Abstract
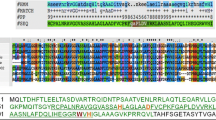
Previously, two genes, designated as lyt and hol, were identified in the lysis module of phage μ1/6. They were cloned and expressed in Escherichia coli. An additional candidate holin gene, hol2, was found downstream from the hol gene based on one predicted transmembrane domain and a highly charged C-terminal sequence of the encoded protein. Expression of hol or hol2 in E. coli was shown to cause cell death. The concomitant expression of λ endolysin (R) and μ1/6 holin resulted in cell lysis. Similarly, the coexpression of the endolysin and holin of phage μ1/6 led to lysis, apparently due to the ability of μ1/6 endolysin to hydrolyze the peptidoglycan layer of this bacterium. In contrast, the simultaneous expression of μ1/6 hol2 and the endolysin gene (λR or μ1/6 lyt) did not cause detectable lysis of the host cells. Demonstration of the holin function in streptomycetes was achieved by providing for the release of μ1/6 endolysin to the periplasm and subsequent cleavage of the peptidoglycan, which strongly suggested that the holin produces lesions in the streptomycete membrane.
Access this article
We’re sorry, something doesn't seem to be working properly.
Please try refreshing the page. If that doesn't work, please contact support so we can address the problem.



Similar content being viewed by others
References
Agu ChA, Klein R, Schwab S et al (2006) The cytotoxic activity of the bacteriophage λ-holin protein reduces tumour growth rates in mammary cancer cell xenograft models. J Gene Med 8:229–241
Altschul SF, Madden TL, Schäffer AA et al (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Birnboim HC, Doly J (1979) A rapid procedure for screening recombinant plasmid DNA. Nucleic Acids Res 7:1513–1523
Blondelet-Rouault M-H, Weiser J, Lebrihi A et al (1997) Antibiotic resistance gene cassettes derived from the Ω interposon for use in E. coli and Streptomyces. Gene 190:315–317
Borysowski J, Weber-Dabrowska B, Górski A (2005) Bacteriophage endolysins as a novel class of antibacterial agents. Exp Biol Med 231:366–377
Delisle AL, Barcak GJ, Guo M (2006) Isolation and expression of the lysis genes of Actinomyces naeslundii phage Av-1. Appl Environ Microbiol 72:1110–1117
Denis F, Brzezinski R (1992) A versatile shuttle cosmid vector for use in Escherichia coli and actinomycetes. Gene 111:115–118
Díaz E, Munthali M, Lűnsdorf H, Höltje J-V, Timmis KN (1996) The two-step lysis system of pneumococcal bacteriophage EJ-1 is functional in Gram-negative bacteria: triggering of the major pneumococcal autolysin in Escherichia coli. Mol Microbiol 19:667–681
Farkašovská J, Godány A, Vlček Č (2003) Identification and characterization of an endolysin encoded by the Streptomyces aureofaciens phage μ1/6. Folia Microbiol 48:737–744
Farkašovská J, Godány A, Vlček Č (2004) Identification of a holin encoded by the Streptomyces aureofaciens phage μ1/6: functional analysis in Escherichia coli system. Folia Microbiol 49:679–684
Farkašovská J, Klucar L, Vlček Č et al (2007) Complete genome sequence and analysis of the Streptomyces aureofaciens phage μ1/6. Folia Microbiol 52:347–358
Hermoso JA, García J, García P (2007) Taking aim on bacterial pathogens: from phage therapy to enzybiotics. Curr Opin Microbiol 10:461–472
Kieser T, Bibb MJ, Buttner MJ et al (2000) Practical Streptomyces Genetics. John Innes Foundation, Norwich, England
Krogh S, Jorgensen ST, Devine KM (1998) Lysis genes of the Bacillus subtilis defective prophage PBSX. J Bacteriol 180:2110–2117
Loessner MJ, Gaeng S, Scherer S (1999) Evidence for a holin-like protein gene fully embedded out of frame in the endolysin gene of Staphylococcus aureus bacteriophage 187. J Bacteriol 181:4452–4460
Loessner MJ (2005) Bacteriophage endolysins: current state of research and applications. Curr Opin Microbiol 8:480–487
Martín AC, López R, García P (1998) Functional analysis of two-gene lysis system of the pneumococcal phage Cp-1 in homologous and heterologous host cells. J Bacteriol 180:210–217
Murakami T, Holt TG, Thompson CJ (1989) Thiostrepton-induced gene expression in S. lividans. J Bacteriol 171:1459–1466
Ramanculov E, Young R (2001) An ancient player unmasked: T4 rI encodes a t-specific antiholin. Mol Microbiol 41:575–583
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Sheenan MM, Stanley E, Fitzgerald GF, van Sinderen D (1999) Identification and characterization of a lysis module present in a large proportion of bacteriophages infecting Streptococcus thermophilus. Appl Environ Microbiol 65:569–577
Sonnhammer ELL, von Heijne G, Krogh A (1998) A hidden Markov model for predicting transmembrane helices in protein sequences. In: Glasgow J, Littlejohn T, Major F et al (eds) Proceedings of the sixth international conference on intelligent systems for molecular biology, AAAI Press, Menlo Park, CA, USA, pp 175–182
Takáč M, Witte A, Bläsi U (2005) Functional analysis of the lysis genes of Staphylococcus aureus phage P68 in Escherichia coli. Microbiology 151:2331–2342
Takano E, White J, Thompson CJ, Bibb MJ (1995) Construction of thiostrepton-inducible, high-copy-number expression vectors for use in Streptomyces spp. Gene 166:133–137
Young Ry, Wang Ing-Nang, Roof WD (2000) Phages will out: strategies of host cell lysis. Trends Microbiol 8:120–128
Young Ry, Wang IN (2006) Phage lysis. In: Calendar R (ed) The Bacteriophages. Oxford University Press, Oxford, pp 104–125
Acknowledgment
This work was supported by VEGA grant no. 2/0121/25 of the Slovak Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Farkašovská, J., Godány, A. The Lysis System of the Streptomyces aureofaciens Phage μ1/6. Curr Microbiol 57, 631–637 (2008). https://doi.org/10.1007/s00284-008-9255-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-008-9255-0