior, sexual health, and sexually transmitted diseases. Obstet Gynecol 129(4):643–654. https://doi.org/10.1097/AOG.0000000000001932
Freire AD, Custódio AI, Filho JQ, Freitas JC, Gonçalves AK, Cobucci RN (2020) The association between abnormal vaginal flora and cytological evidence of HPV with prematurity in high-risk pregnant women. Gynecol Obstet Reprod Med 26(3):173–178
Tortelli BA, Lewis WG, Allsworth JE, Member-Meneh N, Foster LR, Reno HE, Peipert JF, Fay JC, Lewis AL (2020) Associations between the vaginal microbiome and Candida colonization in women of reproductive age. Am J Obstet Gynecol 222(5):471.e1-471.e9. https://doi.org/10.1016/j.ajog.2019.10.008
Curty G, de Carvalho PS, Soares MA (2020) The role of the cervicovaginal microbiome on the genesis and as a biomarker of premalignant cervical intraepithelial neoplasia and invasive cervical cancer. Int J Mol Sci 21(1):222. https://doi.org/10.3390/ijms21010222
Goldstein EJC, Tyrrell KL, Citron DM (2015) Lactobacillus species: taxonomic complexity and controversial susceptibilities. Clin Infect Dis 60(2):S98–S107. https://doi.org/10.1093/cid/civ072
Fan B, Wang C, Song X, Ding X, Wu L, Wu H, Gao X, Borriss R (2018) Bacillus velezensis FZB42 in 2018 the gram-positive model strain for plant growth promotion and biocontrol. Front Microbiol 9:2491. https://doi.org/10.3389/fmicb.2018.02491
Jiang CH, Liao MJ, Wang HK, Zheng MZ, Xu JJ, Guo JH (2018) Bacillus velezensis, a potential and efficient biocontrol agent in control of pepper gray mold caused by Botrytis cinerea. Biol Control 12:147–157. https://doi.org/10.1016/j.biocontrol.2018.07.017
Yi Y, Zhang Z, Zhao F, Liu H, Yu L, Zha J, Wang G (2018) Probiotic potential of Bacillus velezensis JW: antimicrobial activity against fish pathogenic bacteria and immune enhancement effects on Carassius auratus. Fish Shellfish Immunol 78:322–330. https://doi.org/10.1016/j.fsi.2018.04.055
Pandin C, Darsonval M, Mayeur C, Le Coq D, Aymerich S, Briandet R (2019) Biofilm formation and synthesis of Antimicrobial compounds by the biocontrol agent Bacillus velezensis QST713 in an Agaricus bisporus compost micromodel. Appl Environ Microbiol 85(12):e00327-e419. https://doi.org/10.1128/AEM.00327-19
Rabbee MF, Hwang BS, Baek KH (2023) Bacillus velezensis: a beneficial biocontrol agent or facultative phytopathogen for sustainable agriculture. Agronomy 13(3):840. https://doi.org/10.3390/agronomy13030840
Yuan H, Shi B, Wang L, Huang T, Zhou Z, Hou H, Tu H (2022) Isolation and characterization of Bacillus velezensis strain P2–1 for biocontrol of apple postharvest decay caused by Botryosphaeria dothidea. Front Microbiol 12:808938. https://doi.org/10.3389/fmicb.2021.808938
Thurlow CM, Williams MA, Carrias A, Ran C, Newman M, Tweedie J, Allison E, Jescovitch LN, Wilson AE, Terhune JS, Liles MR (2019) Bacillus velezensis AP193 exerts probiotic effects in channel catfish (Ictalurus punctatus) and reduces aquaculture pond eutrophication. Aquaculture 503:347–356. https://doi.org/10.1016/j.aquaculture.2018.11.051
Fooks LJ, Gibson GR (2002) In vitro investigations of the effect of probiotics and prebiotics on selected human intestinal pathogens. FEMS Microbiol Ecol 9(1):67–75. https://doi.org/10.1016/S0168-6496(01)00197-0
Torres-Sánchez A, Pardo-Cacho J, López-Moreno A, Ruiz-Moreno Á, Cerk K, Aguilera M (2021) Antimicrobial effects of potential probiotics of Bacillus spp. isolated from human microbiota: in vitro and in silico methods. Microorganisms 9(8):1615. https://doi.org/10.3390/microorganisms9081615
Borah T, Gogoi B, Khataniar A, Gogoi M, Das A, Borah D (2019) Probiotic characterization of indigenous Bacillus velezensis strain DU14 isolated from Apong, a traditionally fermented rice beer of Assam. Biocatal Agric Biotechnol 18:101008. https://doi.org/10.1016/j.bcab.2019.01.046
Prabhurajeshwar C, Chandrakanth RK (2017) Probiotic potential of Lactobacilli with antagonistic activity against pathogenic strains: an in vitro validation for the production of inhibitory substances. Biomed J 40(5):270–283. https://doi.org/10.1016/j.bj.2017.06.008
