Abstract
Purpose
QT assessment of oncology drugs is generally challenging because they are genotoxic and, of necessity, they require multisite evaluation in cancer patients. Lenvatinib is not genotoxic, therefore, this thorough QT (TQT) study with lenvatinib, a multityrosine kinase inhibitor, was undertaken utilizing healthy volunteers and concentration-effect modeling to project the TQT effect at high plasma levels.
Methods
Fifty-two healthy subjects randomly received single doses of lenvatinib 32 mg, placebo, or moxifloxacin 400 mg in a three-way crossover study. Serial electrocardiograms were recorded, and the effect on placebo-corrected change-from-baseline QTcF (∆∆QTcF) was evaluated. The relationship between lenvatinib plasma concentrations and QTcF was analyzed with linear mixed-effects modeling.
Results
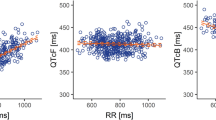
Lenvatinib mildly lowered the heart rate by 5–8 bpm during the first 12 h after dosing. ∆∆QTcF was shortened with a peak effect of −5.72 ms (90 % confidence interval (90 % CI) −7.76 to −3.69 ms) at 6 h postdosing. The upper bound of mean ∆∆QTcF did not exceed 2 ms at any time point postdosing. A concentration-dependent effect of lenvatinib on ∆∆QTcF was identified with an estimated population intercept of −2.96 ms (90 % CI −4.49 to −1.43 ms; P = 0.0016) and a negative slope of −0.0045 (90 % CI −4.49 to −1.43) ms per ng/mL, respectively. The safety profile after a single dose of lenvatinib was acceptable, with adverse events (AEs) of mild-to-moderate severity and no serious AEs.
Conclusions
Lenvatinib had no clinically relevant effect on the QTc interval. Concentration-effect modeling supports the lack of QTc prolongation at high plasma concentrations.





Similar content being viewed by others
References
Hanahan D, Weinberg RA (2000) The hallmarks of cancer. Cell 100:57–70
Matsui J, Yamamoto Y, Funahashi Y, Tsuruoka A, Watanabe T, Wakabayashi T, Uenaka T, Asada M (2008) E7080, a novel inhibitor that targets multiple kinases, has potent antitumor activities against stem cell factor producing human small cell lung cancer H146, based on angiogenesis inhibition. Int J Cancer 122:664–671
Yamada K, Yamamoto N, Yamada Y, Nokihara H, Fujiwara Y, Hirata T, Koizumi F, Nishio K, Koyama N, Tamura T (2011) Phase I dose-escalation study and biomarker analysis of E7080 in patients with advanced solid tumors. Clin Cancer Res 17:2528–2537
Boss DS, Glen H, Beijnen JH, Keesen M, Morrison R, Tait B, Copalu W, Mazur A, Wanders J, O’Brien JP, Schellens JH, Evans TR (2012) A phase I study of E7080, a multitargeted tyrosine kinase inhibitor, in patients with advanced solid tumours. Br J Cancer 106:1598–1604
Shah RR (2002) The significance of QT interval in drug development. Br J Clin Pharmacol 54:188–202
Shah RR (2002) Drug-induced prolongation of the QT interval: why the regulatory concern? Fundam Clin Pharmacol 16:119–124
Shah RR (2002) Drug-induced prolongation of the QT interval: regulatory dilemmas and implications for approval and labelling of a new chemical entity. Fundam Clin Pharmacol 16:147–156
Food and Drug Administration, HHS (2005) International Conference on Harmonisation; guidance on E14 clinical evaluation of QT/QTc interval prolongation and proarrhythmic potential for non-antiarrhythmic drugs; availability. Notice. Fed Regist 70:61134–61135
Rock EP, Finkle J, Fingert HJ, Booth BP, Garnett CE, Grant S, Justice RL, Kovacs RJ, Kowey PR, Rodriguez I, Sanhai WR, Strnadova C, Targum SL, Tsong Y, Uhl K, Stockbridge N (2009) Assessing proarrhythmic potential of drugs when optimal studies are infeasible. Am Heart J 157(827–836):e1
Garnett CE, Zhu H, Malik M, Fossa AA, Zhang J, Badilini F, Li J, Darpö B, Sager P, Rodriguez I (2012) Methodologies to characterize the QT/corrected QT interval in the presence of drug-induced heart rate changes or other autonomic effects. Am Heart J 163:912–930
Couderc JP, Garnett C, Li M, Handzel R, McNitt S, Xia X, Polonsky S, Zareba W (2011) Highly automated QT measurement techniques in 7 thorough QT studies implemented under ICH E14 guidelines. Ann Noninvasive Electrocardiol 16:13–24
E14 Implementation Working Group (2012) ICH E14 guideline: the clinical evaluation of QT/QTc interval prolongation and proarrhythmic potential for non-antiarrhythmic drugs: questions & answers (R1). http://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Efficacy/E14/E14_Q_As_R1_step4.pdf. Accessed 23 May 2013
Hochberg Y, Benjamini Y (1990) More powerful procedures for multiple significance testing. Stat Med 9:811–818
