Abstract
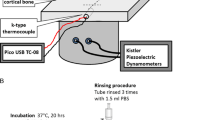
In clinical applications, colonization of metal implants by adhesive and biofilm-forming bacteria not only prolong healing but create additional healthcare costs for implant revision and antimicrobial treatment. An in vitro assay was established investigating the antimicrobial surface activity of external fixation pins intended for use in bone fractures and deformities. Test articles made out of stainless steel and coated with a polymer-containing nanoparticulate silver were compared to non-coated reference controls out of stainless steel, copper and titanium. Staphylococcus epidermidis, known as a predominant cause for implant-related infections was used as test organism. Test pins and bacteria were incubated for a period of 20 h found to be sufficient for initiating biofilm formation. After removing non- and low-adherent bacteria by rinsing, two methods were used to isolate high-adherent (sessile) bacteria from the implant surfaces. Besides shaking the implants in a solution containing small glass beads, a cytobrush technique was used to mechanically harvest viable bacteria. Finally, the amount of detached bacteria was determined by plate counts. Several parameters identified to be critical within the different removal procedures such as the inoculum concentration and the shaking time in the presence of glass beads as well as time of the cytobrush treatment were analysed. The final test scheme resulted in the use of an inoculum of 105 colony forming units (CFU) per millilitre, ten rinsing steps for the removal of low adherent bacteria and 5 min of shaking in the presence of glass beads, detaching the high-adherent bacteria. Due to subjective variations impacting the outcome of the procedure, the cytobrush technique was not favoured and finally rejected. Using the in vitro assay developed, it could be demonstrated that fixation pins coated with silver show a 3 log step reduction in the number of biofilm-forming bacteria compared to a non-coated stainless steel or titanium implant. Pins made out of copper showed the highest antimicrobial efficacy, as the number of detached bacteria was found to be below the detection limit, they served as a positive control within this test.








Similar content being viewed by others
References
Presterl E (2009) Implantatinfektionen, Infektionen von medizinischem Fremdmaterial. Chemother J 18:56–60
An YH, Friedman RJ (eds) (2000) Handbook of bacterial adhesion: principles, methods and applications. Humana Press Inc, Totowa
Zhao L, Chu PK, Zhang Y, Wu Z (2009) Review antibacterial coatings on titanium implants. J Biomed Mat Res B 91:470–480
Montali A (2006) Antibacterial coating systems. Inj Int J Care Injured 37:81–86
Hanna H, Bahna P, Reitzel R, Dvorak T, Chaiban G, Hachem R, Raad I (2006) Comparative in vitro efficacies and antimicrobial durabilities of novel antimicrobial central venous catheters. Antimicrob Agents Chemother 50:3283–3288
Moroni A, Vannini F, Mosca M, Giannini S (2002) Techniques to avoid pin loosening and infection in external fixation. J Orthop Trauma 16:189–195
Collinge CA, Goll G, Seligson D, Easley KJ (1994) Pin tract infections: silver vs. uncoated pins. Orthoped 17:445–448
Masse A, Bruno A, Bosetti M, Biasibetti A, Cannas M, Gallinaro P (2000) Prevention of pin track infection in external fixation with silver coated pins: clinical and microbiological results. J Biomed Mat Res B 53:600–604
Meyer Ch, Keßler J, Alt V, Wenisch S, Hartmann B, Schiefer HG, Schnettler R (2004) Antimicrobial effect of silver-coated external fixator pins. Osteo Trauma Care 12:81–84
Coester LM, Nepola JV, Allen J, Marsh JL (2006) The effects of silver coated external pins. Iowa Orthopaed J 26:48–53
Maki DG, Jarrett F, Sarafin HW (1977) A semiquantitative culture method for identification of catheter-related infection in the burn patient. J Surg Res 22:513–520
Stevens KNJ, Crespo-Biel O, van den Bosch EEM, Dias AA, Knetsch MLW, Aldenhoff YBJ, van der Veen FH, Maessen JG, Stobberingh EE, Koole LH (2009) The relationship between the antimicrobial effect of catheter coatings containing silver nanoparticles and the coagulation of contacting blood. Biomaterials 30:3682–3690
Gollwitzer H, Ibrahim K, Meyer H, Mittelmeier W, Busch R, Stemberger A (2003) Antibacterial poly(d, l-lactid acid) coating of medical implants using a biodegradable drug delivery technology. J Antimic Chemother 51:585–591
Yoshinari M, Oda Y, Kato T, Okuda K (2001) Influence of surface modifications to titanium on antibacterial activity in vitro. Biomaterials 22:2043–2048
Kramer A, Assadian O (eds) (2008) Praxis der Sterilisation Desinfektion Antiseptik und Konservierung. Georg Thieme Verlag Stuttgart, New York, p 846
