Abstract
Purpose
The aim of this multicenter study was to evaluate the clinical outcomes of patients with metastatic renal cell carcinoma (mRCC) who received sunitinib retreatment.
Methods
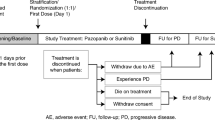
Clinical data from patients treated with sunitinib rechallenge in nine Spanish centers were retrospectively analyzed. All patients received first-line sunitinib until progression or intolerance, followed by one or more successive drugs and rechallenge with sunitinib thereafter.
Results
Thirty-seven patients were included. At first-line treatment, objective response rate (ORR) was 69.4% and median progression-free survival (PFS) was 19.4 months. At rechallenge, ORR was 27.2% and 39.4% of patients obtained stabilization of disease. Median PFS was 6.2 months. Clinical benefit was obtained by 21 patients (75%) with > 6-month interval between sunitinib treatments and by 1 patient (20%) among those with ≤ 6-month interval (P = 0.016). Hemoglobin levels ≥ lower level of normal were associated with clinical benefit (P = 0.019) and with PFS (P = 0.004). Median overall survival from start of first-line sunitinib was 52.7 months. No new adverse events were observed at rechallenge.
Conclusions
Sunitinib rechallenge is a feasible treatment option for selected patients with mRCC.


Similar content being viewed by others
References
Ljungberg B, Campbell SC, Choi HY, Jacqmin D, Lee JE, Weikert S, Kiemeney LA (2011) The epidemiology of renal cell carcinoma. Eur Urol 60:615–621 (Erratum in: Eur Urol 2011; 60:1317. Cho, Han Yong [corrected to Choi, Han Yong])
Rini BI, Small EJ (2005) Biology and clinical development of vascular endothelial growth factor-targeted therapy in renal cell carcinoma. J Clin Oncol 23:1028–1043
Brugarolas J (2014) Molecular genetics of clear-cell renal cell carcinoma. J Clin Oncol 32:1968–1976
Cancer Genome Atlas Research Network (2013) Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499(7456):43–49
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674
Schmelzle T, Hall MN (2000) TOR, a central controller of cell growth. Cell 103:253–262
Fingar DC, Richardson CJ, Tee AR, Cheatham L, Tsou C, Blenis J (2004) mTOR controls cell cycle progression through its cell growth effectors S6K1 and 4E-BP1/eukaryotic translation initiation factor 4E. Mol Cell Biol 24:200–216
Hudson CC, Liu M, Chiang GG, Otterness DM, Loomis DC, Kaper F, Giaccia AJ, Abraham RT (2002) Regulation of hypoxia-inducible factor 1alpha expression and function by the mammalian target of rapamycin. Mol Cell Biol 22:7004–7014
Bamias A, Escudier B, Sternberg CN, Zagouri F, Dellis A, Djavan B, Tzannis K, Kontovinis L, Stravodimos K, Papatsoris A, Mitropoulos D, Deliveliotis C, Dimopoulos MA, Constantinides CA (2017) Current clinical practice guidelines for the treatment of renal cell carcinoma: a systematic review and critical evaluation. Oncologist 22:667–679
Heng DY, Mackenzie MJ, Vaishampayan UN, Bjarnason GA, Knox JJ, Tan MH, Wood L, Wang Y, Kollmannsberger C, North S, Donskov F, Rini BI, Choueiri TK (2012) Primary anti-vascular endothelial growth factor (VEGF)-refractory metastatic renal cell carcinoma: clinical characteristics, risk factors, and subsequent therapy. Ann Oncol 23:1549–1555
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, Oudard S, Negrier S, Szczylik C, Kim ST, Chen I, Bycott PW, Baum CM, Figlin RA (2007) Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356:115–124
