Abstract
Purpose
Sequence-dependent improved efficacy of topoisomerase I followed by topoisomerase 2 inhibitors was assessed in a randomized phase II study in extensive-stage small-cell lung cancer (SCLC).
Methods
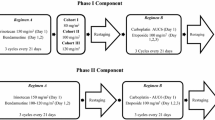
Patients with previously untreated extensive-stage SCLC with measurable disease, ECOG performance status of 0–3 and stable brain metastases were eligible. Arm A consisted of topotecan (0.75 mg/m2) on days 1, 2 and 3, etoposide (70 mg/m2) and cisplatin (20 mg/m2) (PET) on days 8, 9 and 10 in a 3-week cycle. Arm B consisted of irinotecan (50 mg/m2) and cisplatin (20 mg/m2) on days 1 and 8 followed by etoposide (85 mg/m2 PO bid) on days 3 and 10 (PIE) in a 3-week cycle.
Results
We enrolled 140 patients and randomized 66 eligible patients to each arm. Only 54.5 % of all patients completed the planned maximum 6 cycles. There were grade ≥3 treatment-related adverse events in approximately 70 % of the patients on both arms including 6 treatment-related grade 5 events. The overall response rates (CR + PR) were 69.7 % (90 % CI 59.1–78.9, 95 % CI 57.1–80.4 %) for arm A and 57.6 % (90 % CI 46.7–67.9, 95 % CI 44.8–69.7 %) for arm B. The median progression-free survival and overall survival were 6.4 months (95 % CI 5.4–7.5 months) and 11.9 months (95 % CI 9.6–13.7 months) for arm A and 6.0 months (95 % CI 5.4–7.0 months) and 11.0 months (95 % CI 8.6–13.1 months) for arm B.
Conclusion
Sequential administration of topoisomerase inhibitors did not improve on the historical efficacy of standard platinum-doublet chemotherapy for extensive-stage SCLC.




Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A (2012) Cancer statistics, 2012. CA Cancer J Clin 62(1):10–29. doi:10.3322/caac.20138
Owonikoko TK, Ragin CC, Belani CP, Oton AB, Gooding WE, Taioli E, Ramalingam SS (2007) Lung cancer in elderly patients: an analysis of the surveillance, epidemiology, and end results database. J Clin Oncol 25(35):5570–5577
Abrams J, Doyle LA, Aisner J (1988) Staging, prognostic factors, and special considerations in small cell lung cancer. Semin Oncol 15(3):261–277
Zelen M (1973) Keynote address on biostatistics and data retrieval. Cancer chemotherapy reports part 3 4 (2):31–42
Aisner J, Alberto P, Bitran J, Comis R, Daniels J, Hansen H, Ikegami H, Smyth J (1983) Role of chemotherapy in small cell lung cancer: a consensus report of the International Association for the study of lung cancer workshop. Cancer Treat Rep 67(1):37–43
Turrisi AT 3rd, Kim K, Blum R, Sause WT, Livingston RB, Komaki R, Wagner H, Aisner S, Johnson DH (1999) Twice-daily compared with once-daily thoracic radiotherapy in limited small-cell lung cancer treated concurrently with cisplatin and etoposide. N Engl J Med 340(4):265–271
Sundstrom S, Bremnes RM, Kaasa S, Aasebo U, Hatlevoll R, Dahle R, Boye N, Wang M, Vigander T, Vilsvik J, Skovlund E, Hannisdal E, Aamdal S (2002) Cisplatin and etoposide regimen is superior to cyclophosphamide, epirubicin, and vincristine regimen in small-cell lung cancer: results from a randomized phase III trial with 5 years’ follow-up. J Clin Oncol 20(24):4665–4672
Owonikoko TK, Behera M, Chen Z, Bhimani C, Curran WJ, Khuri FR, Ramalingam SS (2012) A systematic analysis of efficacy of second-line chemotherapy in sensitive and refractory small-cell lung cancer. J Thorac Oncol 7(5):866–872. doi:10.1097/JTO.0b013e31824c7f4b
Giaccone G, Ferrati P, Donadio M, Testore F, Calciati A (1987) Reinduction chemotherapy in small cell lung cancer. Eur J Cancer Clin Oncol 23(11):1697–1699
Stuart-Harris R, Raghavan D, Fox RM, Peretz G, Crombie C, Teriana N, Young I, Bye P, Tiver K, Green D (1987) Chemotherapy for small cell lung cancer: induction and reinduction with VOCA. Aust N Z J Med 17(3):279–282
Johnson DH, Greco FA, Strupp J, Hande KR, Hainsworth JD (1990) Prolonged administration of oral etoposide in patients with relapsed or refractory small-cell lung cancer: a phase II trial. J Clin Oncol 8(10):1613–1617
