Abstract
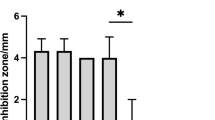
Bacterial disease is one of the most critical problems in aquaculture. Probiotics represent a promising biological approach to control bacterial disease because it is effective against pathogens and environmentally friendly. This study assessed the antagonistic activities of a bacterial strain FIO1408 isolated from deep-sea water against many pathogenic bacteria in aquaculture, including Listonella anguillarum, Vibrio parahaemolyticus, Vibrio alginolyticus, Aeromonas hydrophila, Edwardsiella anguillarum, Edwardsiella tarda, and Edwardsiella piscicida. The complete genome of strain FIO1408 consisted of a circular chromosome of 4,137,639 bp and two plasmids of 16,439 bp and 24,472 bp. Phylogenetic analysis showed strain FIO1408 clustered with Bacillus velezensis strains. 12 genes/gene clusters responsible for the synthesis of secondary metabolites were identified in the FIO1408 genome, including three lipopeptides, three polyketides, three bacteriocins, one siderophore, one dipeptide, and one unknown type. Also identified were 273 unique orthologous genes primarily involved in phage resistance, protein hydrolysis, environmental survivability, and genetic stability compared to B. velezensis KACC 13105, B. velezensis FZB42T, and B. velezensis NRRL B-41580. The principal safety of FIO1408 was demonstrated by genetic analyses and feeding trials. These findings will contribute to studies on the biocontrol mechanisms of B. velezensis FIO1408 and facilitate its application as a potent biological control agent against bacterial pathogens in aquaculture.





Similar content being viewed by others
Data Availability
The genome sequences of B. velezensis FIO1408 have been deposited in NCBI under the GenBank accession numbers CP061938-CP061940 (BioProject: PRJNA664348). The B. velezensis FIO1408 strain is available from the Key laboratory of marine bioactive substances, First Institute of Oceanology, Ministry of Natural Resources, China.
References
Natrah FMI, Defoirdt T, Sorgeloos P, Bossier P (2011) Disruption of bacterial cell-to-cell communication by marine organisms and its relevance to aquaculture. Mar Biotechnol 13(2):109–126. https://doi.org/10.1007/s10126-010-9346-3
Ringø E, Van Doan H, Lee SH, Soltani M, Hoseinifar SH, Harikrishnan R, Song SK (2020) Probiotics, lactic acid bacteria and bacilli: interesting supplementation for aquaculture. J Appl Microbiol 129(1):116–136. https://doi.org/10.1111/jam.14628
Pérez-Sánchez T, Mora-Sánchez B, Balcázar JL (2018) Biological approaches for disease control in aquaculture: advantages limitations and challenges. Trends Microbiol 26(11):896–903. https://doi.org/10.1016/j.tim.2018.05.002
Cabello FC, Godfrey HP, Tomova A, Ivanova L, Dölz H, Millanao A, Buschmann AH (2013) Antimicrobial use in aquaculture re-examined: its relevance to antimicrobial resistance and to animal and human health. Environ Microbiol 15(7):1917–1942. https://doi.org/10.1111/1462-2920.12134
Banerjee G, Ray AK (2017) The advancement of probiotics research and its application in fish farming industries. Res Vet Sci 115:66–77. https://doi.org/10.1016/j.rvsc.2017.01.016
de Bruijn I, Liu Y, Wiegertjes GF, Raaijmakers JM (2018) Exploring fish microbial communities to mitigate emerging diseases in aquaculture. FEMS Microbiol Ecol. https://doi.org/10.1093/femsec/fix161
Olmos J, Acosta M, Mendoza G, Pitones V (2020) Bacillus subtilis, an ideal probiotic bacterium to shrimp and fish aquaculture that increase feed digestibility, prevent microbial diseases, and avoid water pollution. Arch Microbiol 202:427–435. https://doi.org/10.1007/s00203-019-01757-2