Gore ME, Szczylik C, Porta C, Bracarda S, Bjarnason GA, Oudard S, Lee SH, Haanen J, Castellano D, Vrdoljak E, Schöffski P, Mainwaring P, Hawkins RE, Crinò L, Kim TM, Carteni G, Eberhardt WE, Zhang K, Fly K, Matczak E, Lechuga MJ, Hariharan S, Bukowski R (2015) Final results from the large sunitinib global expanded-access trial in metastatic renal cell carcinoma. Br J Cancer 113:12–19
Joosten SC, Hamming L, Soetekouw PM, Aarts MJ, Veeck J, van Engeland M, Tjan-Heijnen VC (2015) Resistance to sunitinib in renal cell carcinoma: from molecular mechanisms to predictive markers and future perspectives. Biochim Biophys Acta 1855:1–16
Ko JS, Zea AH, Rini BI, Ireland JL, Elson P, Cohen P, Golshayan A, Rayman PA, Wood L, Garcia J, Dreicer R, Bukowski R, Finke JH (2009) Sunitinib mediates reversal of myeloid-derived suppressor cell accumulation in renal cell carcinoma patients. Clin Cancer Res 15:2148–2157
Adotevi O, Pere H, Ravel P, Haicheur N, Badoual C, Merillon N, Medioni J, Peyrard S, Roncelin S, Verkarre V, Mejean A, Fridman WH, Oudard S, Tartour E (2010) A decrease of regulatory T cells correlates with overall survival after sunitinib-based antiangiogenic therapy in metastatic renal cancer patients. J Immunother 33:991–998
Liu XD, Hoang A, Zhou L, Kalra S, Yetil A, Sun M, Ding Z, Zhang X, Bai S, German P, Tamboli P, Rao P, Karam JA, Wood C, Matin S, Zurita A, Bex A, Griffioen AW, Gao J, Sharma P, Tannir N, Sircar K, Jonasch E (2015) Resistance to antiangiogenic therapy is associated with an immunosuppressive tumor microenvironment in metastatic renal cell carcinoma. Cancer Immunol Res 3:1017–1029
Mollica V, Di Nunno V, Gatto L, Santoni M, Cimadamore A, Cheng L, Lopez-Beltran A, Montironi R, Pisconti S, Battelli N, Massari F (2019) Novel therapeutic approaches and targets currently under evaluation for renal cell carcinoma: waiting for the revolution. Clin Drug Investig. https://doi.org/10.1007/s40261-019-00773-w
Motzer RJ, Tannir NM, McDermott DF, Arén Frontera O, Melichar B, Choueiri TK, Plimack ER, Barthélémy P, Porta C, George S, Powles T, Donskov F, Neiman V, Kollmannsberger CK, Salman P, Gurney H, Hawkins R, Ravaud A, Grimm MO, Bracarda S, Barrios CH, Tomita Y, Castellano D, Rini BI, Chen AC, Mekan S, McHenry MB, Wind-Rotolo M, Doan J, Sharma P, Hammers HJ, Escudier B, CheckMate 214 Investigators (2018) Nivolumab plus ipilimumab versus sunitinib in advanced renal-cell carcinoma. N Engl J Med 378:1277–1290
Armstrong AJ, Halabi S, Eisen T, Broderick S, Stadler WM, Jones RJ, Garcia JA, Vaishampayan UN, Picus J, Hawkins RE, Hainsworth JD, Kollmannsberger CK, Logan TF, Puzanov I, Pickering LM, Ryan CW, Protheroe A, Lusk CM, Oberg S, George DJ (2016) Everolimus versus sunitinib for patients with metastatic non-clear cell renal cell carcinoma (ASPEN): a multicentre, open-label, randomised phase 2 trial. Lancet Oncol 17:378–388
Tannir NM, Jonasch E, Albiges L, Altinmakas E, Ng CS, Matin SF, Wang X, Qiao W, Dubauskas Lim Z, Tamboli P, Rao P, Sircar K, Karam JA, McDermott DF, Wood CG, Choueiri TK (2016) Everolimus versus sunitinib prospective evaluation in metastatic non-clear cell renal cell carcinoma (ESPN): a randomized multicenter phase 2 trial. Eur Urol 69:866–874
Escudier B, Porta C, Schmidinger M, Rioux-Leclercq N, Bex A, Khoo V, Grünwald V, Gillessen S, Horwich A, ESMO Guidelines Committee (2019) Renal cell carcinoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. https://doi.org/10.1093/annonc/mdz056
Ko JJ, Choueiri TK, Rini BI, Lee JL, Kroeger N, Srinivas S, Harshman LC, Knox JJ, Bjarnason GA, MacKenzie MJ, Wood L, Vaishampayan UN, Agarwal N, Pal SK, Tan MH, Rha SY, Yuasa T, Donskov F, Bamias A, Heng DY (2014) First-, second-, third-line therapy for mRCC: benchmarks for trial design from the IMDC. Br J Cancer 110:1917–1922. https://doi.org/10.1038/bjc.2014.25