Slichenmyer WJ, Rowinsky EK, Donehower RC, Kaufmann SH (1993) The current status of camptothecin analogues as antitumor agents. J Natl Cancer Inst 85(4):271–291
Masuda N, Fukuoka M, Kusunoki Y, Matsui K, Takifuji N, Kudoh S, Negoro S, Nishioka M, Nakagawa K, Takada M (1992) CPT-11: a new derivative of camptothecin for the treatment of refractory or relapsed small-cell lung cancer. J Clin Oncol 10(8):1225–1229
Ohe Y, Saijo N (2001) Topoisomerase I targeting agents in small-cell lung cancer. Curr Oncol Rep 3(2):170–178
Schiller JH, Adak S, Cella D, DeVore RF 3rd, Johnson DH (2001) Topotecan versus observation after cisplatin plus etoposide in extensive-stage small-cell lung cancer: E7593–a phase III trial of the eastern cooperative oncology group. J Clin Oncol 19(8):2114–2122
Noda K, Nishiwaki Y, Kawahara M, Negoro S, Sugiura T, Yokoyama A, Fukuoka M, Mori K, Watanabe K, Tamura T, Yamamoto S, Saijo N (2002) Irinotecan plus cisplatin compared with etoposide plus cisplatin for extensive small-cell lung cancer. N Engl J Med 346(2):85–91. doi:10.1056/NEJMoa003034
Hanna N, Bunn PA Jr, Langer C, Einhorn L, Guthrie T Jr, Beck T, Ansari R, Ellis P, Byrne M, Morrison M, Hariharan S, Wang B, Sandler A (2006) Randomized phase III trial comparing irinotecan/cisplatin with etoposide/cisplatin in patients with previously untreated extensive-stage disease small-cell lung cancer. J Clin Oncol 24(13):2038–2043
Lara PN Jr, Natale R, Crowley J, Lenz HJ, Redman MW, Carleton JE, Jett J, Langer CJ, Kuebler JP, Dakhil SR, Chansky K, Gandara DR (2009) Phase III Trial of Irinotecan/Cisplatin compared with Etoposide/Cisplatin in extensive-stage small-cell lung cancer: clinical and pharmacogenomic results from SWOG S0124. J Clin Oncol 27(15):2530–2535. doi:10.1200/jco.2008.20.1061
Rubin EH, Li TK, Duann P, Liu LF (1996) Cellular resistance to topoisomerase poisons. Cancer Treat Res 87:243–260
Rubin E, Wood V, Bharti A, Trites D, Lynch C, Hurwitz S, Bartel S, Levy S, Rosowsky A, Toppmeyer D et al (1995) A phase I and pharmacokinetic study of a new camptothecin derivative, 9-aminocamptothecin. Clin Cancer Res 1(3):269–276
Saleem A, Ibrahim N, Patel M, Li XG, Gupta E, Mendoza J, Pantazis P, Rubin EH (1997) Mechanisms of resistance in a human cell line exposed to sequential topoisomerase poisoning. Cancer Res 57(22):5100–5106
Mok TS, Wong H, Zee B, Yu KH, Leung TW, Lee TW, Yim A, Chan AT, Yeo W, Chak K, Johnson P (2002) A Phase I-II study of sequential administration of topotecan and oral etoposide (toposiomerase I and II inhibitors) in the treatment of patients with small cell lung carcinoma. Cancer 95(7):1511–1519. doi:10.1002/cncr.10836
Dowlati A, Levitan N, Gordon NH, Hoppel CL, Gosky DM, Remick SC, Ingalls ST, Berger SJ, Berger NA (2001) Phase II and pharmacokinetic/pharmacodynamic trial of sequential topoisomerase I and II inhibition with topotecan and etoposide in advanced non-small-cell lung cancer. Cancer Chemother Pharmacol 47(2):141–148
Aisner J, Musanti R, Beers S, Smith S, Locsin S, Rubin EH (2003) Sequencing topotecan and etoposide plus cisplatin to overcome topoisomerase I and II resistance: a pharmacodynamically based Phase I trial. Clin Cancer Res 9(7):2504–2509
Huisman C, Postmus PE, Giaccone G, Smit EF (2001) A phase I study of sequential intravenous topotecan and etoposide in lung cancer patients. Ann Oncol 12(11):1567–1573
Atkinson EN, Brown BW (1985) Confidence limits for probability of response in multistage phase II clinical trials. Biometrics 41(3):741–744
Schiller JH, Kim K, Hutson P, DeVore R, Glick J, Stewart J, Johnson D (1996) Phase II study of topotecan in patients with extensive-stage small-cell carcinoma of the lung: an Eastern Cooperative Oncology Group Trial. J Clinical Oncol 14(8):2345–2352
von Pawel J, Schiller JH, Shepherd FA, Fields SZ, Kleisbauer JP, Chrysson NG, Stewart DJ, Clark PI, Palmer MC, Depierre A, Carmichael J, Krebs JB, Ross G, Lane SR, Gralla R (1999) Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol 17(2):658–667