Florian JA, Tornøe CW, Brundage R, Parekh A, Garnett CE (2011) Population pharmacokinetic and concentration–QTc models for moxifloxacin: pooled analysis of 20 thorough QT studies. J Clin Pharmacol 51:1152–1162
Garnett CE, Beasley N, Bhattaram VA, Jadhav PR, Madabushi R, Stockbridge N, Tornøe CW, Wang Y, Zhu H, Gobburu JV (2008) Concentration-QT relationships play a key role in the evaluation of proarrhythmic risk during regulatory review. J Clin Pharmacol 48:13–18
Lesimple T, Edeline J, Carrothers TJ, Cvitkovic F, Darpo B, Delord JP, Léna H, Penel N, Edwards GJ, Law K, Wanders J, Kristensen A, Reyderman L (2013) A phase I, open-label, single-arm study for QT assessment of eribulin mesylate in patients with advanced solid tumors. Invest New Drugs 31:900–909
Bello CL, Rosen LS, Mulay M, van Vugt A, Dinolfo M, Levine S, Huang X, Fingert HJ, Toh M, Baum CM (2007) Characterization of electrocardiographic QTc interval in patients (pts) with advanced solid tumors: pharmacokinetic-pharmacodynamic evaluation of sunitinib. Eur J Cancer (suppl; abstr 709)
Krishnaswami S, Kudlacz E, Wang R, Chan G (2011) A supratherapeutic dose of the Janus kinase inhibitor tasocitinib (CP-690,550) does not prolong QTc interval in healthy participants. J Clin Pharmacol 51:1256–1263
Abbas R, Hug BA, Leister C, Sonnichsen D (2012) A randomized, crossover, placebo- and moxifloxacin-controlled study to evaluate the effects of bosutinib (SKI-606), a dual Src/Abl tyrosine kinase inhibitor, on cardiac repolarization in healthy adult subjects. Int J Cancer 131:E304–E311
Darpo B, Agin M, Kazierad DJ, Layton G, Muirhead G, Gray P, Jorkasky DK (2006) Man versus machine: is there an optimal method for QT measurements in thorough QT studies? J Clin Pharmacol 46:598–612
Tolcher AW, Appleman LJ, Shapiro GI, Mita AC, Cihon F, Mazzu A, Sundaresan PR (2011) A phase I open-label study evaluating the cardiovascular safety of sorafenib in patients with advanced cancer. Cancer Chemother Pharmacol 67:751–764
Shumaker R, Aluri J, Fan J, Martinez G, Ren M, Chen K. Evaluation of the effects of formulation and food on the pharmacokinetics of lenvatinib (E7080) in healthy volunteers, Int J Clin Pharmacol Ther (accepted)
Dubbelman et al. Poster presented at the Scientific Meeting of the Dutch Society of Clinical Pharmacology & Biopharmacy (NVKFB), 30 March 2012, Utrecht, The Netherlands
Shumaker R et al. Poster presented at the 24th EORTC-NCI-AACR Symposium on Molecular Targets and Cancer Therapeutics, 6–9 November 2012, Dublin, Ireland
Guidance for Industry. Bioavailability and bioequivalence studies for orally administered drug products—general considerations. U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), March 2003 BP Revision 1
Garnett CE, Zhu H, Malik M, Fossa AA, Zhang J, Badilini F, Li J, Darpö B, Sager P, Rodriguez I (2012) Methodologies to characterize the QT/corrected QT interval in the presence of drug-induced heart rate changes or other autonomic effects. Am Heart J 163:912–930
Darpo B, Fossa AA, Couderc JP, Zhou M, Schreyer A, Ticktin M, Zapesochny A (2011) Improving the precision of QT measurements. Cardiol J 18:401–410
Shumaker R, Fan J, Martinez G, Chen K (2012) Comparative bioavailability study of a 10 mg capsule and a 10 mg tablet of lenvatinib (E7080) in healthy subjects. In 113th ASCPT Annual Meeting 2012; 12–17 March; National Harbor
Acknowledgments
The authors thank the lenvatinib clinical team for their assistance in the conduct, reporting, and quality control of the studies; the study site investigator, Steven F. Komjathy, MD; the study site, Charles River Clinical Services northwest, Inc., Tacoma, WA, and their staff, for conducting the study; and the study subjects for their participation. The authors gratefully acknowledge editorial contributions by Phase Five Communications Inc.
Conflict of interest
B Darpo: consultant to Eisai Product Creation Systems and iCardiac Technologies, Inc.; stock holdings of iCardiac. M Zhou: employee of iCardiac Technologies, Inc. RC Shumaker, J Fan, M Ren, G Martinez, J Aluri: employees of Eisai Product Creation Systems.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shumaker, R.C., Zhou, M., Ren, M. et al. Effect of lenvatinib (E7080) on the QTc interval: results from a thorough QT study in healthy volunteers. Cancer Chemother Pharmacol 73, 1109–1117 (2014). https://doi.org/10.1007/s00280-014-2444-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-014-2444-6