Kuebutornye FKA, Abarike ED, Lu Y, Hlordzi V, Sakyi ME, Afriyie G, Wang Z, Li Y, Xie C (2020) Mechanisms and the role of probiotic bacillus in mitigating fish pathogens in aquaculture. Fish Physiol Biochem 46(3):819–841. https://doi.org/10.1007/s10695-019-00754-y
Zhao X, Kuipers OP (2016) Identification and classification of known and putative antimicrobial compounds produced by a wide variety of bacillales species. BMC Genomics 17(1):882. https://doi.org/10.1186/s12864-016-3224-y
Zhang D, Gao Y, Wang Y, Liu C, Cunbin S (2020) Advances in taxonomy, antagonistic function and application of Bacillus velezensis. Microbiol China 47(11):3634–3649. https://doi.org/10.13344/j.microbiol.china.190947
Yi Y, Zhang Z, Zhao F, Liu H, Yu L, Zha J, Wang G (2018) Probiotic potential of Bacillus Velezensis JW: antimicrobial activity against fish pathogenic bacteria and immune enhancement effects on Carassius Auratus. Fish Shellfish Immunol 78:322–330. https://doi.org/10.1016/j.fsi.2018.04.055
Ye M, Tang X, Yang R, Zhang H, Li F, Tao F, Li F, Wang Z (2018) Characteristics and application of a novel species of bacillus: Bacillus Velezensis. ACS Chem Biol 13(3):500–505. https://doi.org/10.1021/acschembio.7b00874
Zhang D, Gao Y, Ke X, Yi M, Liu Z, Han X, Shi C, Lu M (2019) Bacillus Velezensis LF01 in vitro antimicrobial activity against fish pathogens, growth performance enhancement, and disease resistance against streptococcosis in Nile Tilapia (Oreochromis Niloticus). Appl Microbiol Biotechnol 103(2):9023–9035. https://doi.org/10.1007/s00253-019-10176-8
Eid J, Fehr A, Gray J, Luong K, Lyle J, Otto G, Peluso P, Rank D, Baybayan P, Bettman B et al (2009) Real-time DNA sequencing from single polymerase molecules. Science 323(5910):133–138. https://doi.org/10.1126/science.1162986
Besemer J, Lomsadze A, Borodovsky M (2001) GeneMarkS: a self-training method for prediction of gene starts in microbial genomes implications for finding sequence motifs in regulatory regions. Nucleic Acids Res 29(12):2607–2618. https://doi.org/10.1093/nar/29.12.2607
Lowe TM, Eddy SR (1997) TRNAscan-SE: a program for improved detection of transfer RNA genes in genomic sequence. Nucleic Acids Res 25(5):955–964. https://doi.org/10.1093/nar/25.5.955
Lagesen K, Hallin P, Rødland EA, Staerfeldt H-H, Rognes T, Ussery DW (2007) RNAmmer: consistent and rapid annotation of ribosomal RNA genes. Nucleic Acids Res 35(9):3100–3108. https://doi.org/10.1093/nar/gkm160
Hsiao W, Wan I, Jones SJ, Brinkman FSL (2003) IslandPath: aiding detection of genomic islands in prokaryotes. Bioinformatics 19(3):418–420. https://doi.org/10.1093/bioinformatics/btg004
Zhou Y, Liang Y, Lynch KH, Dennis JJ, Wishart DS (2011) PHAST: a fast phage search tool. Nucleic Acids Res 39(suppl):W347–W352. https://doi.org/10.1093/nar/gkr485
Grissa I, Vergnaud G, Pourcel C (2007) CRISPRFinder: a web tool to identify clustered regularly interspaced short palindromic repeats. Nucleic Acids Res 35:W52–W57. https://doi.org/10.1093/nar/gkm360
Meier-Kolthoff JP, Göker M (2019) TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat Commun 10(1):2182. https://doi.org/10.1038/s41467-019-10210-3
Na S-I, Kim YO, Yoon S-H, Ha S, Baek I, Chun J (2018) UBCG: up-to-date bacterial core gene set and pipeline for phylogenomic tree reconstruction. J Microbiol 56(4):280–285. https://doi.org/10.1007/s12275-018-8014-6
Yoon S-H, Ha S-M, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67(5):1613–1617. https://doi.org/10.1099/ijsem.0.001755
