Abstract
Purpose
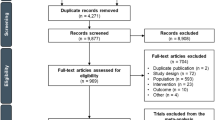
We conducted a systematic review and meta-analysis on survival impact of post-progression chemotherapy (post-Cx) after first-line chemotherapy (1st-Cx) and after second-line chemotherapy (2nd-Cx), and survival benefit of third-line chemotherapy (3rd-Cx) for advanced gastric cancer (AGC).
Methods
Phase III trials of systemic chemotherapy for AGC published in English between 2005 and 2015 or presented at annual meetings of ASCO or ESMO between 2013 and 2015 were searched. Numbers of patients, types of chemotherapy, patient baseline, proportion of patients receiving post-Cx (post-Cx%), median progression-free survival (mPFS), and median overall survival (mOS) of each treatment arm were surveyed; trials not reporting these parameters were excluded. Median post-progression survival (mPPS) was calculated as the difference between mOS and mPFS. Weighted Spearman’s correlation coefficients between post-Cx% and survival outcomes (mOS and mPPS) were calculated. The effect of post-Cx% on survival outcomes adjusted for the types of chemotherapy and patient characteristics was evaluated by meta-regression.
Results
Overall, 25 phase III trials of AGC were selected: 15 trials with 31 arms for 1st-Cx, and 10 trials with 16 arms for 2nd-Cx. Weighted Spearman’s correlation coefficients for post-Cx% and mOS/mPPS were 0.520/0.739 for 1st-Cx, and 0.767/0.823 for 2nd-Cx. Meta-regression analyses adjusting for types of chemotherapy, age, and PS showed that a 10% increase in post-Cx% was associated with prolongation of mOS by 1.033 months for 1st-Cx and 0.344 months for 2nd-Cx.
Conclusions
Post-Cx% both after 1st-Cx and 2nd-Cx were correlated with mOS/mPPS, suggesting a survival benefit of 3rd-Cx in addition to that of 2nd-Cx for AGC.


Similar content being viewed by others
References
Ferlay J, Soerjomataram I, Dikshit R et al (2015) Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 136(5):E359-386. https://doi.org/10.1002/ijc.29210
Wagner AD, Grothe W, Haerting J, Kleber G, Grothey A, Fleig WE (2006) Chemotherapy in advanced gastric cancer: a systematic review and meta-analysis based on aggregate data. J Clin Oncol 24(18):2903–2909. https://doi.org/10.1200/jco.2005.05.0245
Thuss-Patience PC, Kretzschmar A, Bichev D et al (2011) Survival advantage for irinotecan versus best supportive care as second-line chemotherapy in gastric cancer—a randomised phase III study of the Arbeitsgemeinschaft Internistische Onkologie (AIO). Eur J Cancer 47(15):2306–2314. https://doi.org/10.1016/j.ejca.2011.06.002
Kang JH, Lee SI, Lim do H et al (2012) Salvage chemotherapy for pretreated gastric cancer: a randomized phase III trial comparing chemotherapy plus best supportive care with best supportive care alone. J Clin Oncol 30(13):1513–1518. https://doi.org/10.1200/jco.2011.39.4585
Ford HE, Marshall A, Bridgewater JA et al (2014) Docetaxel versus active symptom control for refractory oesophagogastric adenocarcinoma (COUGAR-02): an open-label, phase 3 randomised controlled trial. Lancet Oncol 15(1):78–86. https://doi.org/10.1016/s1470-2045(13)70549-7
Wilke H, Muro K, Van Cutsem E et al (2014) Ramucirumab plus paclitaxel versus placebo plus paclitaxel in patients with previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (RAINBOW): a double-blind, randomised phase 3 trial. Lancet Oncol 15(11):1224–1235. https://doi.org/10.1016/s1470-2045(14)70420-6
Fuchs CS, Tomasek J, Yong CJ et al (2014) Ramucirumab monotherapy for previously treated advanced gastric or gastro-oesophageal junction adenocarcinoma (REGARD): an international, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet 383(9911):31–39. https://doi.org/10.1016/s0140-6736(13)61719-5
Hironaka S, Ueda S, Yasui H et al (2013) Randomized, open-label, phase III study comparing irinotecan with paclitaxel in patients with advanced gastric cancer without severe peritoneal metastasis after failure of prior combination chemotherapy using fluoropyrimidine plus platinum: WJOG 4007 trial. J Clin Oncol 31(35):4438–4444. https://doi.org/10.1200/jco.2012.48.5805