Dunne C, O’Mahony L, Murphy L, Thornton G, Morrissey D, O’Halloran S, Feeney M, Flynn S, Fitzgerald G, Daly C, Kiely B, O’Sullivan GC, Shanahan F, Collins JK (2001) In vitro selection criteria for probiotic bacteria of human origin: correlation with in vivo findings. Am J Clin Nutr 73(2):386S-392S. https://doi.org/10.1093/ajcn/73.2.386s
Mandal S, Puniya AK, Singh K (2006) Effect of alginate concentrations on survival of microencapsulated Lactobacillus casei NCDC-298. Int Dairy J 16(10):1190–1195. https://doi.org/10.1016/j.idairyj.2005.10.005
Darmastuti A, Hasan PN, Wikandari R, Utami T, Rahayu ES, Suroto DA (2021) Adhesion properties of Lactobacillus plantarum Dad-13 and Lactobacillus plantarum Mut-7 on Sprague Dawley rat intestine. Microorganisms 9(11):2336. https://doi.org/10.3390/microorganisms9112336
Feito J, Contente D, Ponce-Alonso M, Díaz-Formoso L, Araújo C, Peña N, Borrero J, Gómez-Sala B, Del Campo R, Muñoz-Atienza E, Hernández PE, Cintas LM (2022) Draft genome sequence of Lactococcus lactis subsp. cremoris WA2–67: a promising Nisin-producing probiotic strain isolated from the rearing environment of a Spanish rainbow trout (Oncorhynchus mykiss, Walbaum) farm. Microorganisms 10(3):521. https://doi.org/10.3390/microorganisms10030521
Kamble A, Naik S, Talathi M, Jadhav D, Pingale S, Kaul-Ghanekar R (2022) Cervicovaginal microbiota isolated from healthy women exhibit probiotic properties and antimicrobial activity against pathogens isolated from cervical cancer patients. Arch Microbiol 204(8):491. https://doi.org/10.1007/s00203-022-03103-5
Ranjit E, Raghubanshi BR, Maskey S, Parajuli P (2018) Prevalence of bacterial vaginosis and its association with risk factors among nonpregnant women: a hospital based study. Int J Microbiol 2018:8349601. https://doi.org/10.1155/2018/8349601
Zhang W, Guo H, Cao C, Li L, Kwok LY, Zhang H, Sun Z (2017) Adaptation of Lactobacillus casei Zhang to gentamycin involves an alkaline shock protein. Front Microbiol 8:2316. https://doi.org/10.3389/fmicb.2017.02316
Mancabelli L, Mancino W, Lugli GA, Argentini C, Longhi G, Milani C, Viappiani A, Anzalone R, Bernasconi S, van Sinderen D, Ventura M, Turroni F (2021) Amoxicillin-clavulanic acid resistance in the genus Bifidobacterium. Appl Environ Microbiol 87(7):e03137-e3220. https://doi.org/10.1128/AEM.03137-20
Duche RT, Singh A, Wandhare AG, Sangwan V, Sihag MK, Nwagu TNT, Panwar H, Ezeogu LI (2023) Antibiotic resistance in potential probiotic lactic acid bacteria of fermented foods and human origin from Nigeria. BMC Microbiol 23:142. https://doi.org/10.1186/s12866-023-02883-0
Ozturk S, Erbas G (2017) Investigation of antibiotic sensivity, isolation and identification of Gardnerella vaginalis collected from Ketem/Aydın Province. Kocatepe Med J 18:61–66
Devi S, Kiesewalter HT, Kovács R, Frisvad JC, Weber T, Larsen TO, Kovács ÁT, Ding L (2019) Depiction of secondary metabolites and antifungal activity of Bacillus velezensis DTU001. Synth Syst Biotechnol 4(3):142–149. https://doi.org/10.1016/j.synbio.2019.08.002
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
Conceptualization: GS, HGD, MSB; Sample collection: HGD and MSB; Investigation: GS and HGD; Data analysis: GS and HGD; Writing—first draft: GS and HGD; Supervision: MSB; Review and editing: MSB. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no relevant financial or non-financial interests to disclose.
Ethical Approval
This study was approved by “Hacettepe University Ethics Committee” (GO19/507).
Informed Consent
The written informed consent was obtained from all individual participants in accordance with the principles of the declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sahal, G., Donmez, H.G. & Beksac, M.S. Cervicovaginal Bacillus velezensis Isolate: A Potential Probiotic and an Antagonist Against Candida and Staphylococcus. Curr Microbiol 80, 332 (2023). https://doi.org/10.1007/s00284-023-03447-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03447-1