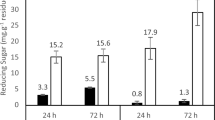
Abstract
Polygalacturonase (PGase) production by Aureobasidium pullulans P56 under submerged fermentation was investigated using agro-industrial wastes and commercial carbon and nitrogen sources. The maximum PGase concentration was equivalent to 8.6 U/mL that was obtained in presence of citrus pectin at 150 rpm, 30 °C, pH = 5.5, and 60 h of fermentation conditions. However, a significant amount of enzyme production was also recorded upon the utilization of corncob (5.3 U/mL) and wheat bran (4.4 U/mL) as carbon sources. Amongst the different nitrogen sources, the highest enzyme production (8.2 U/mL) was obtained in presence of ammonium sulphate and yeast extract simultaneously at a ratio of 1:1. The enzyme was partially purified by gel filtration using Sephadex G50 equilibrated and washed with 50 mM-sodium acetate buffer. The obtained yield and specific activity were determined equivalent to 17% and 9.53 U/mg, respectively. The molecular weight of the partially purified enzyme was estimated as 54 kDa on SDS-PAGE. The conditions affecting the enzyme activity were determined and the highest enzyme activity was recorded at 40 °C and 4.5 pH. Amongst the tested metal ions, 2 and 5 mM of CaCl2 concentrations increased the enzymatic activity by 30%. Overall, the use of corncob (2.5%) to produce PGase by A. pullulans represents an attractive agro-industrial substrate.





Similar content being viewed by others
Data Availability
The data used to support the findings of this study are available upon reasonable request. A. pullulans p56 has been deposited at the Culture Collection of the Biology Department of the Manisa Celal Bayar University, Turkey with an accession number CCB 012.
Code Availability
Not applicable.
References
Kashyap DR, Vohra PK, Chopra S, Tewari R (2001) Applications of pectinases in the commercial sector: a review. Bioresour Technol 77:215–227. https://doi.org/10.1016/S0960-8524(00)00118-8
Pedrolli DB, Monteiro AC, Gomes E, Carmona EC (2009) Pectin and pectinases: production, characterization and industrial application of microbial pectinolytic enzymes. Open Biotechnol J 3:9–18. https://doi.org/10.2174/1874070700903010009
de Souza TSP, Kawaguti HY (2021) Cellulases, hemicellulases, and pectinases: applications in the food and beverage industry. Food Bioprocess Technol 14:1446–1477. https://doi.org/10.1007/s11947-021-02678-z
Amin F, Bhatti HN, Bilal M, Asgher M (2017) Improvement of activity, thermo-stability and fruit juice clarification characteristics of fungal exo-polygalacturonase. Int J Biol Macromol 95:974–984. https://doi.org/10.1016/j.ijbiomac.2016.10.086
Satapathy S, Rout JR, Kerry RG, Thatoi H, Sahoo SL (2020) Biochemical prospects of various microbial pectinase and pectin: an approachable concept in pharmaceutical bioprocessing. Front Nutr 7:117. https://doi.org/10.3389/fnut.2020.00117
Shrestha S, Rahman MS, Qin W (2021) New insights in pectinase production development and industrial applications. Appl Microbiol Biotechnol 105:9069–9087. https://doi.org/10.1007/s00253-021-11705-0
Sharma N, Rathore M, Sharma M (2013) Microbial pectinase: sources, characterization and applications. Rev Environ Sci Biotechnol 12:45–60. https://doi.org/10.1007/s11157-012-9276-9
Garg G, Singh A, Kaur A, Singh R, Kaur J, Mahajan R (2016) Microbial pectinases: an ecofriendly tool of nature for industries. 3Biotech 6:1–13. https://doi.org/10.1007/s13205-016-0371-4
