Abstract
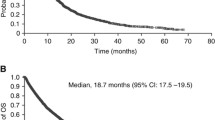
A global, open-label, expanded-access trial (EAT) provided sunitinib treatment on a compassionate-use basis to patients with metastatic renal cell carcinoma (mRCC) between 2005 and 2011. This retrospective analysis examines outcomes in patients from Central and East European (CEE) countries participating in the global EAT. Sunitinib (starting dose 50 mg orally once daily, with dose reduction for toxicity) was administered in repeated 6-week cycles (4 weeks on and 2 weeks off) until occurrence of disease progression or unacceptable toxicity. Tumor assessments were guided by Response Evaluation Criteria in Solid Tumors (RECIST) criteria but were performed according to local standards of care. In total, 401 CEE patients received sunitinib (median treatment duration 9.6 months), of whom 378 were evaluable for tumor response. The most frequent grade ≥3 toxicities were fatigue (7.5 %), hypertension (7.0 %), thrombocytopenia (6.5 %), diarrhea (4.2 %), nausea and hand-foot syndrome (both 3.7 %) and neutropenia (3.0 %). Median overall survival was 30.7 months (95 % CI 23.3, ‒ months). Overall survival tended to be longer in cytokine-naïve than cytokine-experienced patients (median 60.8 vs. 27.5 months; P = 0.1324). Among patients with evaluable tumors, 4.0 % achieved a complete and 14.6 % a partial response [objective response rate (ORR) 18.5 % (95 % CI 14.7, 22.8 %)]. Median progression-free survival was 11.6 months (95 % CI 10.3, 12.8 months). Sunitinib demonstrates safety and effectiveness in real-world mRCC patients in CEE countries. Expanded-access program patients showed a lower tumor response rate but similar survival outcomes to patients in the pivotal Phase III clinical trial of sunitinib in mRCC.



Similar content being viewed by others
References
Mena AC, Pulido EG, Guillén-Ponce C. Understanding the molecular-based mechanism of action of the tyrosine kinase inhibitor: sunitinib. Anti-Cancer Drugs 21 Suppl. 1: S3-S11. doi: 10.1097/01.cad.0000361534.44052.c5
Escudier B, Eisen T, Porta C, Patard JJ, Khoo V, Algaba F, Mulders P, Kataja V (2012) Renal cell carcinoma: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol 23(7):vii65–vii71. doi:10.1093/annonc/mds227
Patard JJ, Pignot G, Escudier B, Eisen T, Bex A, Sternberg C, Rini B, Roigas J, Choueiri T, Bukowski R, Motzer R, Kirkali Z, Mulders P, Bellmunt J (2011) ICUD-EAU International consultation on kidney cancer 2010: treatment of metastatic disease. Eur Urol 60(4):684–690. doi:10.1016/j.eururo.2011.06.017
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O, Oudard S, Negrier S, Szczylik C, Kim ST, Chen I, Bycott PW, Baum CM, Figlin RA (2007) Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 356(2):115–124. doi:10.1056/NEJMoa065044
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Oudard S, Negrier S, Szczylik C, Pili R, Bjarnason GA, Garcia-del-Muro X, Sosman JA, Solska E, Wilding G, Thompson JA, Kim ST, Chen I, Huang X, Figlin RA (2009) Overall survival and updated results for sunitinib compared with interferon alfa in patients with metastatic renal cell carcinoma. J Clin Oncol 27(22):3584–3590. doi:10.1200/JCO.2008.20.1293