Shitara K, Matsuo K, Muro K, Doi T, Ohtsu A (2014) Correlation between overall survival and other endpoints in clinical trials of second-line chemotherapy for patients with advanced gastric cancer. Gastric Cancer 17(2):362–370. https://doi.org/10.1007/s10120-013-0274-6
Shitara K, Muro K, Shimada Y et al (2016) Subgroup analyses of the safety and efficacy of ramucirumab in Japanese and Western patients in RAINBOW: a randomized clinical trial in second-line treatment of gastric cancer. Gastric Cancer 19(3):927–938. https://doi.org/10.1007/s10120-015-0559-z
Kang YK, Boku N, Satoh T et al (2017) Nivolumab in patients with advanced gastric or gastro-oesophageal junction cancer refractory to, or intolerant of, at least two previous chemotherapy regimens (ONO-4538-12, ATTRACTION-2): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet 390:2461–2471. https://doi.org/10.1016/S0140-6736(17)31827-5
Takashima A, Iizumi S, Boku N (2017) Survival after failure of first-line chemotherapy in advanced gastric cancer patients: differences between Japan and the rest of the world. Jpn J Clin Oncol 47(7):583–589. https://doi.org/10.1093/jjco/hyx044
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing. https://www.r-project.org/. Accessed 22 Dec 2017
Van Cutsem E, Moiseyenko VM, Tjulandin S et al (2006) Phase III study of docetaxel and cisplatin plus fluorouracil compared with cisplatin and fluorouracil as first-line therapy for advanced gastric cancer: a report of the V325 Study Group. J Clin Oncol 24(31):4991–4997. https://doi.org/10.1200/jco.2006.06.8429
Al-Batran SE, Hartmann JT, Probst S et al (2008) Phase III trial in metastatic gastroesophageal adenocarcinoma with fluorouracil, leucovorin plus either oxaliplatin or cisplatin: a study of the Arbeitsgemeinschaft Internistische Onkologie. J Clin Oncol 26(9):1435–1442. https://doi.org/10.1200/jco.2007.13.9378
Koizumi W, Narahara H, Hara T et al (2008) S-1 plus cisplatin versus S-1 alone for first-line treatment of advanced gastric cancer (SPIRITS trial): a phase III trial. Lancet Oncol 9(3):215–221. https://doi.org/10.1016/s1470-2045(08)70035-4
Boku N, Yamamoto S, Fukuda H et al (2009) Fluorouracil versus combination of irinotecan plus cisplatin versus S-1 in metastatic gastric cancer: a randomised phase 3 study. Lancet Oncol 10(11):1063–1069. https://doi.org/10.1016/s1470-2045(09)70259-1
Ajani JA, Rodriguez W, Bodoky G et al (2010) Multicenter phase III comparison of cisplatin/S-1 with cisplatin/infusional fluorouracil in advanced gastric or gastroesophageal adenocarcinoma study: the FLAGS trial. J Clin Oncol 28(9):1547–1553. https://doi.org/10.1200/jco.2009.25.4706
Bang YJ, Van Cutsem E, Feyereislova A et al (2010) Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): a phase 3, open-label, randomised controlled trial. Lancet 376(9742):687–697. https://doi.org/10.1016/s0140-6736(10)61121-x
Narahara H, Iishi H, Imamura H et al (2011) Randomized phase III study comparing the efficacy and safety of irinotecan plus S-1 with S-1 alone as first-line treatment for advanced gastric cancer (study GC0301/TOP-002). Gastric Cancer 14(1):72–80. https://doi.org/10.1007/s10120-011-0009-5
Ohtsu A, Shah MA, Van Cutsem E et al (2011) Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: a randomized, double-blind, placebo-controlled phase III study. J Clin Oncol 29(30):3968–3976. https://doi.org/10.1200/jco.2011.36.2236
Lordick F, Kang YK, Chung HC et al (2013) Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): a randomised, open-label phase 3 trial. Lancet Oncol 14(6):490–499. https://doi.org/10.1016/s1470-2045(13)70102-5