Thakur A, Pahwa R, Singh S, Gupta R (2010) Production, purification, and characterization of polygalacturonase from Mucor circinelloides ITCC 6025. Enzyme Res. https://doi.org/10.4061/2010/170549
Roosdiana A, Prasetyawan S, Mahdi C, Sutrisno S (2013) Production and characterization of Bacillus firmus pectinase. J Pure Appl Chem Res 2:35–41. https://doi.org/10.21776/ub.jpacr.2013.002.01.111
Merín MG, Mendoza LM, Morata de Ambrosini VI (2014) Pectinolytic yeasts from viticultural and enological environments: Novel finding of Filobasidium capsuligenum producing pectinases. J Basic Microbiol 54:835–842. https://doi.org/10.1002/jobm.201200534
Cooke WB (1959) An ecological life history of Aureobasidium pullulans (de Bary) Arnaud. Mycopathol Mycol Appl 12:1–45. https://doi.org/10.1007/BF02118435
Chi Z, Wang F, Chi Z, Yue L, Liu G, Zhang T (2009) Bioproducts from Aureobasidium pullulans, a biotechnologically important yeast. Appl Microbiol Biotechnol 82:793–804. https://doi.org/10.1007/s00253-009-1882-2
Stratilová E, Dzúrová M, Breierová E, Malovíková A, Omelková J (2006) The life style of Aureobasidium pullulans and the multiple forms of its polygalacturonase. Biologia (Bratisl) 61:257–262. https://doi.org/10.2478/s11756-006-0047-3
Cheng KC, Demirci A, Catchmark JM (2011) Pullulan: biosynthesis, production, and applications. Appl Microbiol Biotechnol 92:29–44. https://doi.org/10.1007/s00253-011-3477-y
Manitchotpisit P, Watanapoksin R, Price NPJ, Bischoff KM, Tayeh M, Teeraworawit S, Kriwong S, Leathers TD (2014) Aureobasidium pullulans as a source of liamocins (heavy oils) with anticancer activity. World J Microbiol Biotechnol 30:2199–2204. https://doi.org/10.1007/s11274-014-1639-7
Federici F (1982) Extracellular enzymatic activities in Aureobasidium pullulans. Mycologia 74:738. https://doi.org/10.2307/3792859
Molnárová J, Vadkertiová R, Stratilová E (2014) Extracellular enzymatic activities and physiological profiles of yeasts colonizing fruit trees. J Basic Microbiol 54(Suppl 1):S74-84. https://doi.org/10.1002/jobm.201300072
Leathers TD, Rich JO, Anderson AM, Manitchotpisit P (2013) Lipase production by diverse phylogenetic clades of Aureobasidium pullulans. Biotechnol Lett 35:1701–1706. https://doi.org/10.1007/s10529-013-1268-5
Bankeeree W, Lotrakul P, Prasongsuk S, Chaiareekij S, Eveleigh DE, Kim SW, Punnapayak H (2014) Effect of polyols on thermostability of xylanase from a tropical isolate of Aureobasidium pullulans and its application in prebleaching of rice straw pulp. Springerplus 3:1–11. https://doi.org/10.1186/2193-1801-3-37
Rich JO, Leathers TD, Anderson AM, Bischoff KM, Manitchotpisit P (2013) Laccases from Aureobasidium pullulans. Enzyme Microb Technol 53:33–37. https://doi.org/10.1016/j.enzmictec.2013.03.015
Manitchotpisit P, Skory CD, Leathers TD, Lotrakul P, Eveleigh DE, Prasongsuk S, Punnapayak H (2011) α-Amylase activity during pullulan production and α-amylase gene analyses of Aureobasidium pullulans. J Ind Microbiol Biotechnol 38:1211–1218. https://doi.org/10.1007/s10295-010-0899-y
Mari M, Martini C, Spadoni A, Rouissi W, Bertolini P (2012) Biocontrol of apple postharvest decay by Aureobasidium pullulans. Postharvest Biol Technol 73:56–62. https://doi.org/10.1016/j.postharvbio.2012.05.014
Roukas T (1999) Pullulan production from brewery wastes by Aureobasidium pullulans. World J Microbiol Biotechnol 15:447–450. https://doi.org/10.1023/A:1008996522115
Göksungur Y, Uçan A, Güvenç U (2004) Production of pullulan from beet molasses and synthetic medium by Aureobasidium pullulans. Turkish J Biol 28:23–30