Gore ME, Szczylik C, Porta C, Bracarda S, Bjarnason GA, Oudard S, Hariharan S, Lee SH, Haanen J, Castellano D, Vrdoljak E, Schoffski P, Mainwaring P, Nieto A, Yuan J, Bukowski R (2009) Safety and efficacy of sunitinib for metastatic renal-cell carcinoma: an expanded-access trial. Lancet Oncol 10(8):757–763. doi:10.1016/S1470-2045(09)70162-7
Gore ME, Porta C, Bracarda S, Bjarnason GA, Oudard S, Lee S, Crino L, Kim TM, Fly K, Szczylik C (2012) Sunitinib global expanded-access trial in metastatic renal cell carcinoma (mRCC): final results. Ann Oncol 23(9):271, Abstract 820P
Curado MP, Edwards B, Shin HR, Storm H, Ferlay J, Heanue M, Boyle P (2007). In: Cancer Incidence in Five Continents Vol. IX, vol Scientific Publication No. 160. IARC, Lyon, France
Levi F, Lucchini F, Negri E, Boyle P, La Vecchia C (2004) Cancer mortality in Europe, 1995-1999, and an overview of trends since 1960. Int J Cancer 110(2):155–169. doi:10.1002/ijc.20097
Levi F, Lucchini F, Negri E, Zatonski W, Boyle P, La Vecchia C (2004) Trends in cancer mortality in the European Union and accession countries, 1980-2000. Ann Oncol 15(9):1425–1431. doi:10.1093/annonc/mdh346
Berrino F, De Angelis R, Sant M, Rosso S, Bielska-Lasota M, Coebergh JW, Santaquilani M (2007) Survival for eight major cancers and all cancers combined for European adults diagnosed in 1995-99: results of the EUROCARE-4 study. Lancet Oncol 8(9):773–783. doi:10.1016/S1470-2045(07)70245-0
Castellano D, Duh MS, Korves C, Suthoff ED, Neary M, Hernández Pastor LJ, Bellmunt J (2013) Safety and treatment patterns of angiogenesis inhibitors in patients with advanced renal cell carcinoma in Spain. Expert Opin Drug Saf 12(4):455–463. doi:10.1517/14740338.2013.781581
Porta C, Paglino C, Imarisio I, Canipari C, Chen K, Neary M, Duh MS (2011) Safety and treatment patterns of multikinase inhibitors in patients with metastatic renal cell carcinoma at a tertiary oncology center in Italy. BMC Cancer 11:105. doi:10.1186/1471-2407-11-105
Ansari J, Fatima A, Fernando K, Collins S, James ND, Porfiri E (2010) Sunitinib in patients with metastatic renal cell carcinoma: Birmingham experience. Oncol Rep 24(2):507–510
Hess G, Borker R, Fonseca E (2013) Treatment patterns: targeted therapies indicated for first-line management of metastatic renal cell carcinoma in a real-world setting. Clin Genitourin Cancer 11(2):161–167. doi:10.1016/j.clgc.2012.10.003
Filson CP, Redman BG, Dunn RL, Miller DC (2011) Initial patterns of care with oral targeted therapies for patients with renal cell carcinoma. Urology 77(4):825–830. doi:10.1016/j.urology.2010.11.003
Choueiri TK, Duh MS, Clement J, Brick AJ, Rogers MJ, Kwabi C, Shah K, Percy AG, Antras L, Jayawant SS, Chen K, Wang ST, Luka A, Neary MP, McDermott D, Oh WK (2010) Angiogenesis inhibitor therapies for metastatic renal cell carcinoma: effectiveness, safety and treatment patterns in clinical practice-based on medical chart review. BJU Int 105(9):1247–1254. doi:10.1111/j.1464-410X.2009.08972.x
Feinberg BA, Jolly P, Wang ST, Fortner B, Scott J, Gilmore J, Neary MP, Duh MS (2012) Safety and treatment patterns of angiogenesis inhibitors in patients with metastatic renal cell carcinoma: evidence from US community oncology clinics. Med Oncol 29(2):786–794. doi:10.1007/s12032-011-9922-z
Hong MH, Kim HS, Kim C, Ahn JR, Chon HJ, Shin SJ, Ahn JB, Chung HC, Rha SY (2009) Treatment outcomes of sunitinib treatment in advanced renal cell carcinoma patients: a single cancer center experience in Korea. Cancer Res Treat 41(2):67–72. doi:10.4143/crt.2009.41.2.67