Weber T, Blin K, Duddela S, Krug D, Kim HU, Bruccoleri R, Lee SY, Fischbach MA, Müller R, Wohlleben W (2015) AntiSMASH 3.0—a comprehensive resource for the genome mining of biosynthetic gene clusters. Nucleic Acids Res 43(W1):W237–W243. https://doi.org/10.1093/nar/gkv437
de Jong A, Van Hijum SAFT, Bijlsma JJE, Kok J, Kuipers OP (2006) BAGEL: a web-based bacteriocin genome mining tool. Nucleic Acids Res 34:W273–W279. https://doi.org/10.1093/nar/gkl237
Emms DM, Kelly S (2019) OrthoFinder: phylogenetic orthology inference for comparative genomics. Genome Biol 20(1):238. https://doi.org/10.1186/s13059-019-1832-y
Bardou P, Mariette J, Escudié F, Djemiel C, Klopp C (2014) Jvenn: an interactive venn diagram viewer. BMC Bioinformatics 15(1):293. https://doi.org/10.1186/1471-2105-15-293
Jacques P (2011) Surfactin and Other Lipopeptides from Bacillus spp. In: Jacques Philippe (ed) Biosurfactants. Springer, Berlin, pp 57–91. https://doi.org/10.1007/978-3-642-14490-5_3
Scholz R, Vater J, Budiharjo A, Wang Z, He Y, Dietel K, Schwecke T, Herfort S, Lasch P, Borriss R (2014) Amylocyclicin, a novel circular bacteriocin produced by bacillus Amyloliquefaciens FZB42. J Bacteriol 196(10):1842–1852. https://doi.org/10.1128/JB.01474-14
Dias L, Caetano T, Pinheiro M, Mendo S (2015) The lanthipeptides of bacillus methylotrophicus and their association with genomic islands. Syst Appl Microbiol 38(8):525–533. https://doi.org/10.1016/j.syapm.2015.10.002
Kuebutornye FKA, Abarike ED, Lu Y (2019) A review on the application of bacillus as probiotics in aquaculture. Fish Shellfish Immunol 87:820–828. https://doi.org/10.1016/j.fsi.2019.02.010
Ina-Salwany MY, Al-Saari N, Mohamad A, Mursidi F-A, Mohd-Aris A, Amal MNA, Kasai H, Mino S, Sawabe T, Zamri-Saad M (2019) Vibriosis in fish: a review on disease development and prevention. J Aquat Anim Health 31(1):3–22. https://doi.org/10.1002/aah.10045
Rasmussen-Ivey CR, Figueras MJ, McGarey D, Liles MR (2016) Virulence factors of Aeromonas Hydrophila: in the wake of reclassification. Fron Microbiol. https://doi.org/10.3389/fmicb.2016.01337
Buján N, Mohammed H, Balboa S, Romalde JL, Toranzo AE, Arias CR, Magariños B (2018) Genetic studies to re-affiliate Edwardsiella Tarda fish isolates to Edwardsiella Piscicida and Edwardsiella Anguillarum species. Syst Appl Microbiol 41(1):30–37. https://doi.org/10.1016/j.syapm.2017.09.004
Fazle RM, Baek K-H (2020) Antimicrobial activities of lipopeptides and polyketides of bacillus velezensis for agricultural applications. Molecules (Basel, Switzerland) 25(21):E4973. https://doi.org/10.3390/molecules25214973
Rabbee M, Ali Md, Choi J, Hwang B, Jeong S, Baek K (2019) Bacillus Velezensis: a valuable member of bioactive molecules within plant microbiomes. Molecules 24(6):1046. https://doi.org/10.3390/molecules24061046
Özcengiz G, Öğülür İ (2015) Biochemistry, genetics and regulation of bacilysin biosynthesis and its significance more than an antibiotic. New Biotechnol 32(6):612–619. https://doi.org/10.1016/j.nbt.2015.01.006
Lv J, Da R, Cheng Y, Tuo X, Wei J, Jiang K, Monisayo AO, Han B (2020) Mechanism of antibacterial activity of Bacillus Amyloliquefaciens C-1 lipopeptide toward anaerobic clostridium difficile. Biomed Res Int 2020:1–12. https://doi.org/10.1155/2020/3104613
Kröber M, Verwaaijen B, Wibberg D, Winkler A, Pühler A, Schlüter A (2016) Comparative transcriptome analysis of the biocontrol strain bacillus Amyloliquefaciens FZB42 as response to biofilm formation analyzed by RNA sequencing. J Biotechnol 231:212–223. https://doi.org/10.1016/j.jbiotec.2016.06.013