Chiang WC, Tseng IS, Moller P, Hilbert LR, Tolker-Nielsen T, Wu JK (2010) Influence of silver additions to type 316 stainless steels on bacterial inhibition, mechanical properties, and corrosion resistance. Mat Chem Phys 119:123–130
Furno F, Morley KS, Wong B, Sharp BL, Arnold PL, Howdle SM, Bayston R, Brown PD, Winship PD, Reid HJ (2004) Silver nanoparticles and polymeric medical devices: a new approach to prevention of infection? Antimic Chemother 54:1019–1024
Roe D, Karandikar B, Bonn-Savage N, Gibbins B, Roullet JB (2008) Antimicrobial surface functionalization of plastic catheters by silver nanoparticles. Antimic Chemother 61:869–876
Gabriel MM, Sawant AD, Simmons RB, Ahearn DG (1995) Effects of silver on adherence of bacteria to urinary catheters: in vitro studies. Curr Microbiol 30:17–22
Ahearn DG, Grace DT, Jennings MJ, Borazjani RN, Boles KJ, Rose LJ, Simmons RB, Ahanotu EN (2000) Effects of hydrogel/silver coatings on in vitro adhesion to catheters of bacteria associated with urinary tract infections. Curr Microbiol 41:120–125
Trampuz A, Osmon DR, Hanssen AD, Steckelberg JM, Patel R (2003) Molecular and antibiofilm approaches to prosthetic joint infection. Clin Orthop Relat Res 414:69–88
Trachoo N (2004) Biofilm removal technique using sands as a research tool for assessing microbial attachment on surface. Songklanakarin J Sci Technol 26:109–115
Peters G, Locci R, Pulverer G (1982) Adherence and growth of coagulase-negative staphylococci on surfaces of intravenous catheters. Infect Dis 146:479–482
Pharmacopoeia Europeae 6.0 (2008) Deutscher Apotheker Verlag Stuttgart, Germany
Nissen S, Furkert FH (2000) Antimikrobielle Wirksamkeit einer Silberbeschichtung von Hydrogellinsen. Ophthalmologe 97:640–643
Yorganci K, Krepel C, Weigelt JA, Edmiston CE (2002) In vitro evaluation of the antibacterial activity of three different central venous catheters against gram-positive bacteria. Eur J Clin Microbiol Infect Dis 21:379–384
Secinti KD, Özalp h, Attar A, Sargon MF (2011) Nanoparticle silver ion coatings inhibit biofilm formation on titanium implants. Clin Neurosci 18:391–395
Standard test method for determining the activity of incorporated antimicrobial agent(s) in polymeric or hydrophobic materials (2002) American Society for Testing and Materials E 2180-01 ASTM, West Conshohocken, PA, USA
Japanese Standards Association (2000) Antimicrobial products-test for antimicrobial activity and efficacy. Japanese Industrial Standard JIS Z 2801:2000
Standard test method for determining the antimicrobial activity of immobilized antimicrobial agents under dynamic contact conditions (2001) American Society for Testing and Materials E 2149-01, ASTM West Conshohocken, PA, USA
Chemische Desinfektionsmittel und Antiseptika, Bakterizide Wirkung (Basistest)-Prüfverfahren und Anforderungen. DIN EN 1040 2006-03 (D) (2006) Beuth Verlag Berlin, Germany
Oliver JD (2010) Recent findings on the viable but nonculturable state in pathogenic bacteria. FEMS Microbiol Rev 34:415–425
Colwell RR (2009) Viable but not cultivable bacteria. Microbiol Monogr 10:121–129
Grey B, Steck T (2001) Concentrations of copper thought to be toxic to Escherichia coli can induce the viable but nonculturable condition. Appl Environ Microbiol 67:5325–5327
Sardessai YN (2005) Viable but non-culturable bacteria: their impact on public health. Curr Sci 89:1650
Kobayashi N, Bauer TW, Tuohy MJ, Fujishiro T, Procop GW (2007) Brief ultrasonication improves detection of biofilm-formative bacteria around a metal implant. Clin Orthop Relat Res 457:210–213
Kobayashi H, Oethinger M, Tuohy MJ, Procop GW, Bauer TW (2009) Improved detection of biofilm-formative bacteria by vortexing and sonication. Clin Orthop Relat Res 467:1360–1364
Wassall MA, Santin M, Isalberti C, Cannas M, Denyer SP (1997) Adhesion of bacteria to stainless steel and silver-coated orthopaedic external fixation pins. Biomed Mat Res 36:325–330
Samuel U, Guggenbichler JP (2004) Prevention of catheter-related infections: the potential of a new nano-silver impregnated catheter. Int J Antimicrob Agents 23:S75–S78
Valappil SP, Pickup DM, Carroll DL, Hope CK, Pratten J, Newport RJ, Smith ME, Wilson M, Knowles JC (2007) Effect of silver content on the structure and antibacterial activity of silver-doped phosphate-based glasses. Antimicrob Agents Chemother 51:4453–4461
Acknowledgments
Atomic Force analyses were provided by Dr. S. Wille, Functional Nanomaterials, Institute for Materials Science, Faculty of Engineering, Christian Albrecht University, Kiel, Germany
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Furkert, F.H., Sörensen, J.H., Arnoldi, J. et al. Antimicrobial Efficacy of Surface-Coated External Fixation Pins. Curr Microbiol 62, 1743–1751 (2011). https://doi.org/10.1007/s00284-011-9923-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-011-9923-3