Göksungur Y, Dǎgbaǧli S, Uçan A, Güvenç U (2005) Optimization of pullulan production from synthetic medium by Aureobasidium pullulans in a stirred tank reactor by response surface methodology. J Chem Technol Biotechnol 80:819–827. https://doi.org/10.1002/jctb.1254
Göksungur Y, Uzunogullari P, Daǧbaǧli S (2011) Optimization of pullulan production from hydrolysed potato starch waste by response surface methodology. Carbohydr Polym 83:1330–1337. https://doi.org/10.1016/j.carbpol.2010.09.047
Oskay M, Yalçin HT (2015) Screening of yeast strains for pectinolytic activity: effects of different carbon and nitrogen sources in submerged fermentations. Online J Biol Sci 15:89–96. https://doi.org/10.3844/ojbsci.2015.89.96
Lan Pham P, Taillandier P, Delmas M, Strehaiano P (1998) Production of xylanases by Bacillus polymyxa using lignocellulosic wastes. Ind Crops Prod 2–3:195–203. https://doi.org/10.1016/S0926-6690(97)00048-4
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Bennett G (1992) Lowry’s handbook of right-to-know emergency planning. J Hazard Mater 30:361–362. https://doi.org/10.1016/0304-3894(92)87011-4
Manachini PL, Parini C, Fortina MG (1988) Pectic enzymes from Aureobasidium pullulans LV 10. Enzyme Microb Technol 10:682–685. https://doi.org/10.1016/0141-0229(88)90060-9
Merín MG, Martín MC, Rantsiou K, Cocolin L, de Ambrosini VIM (2015) Characterization of pectinase activity for enology from yeasts occurring in Argentine bonarda grape. Brazilian J Microbiol 46:815–823. https://doi.org/10.1590/S1517-838246320140160
Sudeep KC, Upadhyaya J, Joshi DR, Lekhak B, Chaudhary DK, Pant BR, Bajgai TR, Dhital R, Khanal S, Koirala N, Raghavan V (2020) Production, characterization, and industrial application of pectinase enzyme isolated from fungal strains. Fermentation 6:59. https://doi.org/10.3390/fermentation6020059
Munir M, Abdullah R, Ul Haq I, Afshan Kaleem A, Iqtedar M, Naz S (2020) Strain improvement by random mutagenesis of Aspergillus tamarii RMLC-10 for improved biosynthesis of polygalacturonase. Pakistan J Bot 52:1809–1813. https://doi.org/10.30848/PJB2020-5(29)
Onetto CA, Borneman AR, Schmidt SA (2020) Investigating the effects of Aureobasidium pullulans on grape juice composition and fermentation. Food Microbiol. https://doi.org/10.1016/j.fm.2020.103451
Šelo G, Planinić M, Tišma M, Tomas S, Komlenić DK, Bucić-Kojić A (2021) A comprehensive review on valorization of agro-food industrial residues by solid-state fermentation. Foods 10:103451. https://doi.org/10.3390/foods10050927
El Gamal NG, Atalla SMM, El-mougy NS, Abdel-Kader MM (2018) Production of pectinase by Saccharomyces cerevisiae and its application as biocontrol agent against navel orange and apple fruits decay. Biosci Res 15:4136–4147
Moresi M, Petruccioli M, Federici F (1991) Modelling of cyclic fed-batch plus batch polygalacturonase production by Aureobasidium pullulans on raw orange peel. Appl Microbiol Biotechnol 34:742–748. https://doi.org/10.1007/BF00169344
Bennamoun L, Hiligsmann S, Dakhmouche S, Ait-Kaki A, Labbani FZK, Nouadri T, Meraihi Z, Turchetti B, Buzzini P, Thonart P (2016) Production and properties of a thermostable, pH-stable exo-polygalacturonase using Aureobasidium pullulans isolated from saharan soil of algeria grown on tomato pomace. Foods 5:1–20. https://doi.org/10.3390/foods5040072
Galiotou-Panayotou M, Kalantzi O, Aggelis G (1998) Modelling of simultaneous production of polygalacturonase and exopolysaccharide by Aureobasidium pullulans ATHUM 2915. Antonie Van Leeuwenhoek 73:155–162. https://doi.org/10.1023/A:1000657403593