Guimbaud R, Louvet C, Ries P et al (2014) Prospective, randomized, multicenter, phase III study of fluorouracil, leucovorin, and irinotecan versus epirubicin, cisplatin, and capecitabine in advanced gastric adenocarcinoma: a French intergroup (Federation Francophone de Cancerologie Digestive, Federation Nationale des Centres de Lutte Contre le Cancer, and Groupe Cooperateur Multidisciplinaire en Oncologie) study. J Clin Oncol 32(31):3520–3526. https://doi.org/10.1200/jco.2013.54.1011
Koizumi W, Kim YH, Fujii M et al (2014) Addition of docetaxel to S-1 without platinum prolongs survival of patients with advanced gastric cancer: a randomized study (START). J Cancer Res Clin Oncol 140(2):319–328. https://doi.org/10.1007/s00432-013-1563-5
Ochenduszko S, Puskulluoglu M, Konopka K et al (2015) Comparison of efficacy and safety of first-line palliative chemotherapy with EOX and mDCF regimens in patients with locally advanced inoperable or metastatic HER2-negative gastric or gastroesophageal junction adenocarcinoma: a randomized phase 3 trial. Med Oncol 32(10):242. https://doi.org/10.1007/s12032-015-0687-7
Ryu MH, Baba E, Lee KH et al (2015) Comparison of two different S-1 plus cisplatin dosing schedules as first-line chemotherapy for metastatic and/or recurrent gastric cancer: a multicenter, randomized phase III trial (SOS). Ann Oncol 26(10):2097–2101. https://doi.org/10.1093/annonc/mdv316
Shen L, Li J, Xu J et al (2015) Bevacizumab plus capecitabine and cisplatin in Chinese patients with inoperable locally advanced or metastatic gastric or gastroesophageal junction cancer: randomized, double-blind, phase III study (AVATAR study). Gastric Cancer 18(1):168–176. https://doi.org/10.1007/s10120-014-0351-5
Yamada Y, Higuchi K, Nishikawa K et al (2015) Phase III study comparing oxaliplatin plus S-1 with cisplatin plus S-1 in chemotherapy-naive patients with advanced gastric cancer. Ann Oncol 26(1):141–148. https://doi.org/10.1093/annonc/mdu472
Ohtsu A, Ajani JA, Bai YX et al (2013) Everolimus for previously treated advanced gastric cancer: results of the randomized, double-blind, phase III GRANITE-1 study. J Clin Oncol 31(31):3935–3943. https://doi.org/10.1200/jco.2012.48.3552
Higuchi K, Tanabe S, Shimada K et al (2014) Biweekly irinotecan plus cisplatin versus irinotecan alone as second-line treatment for advanced gastric cancer: a randomised phase III trial (TCOG GI-0801/BIRIP trial). Eur J Cancer 50(8):1437–1445. https://doi.org/10.1016/j.ejca.2014.01.020
Satoh T, Xu RH, Chung HC et al (2014) Lapatinib plus paclitaxel versus paclitaxel alone in the second-line treatment of HER2-amplified advanced gastric cancer in Asian populations: TyTAN—a randomized, phase III study. J Clin Oncol 32(19):2039–2049. https://doi.org/10.1200/jco.2013.53.6136
Nishikawa K, Fujitani K, Inagaki H et al (2015) Randomised phase III trial of second-line irinotecan plus cisplatin versus irinotecan alone in patients with advanced gastric cancer refractory to S-1 monotherapy: TRICS trial. Eur J Cancer 51(7):808–816. https://doi.org/10.1016/j.ejca.2015.02.009
Tanabe K, Fujii M, Nishikawa K et al (2015) Phase II/III study of second-line chemotherapy comparing irinotecan-alone with S-1 plus irinotecan in advanced gastric cancer refractory to first-line treatment with S-1 (JACCRO GC-05). Ann Oncol 26(9):1916–1922. https://doi.org/10.1093/annonc/mdv265
Cunningham D, Starling N, Rao S et al (2008) Capecitabine and oxaliplatin for advanced esophagogastric cancer. N Engl J Med 358(1):36–46. https://doi.org/10.1056/NEJMoa073149
Dank M, Zaluski J, Barone C et al (2008) Randomized phase III study comparing irinotecan combined with 5-fluorouracil and folinic acid to cisplatin combined with 5-fluorouracil in chemotherapy naive patients with advanced adenocarcinoma of the stomach or esophagogastric junction. Ann Oncol 19(8):1450–1457. https://doi.org/10.1093/annonc/mdn166
Kang YK, Kang WK, Shin DB et al (2009) Capecitabine/cisplatin versus 5-fluorouracil/cisplatin as first-line therapy in patients with advanced gastric cancer: a randomised phase III noninferiority trial. Ann Oncol 20(4):666–673. https://doi.org/10.1093/annonc/mdn717