Schmidinger M, Larkin J, Ravaud A (2012) Experience with sunitinib in the treatment of metastatic renal cell carcinoma. Ther Adv Urol 4(5):253–265. doi:10.1177/1756287212454933
Heck JE, Charbotel B, Moore LE, Karami S, Zaridze DG, Matveev V, Janout V, Kollarova H, Foretova L, Bencko V, Szeszenia-Dabrowska N, Lissowska J, Mates D, Ferro G, Chow WH, Rothman N, Stewart P, Brennan P, Boffetta P (2010) Occupation and renal cell cancer in central and Eastern Europe. Occup Environ Med 67(1):47–53. doi:10.1136/oem.2009.046250
Moore LE, Hung R, Karami S, Boffetta P, Berndt S, Hsu CC, Zaridze D, Janout V, Kollarova H, Bencko V, Navratilova M, Szeszenia-Dabrowska N, Mates D, Mukeria A, Holcatova I, Yeager M, Chanock S, Garcia-Closas M, Rothman N, Chow WH, Brennan P (2008) Folate metabolism genes, vegetable intake and renal cancer risk in central Europe. Int J Cancer 122(8):1710–1715. doi:10.1002/ijc.23318
Fabiánová E, Szeszenia-Dabrowska N, Kjaerheim K, Boffetta P (1999) Occupational cancer in central European countries. Environ Health Perspect 107(Suppl 2):279–282
Döbrossy L (2002) Cancer mortality in central-eastern Europe: facts behind the figures. Lancet Oncol 3(6):374–381
Antunes JL, Toporcov TN, de Andrade FP (2003) Trends and patterns of cancer mortality in European countries. Eur J Cancer Prev 12(5):367–372. doi:10.1097/01.cej.0000090182.08740.97
Zatonski W, Didkowska J (2008) Closing the gap: cancer in Central and Eastern Europe (CEE). Eur J Cancer 44(10):1425–1437. doi:10.1016/j.ejca.2008.02.014
Vrdoljak E, Wojtukiewicz MZ, Pienkowski T, Bodoky G, Berzinec P, Finek J, Todorovic V, Borojevic N, Croitoru A (2011) Cancer epidemiology in Central, South and Eastern European countries. Croat Med J 52(4):478–487. doi:10.3325/cmj.2011.52.478
Acknowledgments
This study was sponsored by Pfizer Inc. Medical writing support was provided by Andrew Fitton, PhD, of Engage Scientific Solutions and was funded by Pfizer Inc.
Eduard Vrdoljak has received funding for clinical trials from Roche and Pfizer Inc,, and has received consultancy fees/honoraria from Pfizer Inc., Roche, Glaxo, Novartis and Bayer. Lajos Géczi has received consultancy fees/honoraria from Pfizer Inc., Sanofi-Aventis, Novartis and Bayer. Jozef Mardiak has received research funding from Novartis, and consultancy fees/honoraria from Pfizer Inc., Roche and Novartis. Tudor-Eliade Ciuleanuhas received consultancy fees/honoraria from Pfizer Inc., and has acted as a consultant and/or speakers’ bureau member for Pfizer Inc., Amgen, Sandoz, Merck, Roche, Bristol Myers Squibb, Novartis, Lilly, AstraZeneca, Janssen and Bayer. Laszlo Torday has received funding for clinical trials from Roche, Imatics Research, Amgen, Astra Zeneca, Astellas, Bayer, Helsinn and Pfizer Inc., and consulting honoraria from Pfizer, Roche, Bayer and Novartis. Sophie Leyman, Ke Zhang and Peter Sajben were full-time employees of Pfizer Inc. during the conduct of this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vrdoljak, E., Géczi, L., Mardiak, J. et al. Central and Eastern European Experience with Sunitinib in Metastatic Renal Cell Carcinoma: A Sub-analysis of the Global Expanded-Access Trial. Pathol. Oncol. Res. 21, 775–782 (2015). https://doi.org/10.1007/s12253-014-9889-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12253-014-9889-0