Zama IN, Hutson TE, Elson P, Cleary JM, Choueiri TK, Heng DY, Ramaiya N, Michaelson MD, Garcia JA, Knox JJ, Escudier B, Rini BI (2010) Sunitinib rechallenge in metastatic renal cell carcinoma patients. Cancer 116:5400–5406
Grünwald V, Weikert S, Seidel C, Busch J, Johannsen A, Fenner M, Reuter C, Ganser A, Johannsen M (2011) Efficacy of sunitinib re-exposure after failure of an mTOR inhibitor in patients with metastatic RCC. Onkologie 34:310–314
Nagyiványi K, Budai B, Gyergyay F, Küronya Z, Bíró K, Géczi L (2019) Sunitinib rechallenge after other targeted therapies in metastatic renal cell carcinoma patients: a single-center. Clin Drug Investig, Retrospective Study. https://doi.org/10.1007/s40261-019-00778-5
Oudard S, Geoffrois L, Guillot A, Chevreau C, Deville JL, Falkowski S, Boyle H, Baciuchka M, Gimel P, Laguerre B, Laramas M, Pfister C, Topart D, Rolland F, Legouffe E, Denechere G, Amela EY, Abadie-Lacourtoisie S, Gross-Goupil M (2016) Clinical activity of sunitinib rechallenge in metastatic renal cell carcinoma-Results of the REchallenge with SUnitinib in MEtastatic RCC (RESUME) Study. Eur J Cancer 62:28–35
Motzer RJ, Escudier B, Bukowski R, Rini BI, Hutson TE, Barrios CH, Lin X, Fly K, Matczak E, Gore ME (2013) Prognostic factors for survival in 1059 patients treated with sunitinib for metastatic renal cell carcinoma. Br J Cancer 108:2470–2477
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247
Shin SJ, Jeon YK, Cho YM, Lee JL, Chung DH, Park JY, Go H (2015) The association between PD-L1 expression and the clinical outcomes to vascular endothelial growth factor-targeted therapy in patients with metastatic clear cell renal cell carcinoma. Oncologist 20:1253–1260
Zhou L, Liu XD, Sun M, Zhang X, German P, Bai S, Ding Z, Tannir N, Wood CG, Matin SF, Karam JA, Tamboli P, Sircar K, Rao P, Rankin EB, Laird DA, Hoang AG, Walker CL, Giaccia AJ, Jonasch E (2015) Targeting MET and AXL overcomes resistance to sunitinib therapy in renal cell carcinoma. Oncogene. https://doi.org/10.1038/onc.2015.343
Kasikara C, Kumar S, Kimani S, Tsou WI, Geng K, Davra V, Sriram G, Devoe C, Nguyen KQ, Antes A, Krantz A, Rymarczyk G, Wilczynski A, Empig C, Freimark BD, Gray M, Schlunegger K, Hutchins J, Kotenko SV, Birge RB (2017) Phosphatidylserine sensing by TAM receptors regulates AKT-dependent chemoresistance and PD-L1 expression. Mol Cancer Res. https://doi.org/10.1158/1541-7786.mcr-16-0350
Nadal R, Amin A, Geynisman DM, Voss MH, Weinstock M, Doyle J, Zhang Z, Viudez A, Plimack ER, McDermott DF, Motzer R, Rini B, Hammers HJ (2016) Safety and clinical activity of vascular endothelial growth factor receptor (VEGFR)-tyrosine kinase inhibitors after programmed cell death 1 inhibitor treatment in patients with metastatic clear cell renal cell carcinoma. Ann Oncol 27:1304–1311
Funding
This work is supported by Pfizer, Inc.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Gaspar Reynés, Dr. Puente and Dr Climent have received travel support and honoraria from Pfizer. The other authors declare to have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Munárriz, J., Reynés, G., Sánchez-Lorenzo, L. et al. Sunitinib rechallenge in advanced renal cell carcinoma: outcomes of a multicenter retrospective study. Cancer Chemother Pharmacol 84, 781–789 (2019). https://doi.org/10.1007/s00280-019-03913-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-019-03913-3