Wang W, Li J, Chi H (2021) Purification and antimicrobial mechanism of amylocyclicin W5 produced by bacillus amyloliquefaciens DH8030. Food Sci 42(07):29–34. https://doi.org/10.7506/spkx1002-6630-20200407-082
Brotz H, Bierbaum G, Markus A, Molitor E, Sahl HG (1995) Mode of action of the Lantibiotic Mersacidin: inhibition of peptidoglycan biosynthesis via a novel mechanism? Antimicrob Agents Chemother 39(3):714–719. https://doi.org/10.1128/AAC.39.3.714
Goldfarb T, Sberro H, Weinstock E, Cohen O, Doron S, Charpak-Amikam Y, Afik S, Ofir G, Sorek R (2015) BREX is a novel phage resistance system widespread in microbial genomes. EMBO J 34(2):169–183. https://doi.org/10.15252/embj.201489455
Ferrières L, Aslam SN, Cooper RM, Clarke DJ (2007) The yjbEFGH locus in Escherichia coli K-12 is an operon encoding proteins involved in exopolysaccharide production. Microbiol (Reading, England) 153(Pt 4):1070–1080. https://doi.org/10.1099/mic.0.2006/002907-0
Li M, Feng Y, Gao Y, Wu Q (2007) Glutaredoxin system and its regulation to the cytosolic thiol-redox status. Acta Biophysica Sinica 23(5):343–350. https://doi.org/10.3321/j.issn:1000-6737.2007.05.004
Pilo P, Frey J (2018) Pathogenicity, population genetics and dissemination of Bacillus anthracis. Infect Genet Evol 64:115–125. https://doi.org/10.1016/j.meegid.2018.06.024
Ehling-Schulz M, Lereclus D, Koehler TM (2019) The Bacillus cereus group: Bacillus species with pathogenic potential. Microbiol Spectr. https://doi.org/10.1128/microbiolspec.GPP3-0032-2018
Wang G, Li X, Wang Z (2016) APD3: The Antimicrobial Peptide Database as a Tool for Research and Education. Nucleic Acids Res 44(D1):D1087–D1093. https://doi.org/10.1093/nar/gkv1278
Adeniji AA, Aremu OS, Babalola OO (2019) Selecting lipopeptide-producing, fusarium-suppressing bacillus Spp: metabolomic and genomic probing of bacillus velezensis NWUMFkBS105. MicrobiologyOpen 8(6):e00742. https://doi.org/10.1002/mbo3.742
Acknowledgements
This work was supported by Laoshan Laboratory (LSKJ202203901) and the National Natural Science Foundation of China (Grant Number 42106153).
Author information
Authors and Affiliations
Contributions
WH and LQ: conceived the idea of the study; WHu and PG: analyzed the data; WH and PG and GD: interpreted the results; WH: wrote the paper; all authors discussed the results and revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
All procedures performed in the study were in accordance with the Laboratory Animal Welfare and Ethics Committee CALAS, Beijing, China. The ethical principles (GB/T 35892–2018) established by General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China were followed. We have obtained permission from the National Key Basic Research Program of China for the provision of infrastructure and support for rearing of Scophthalmus maximus (an aquaculture fish species) in indoor conditions related to our study to promote aquaculture practices. During the experiment, the rearing method simulated the aquaculture in a flowing water environment (no harm to S. maximus) and the probiotic supplementation was conducted through feed addition as a modest method. At the end of the experiment, all fish were euthanized according to Guideline for ethical review of animal welfare (GB/T 35892-2018).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, W., Qu, L., Gao, P. et al. Bioassay and Whole-Genome Analysis of Bacillus velezensis FIO1408, a Biocontrol Agent Against Pathogenic Bacteria in Aquaculture. Curr Microbiol 80, 354 (2023). https://doi.org/10.1007/s00284-023-03423-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-023-03423-9