Lee KH, Hyun MS, Kim HK et al (2009) Randomized, multicenter, phase III trial of heptaplatin 1-hour infusion and 5-fluorouracil combination chemotherapy comparing with cisplatin and 5-fluorouracil combination chemotherapy in patients with advanced gastric cancer. Cancer Res Treat 41(1):12–18. https://doi.org/10.4143/crt.2009.41.1.12
Shirao K, Boku N, Yamada Y et al (2013) Randomized Phase III study of 5-fluorouracil continuous infusion vs. sequential methotrexate and 5-fluorouracil therapy in far advanced gastric cancer with peritoneal metastasis (JCOG0106). Jpn J Clin Oncol 43(10):972–980. https://doi.org/10.1093/jjco/hyt114
Waddell T, Chau I, Cunningham D et al (2013) Epirubicin, oxaliplatin, and capecitabine with or without panitumumab for patients with previously untreated advanced oesophagogastric cancer (REAL3): a randomised, open-label phase 3 trial. Lancet Oncol 14(6):481–489. https://doi.org/10.1016/s1470-2045(13)70096-2
Hecht JR, Bang Y-J, Qin S, Chung H-C, Xu J-M, Park JO (2013) Lapatinib in combination with capecitabine plus oxaliplatin (CapeOx) in HER2-positive advanced or metastatic gastric, esophageal, or gastroesophageal adenocarcinoma (AC): the TRIO-013/LOGiC Trial. J Clin Oncol 34(5):suppl; abstr LBA4001. https://doi.org/10.1200/jco.2013.31.18_suppl.lba4001
Xu R-h, Sun G-p, Lu H-s, Peng LY, Xu J-m, Zhong M-z (2013) A phase III study of S-1 plus cisplatin versus fluorouracil plus cisplatin in patients with advanced gastric or gastroesophageal junction adenocarcinoma. J Clin Oncol 31(15):suppl 4025–4025. https://doi.org/10.1200/jco.2013.31.15_suppl.4025
Li J, Qin S, Xu J et al (2014) Phase III study of apatinib in advanced gastric cancer: a randomized, double-blind, placebo-controlled trial. Ann Oncol 25(suppl_2):ii117. https://doi.org/10.1093/annonc/mdu193.29
Ajani JA, Abramov M, Bondar V, Bondarenko I, Shparyk YV (2015) Untreated metastatic diffuse gastric adenocarcinoma (DGAC): randomized phase III study of S-1 and cisplatin vs. 5-FU and cisplatin (the DIGEST trial). J Clin Oncol 33(15):suppl 4015–4015. https://doi.org/10.1200/jco.2015.33.15_suppl.4015
Cunningham D, Tebbutt NC, Davidenko I, Murad AM, Al-Batran S-E (2015) Phase III, randomized, double-blind, multicenter, placebo (P)-controlled trial of rilotumumab (R) plus epirubicin, cisplatin and capecitabine (ECX) as first-line therapy in patients (pts) with advanced MET-positive (pos) gastric or gastroesophageal junction (G/GEJ) cancer: RILOMET-1 study. J Clin Oncol 33(15):suppl 4000–4000. https://doi.org/10.1200/jco.2015.33.15_suppl.4000
Hwang IG, Lee HR, Lee HY, Ji JH, Kang JH, Lee SI (2015) First-line capecitabine (X) monotherapy versus capecitabine plus oxaliplatin (XELOX) in elderly patients with advanced gastric cancer (AGC): results from the first interim analysis. J Clin Oncol 33(15):suppl 4051–4051. https://doi.org/10.1200/jco.2015.33.15_suppl.4051
Nishikawa K, Yoshino S, Morita S, Takahashi T, Sakata K, Nagao J (2015) A randomized phase III study of S-1 alone versus S-1 plus immunomodulator lentinan for unresectable or recurrent gastric cancer (JFMC36–0701). Eur J Cancer 51(Supplement 3):S442
Japanese Gastric Cancer Association (2017) Japanese gastric cancer treatment guidelines 2014 (ver. 4). Gastric Cancer 20:1–19. https://doi.org/10.1007/s10120-016-0622-4
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflicts of interest.
Ethical approval
This study itself does not involve any interventions on human participants or animals performed by any of the authors. Ethical approval was not required for this study because only publicly available data were used.
Informed consent
Informed consent was not required for this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Iizumi, S., Takashima, A., Sakamaki, K. et al. Survival impact of post-progression chemotherapy in advanced gastric cancer: systematic review and meta-analysis. Cancer Chemother Pharmacol 81, 981–989 (2018). https://doi.org/10.1007/s00280-018-3569-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-018-3569-9