Yegin S, Altinel B, Tuluk K (2018) A novel extremophilic xylanase produced on wheat bran from Aureobasidium pullulans NRRL Y-2311-1: effects on dough rheology and bread quality. Food Hydrocoll 81:389–397. https://doi.org/10.1016/j.foodhyd.2018.03.012
Sakai T, Takaoka A (1985) Purification, crystallization, and some properties of endo-polygalacturonase from Aureobasidium pullulans. Agric Biol Chem 49:449–458. https://doi.org/10.1271/bbb1961.49.449
Pagnonceli J, Rasbold LM, Rocha GB, Silva JLC, Kadowaki MK, Simão RCG, Maller A (2019) Biotechnological potential of an exo-polygalacturonase of the new strain Penicillium janthinellum VI2R3M: biochemical characterization and clarification of fruit juices. J Appl Microbiol 127:1706–1715. https://doi.org/10.1111/jam.14426
Biely P, Heinrichová K, Kruz̃iková M, (1996) Induction and inducers of the pectolytic system in Aureobasidium pullulans. Curr Microbiol 33:6–10. https://doi.org/10.1007/s002849900065
Buzzini P, Martini A (2002) Extracellular enzymatic activity profiles in yeast and yeast-like strains isolated from tropical environments. J Appl Microbiol 93:1020–1025. https://doi.org/10.1046/j.1365-2672.2002.01783.x
Li H, Chi Z, Wang X, Duan X, Ma L, Gao L (2007) Purification and characterization of extracellular amylase from the marine yeast Aureobasidium pullulans N13d and its raw potato starch digestion. Enzyme Microb Technol 40:1006–1012. https://doi.org/10.1016/j.enzmictec.2006.07.036
Kudanga T, Mwenje E (2005) Extracellular cellulase production by tropical isolates of Aureobasidium pullulans. Can J Microbiol 51:773–776. https://doi.org/10.1139/W05-053
Leite RSR, Bocchini DA, Martins EDS, Silva D, Gomes E, Da Silva R (2007) Production of cellulolytic and hemicellulolytic enzymes from Aureobasidium pullulans on solid-state fermentation. Appl Biochem Biotechnol 137:281–288. https://doi.org/10.1007/s12010-007-9058-y
Leite RSR, Alves-Prado HF, Cabral H, Pagnocca FC, Gomes E, Da-Silva R (2008) Production and characteristics comparison of crude β-glucosidases produced by microorganisms Thermoascus aurantiacus e Aureobasidium pullulans in agricultural wastes. Enzyme Microb Technol 43:391–395. https://doi.org/10.1016/j.enzmictec.2008.07.006
Acknowledgements
I would like to thank the Manisa Celal Bayar University for Partial financial support.
Funding
Partial financial support was received from Manisa Celal Bayar University under project number 2015–093.
Author information
Authors and Affiliations
Contributions
The author, MO: Contributed to all processes of the study such as the design of the research, conducting laboratory experiments, interpreting the data, and writing and finalizing the article.
Corresponding author
Ethics declarations
Conflict of interest
The author has no conflict of interest to declare that are relevant to the content of this article.
Ethical Approval
There are no ethical issues related to the research. The author also confirmed that the study did not involve any endangered or protected species, any human subjects or animal handling procedures.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Oskay, M. Production, Partial Purification, and Characterization of Polygalacturonase from Aureobasidium pullulans P56 under Submerged Fermentation Using Agro-Industrial Wastes. Curr Microbiol 79, 296 (2022). https://doi.org/10.1007/s00284-022-02991-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00284-022-02991-6