Hosomi Y, Shibuya M, Niho S, Ichinose Y, Kiura K, Sakai H, Takeda K, Kudo S, Eguchi K, Matsui K, Masuda N, Ando M, Watanabe K (2011) Phase II study of topotecan with cisplatin in Japanese patients with small cell lung cancer. Anticancer Res 31(10):3449–3456
Sorensen M, Lassen U, Jensen PB, Osterlind K, Jeppesen N, Jensen BB, Mellemgaard A, Rytter C, Langer SW (2008) Phase II study of a 3-day schedule with topotecan and cisplatin in patients with previously untreated small cell lung cancer and extensive disease. J Thorac Oncol 3(8):902–906. doi:10.1097/JTO.0b013e31817e0f58
Lyss AP, Herndon JE, 2nd, Lynch TJ, Jr., Turrisi AT, Watson DM, Grethlein SJ, Green MR (2002) Novel doublets in extensive-stage small-cell lung cancer: a randomized phase II study of topotecan plus cisplatin or paclitaxel (CALGB 9430). Clin Lung Cancer 3 (3):205–210; discussion 211–202
Felip E, Rosell R, Domine M, Santome L, Garrido P, Font A, Carrato A, Terrasa J, Vadell C, Mane JM, Baselga J (2003) Sequential dose-dense paclitaxel followed by topotecan in untreated extensive-stage small-cell lung cancer: a Spanish Lung Cancer Group phase II study. Ann Oncol 14(10):1549–1554
Jett JR, Hatfield AK, Hillman S, Bauman MD, Mailliard JA, Kugler JW, Morton RF, Marks RS, Levitt R (2003) Alternating chemotherapy with etoposide plus cisplatin and topotecan plus paclitaxel in patients with untreated, extensive-stage small cell lung carcinoma: a phase II trial of the North Central Cancer Treatment Group. Cancer 97(10):2498–2503. doi:10.1002/cncr.11377
Fink TH, Huber RM, Heigener DF, Eschbach C, Waller C, Steinhauer EU, Virchow JC, Eberhardt F, Schweisfurth H, Schroeder M, Ittel T, Hummler S, Banik N, Bogenrieder T, Acker T, Wolf M (2012) Topotecan/cisplatin compared with cisplatin/etoposide as first-line treatment for patients with extensive disease small-cell lung cancer: final results of a randomized phase III trial. J Thorac Oncol 7(9):1432–1439. doi:10.1097/JTO.0b013e318260de75
Eckardt JR, von Pawel J, Papai Z, Tomova A, Tzekova V, Crofts TE, Brannon S, Wissel P, Ross G (2006) Open-label, multicenter, randomized, phase III study comparing oral topotecan/cisplatin versus etoposide/cisplatin as treatment for chemotherapy-naive patients with extensive-disease small-cell lung cancer. J Clin Oncol 24(13):2044–2051. doi:10.1200/JCO.2005.03.3332
Baka S, Agelaki S, Kotsakis A, Veslemes M, Papakotoulas P, Agelidou M, Agelidou A, Tsaroucha E, Pavlakou G, Gerogianni A, Androulakis N, Vamvakas L, Kalbakis K, Mavroudis D, Georgoulias V (2010) Phase III study comparing sequential versus alternate administration of cisplatin-etoposide and topotecan as first-line treatment in small cell lung cancer. Anticancer Res 30(7):3031–3038
Schmittel A, Sebastian M, von Fischer Weikersthal L, Martus P, Gauler TC, Kaufmann C, Hortig P, Fischer JR, Link H, Binder D, Fischer B, Caca K, Eberhardt WE, Keilholz U (2011) A German multicenter, randomized phase III trial comparing irinotecan-carboplatin with etoposide-carboplatin as first-line therapy for extensive-disease small-cell lung cancer. Ann Oncol 22(8):1798–1804. doi:10.1093/annonc/mdq652
Schmittel A, Fischer von Weikersthal L, Sebastian M, Martus P, Schulze K, Hortig P, Reeb M, Thiel E, Keilholz U (2006) A randomized phase II trial of irinotecan plus carboplatin versus etoposide plus carboplatin treatment in patients with extended disease small-cell lung cancer. Ann Oncol 17(4):663–667. doi:10.1093/annonc/mdj137
Charpidou A, Tsagouli S, Tsimpoukis S, Vassias A, Makrilia N, Stratakos G, Gkiozos I, Syrigos K (2010) Triplet combination of carboplatin, irinotecan, and etoposide in the first-line treatment of extensive small-cell lung cancer: a single-institution phase II study. Anticancer Drugs 21(6):651–655. doi:10.1097/CAD.0b013e3283393718
Ihde DC, Mulshine JL, Kramer BS, Steinberg SM, Linnoila RI, Gazdar AF, Edison M, Phelps RM, Lesar M, Phares JC et al (1994) Prospective randomized comparison of high-dose and standard-dose etoposide and cisplatin chemotherapy in patients with extensive-stage small-cell lung cancer. J Clin Oncol 12(10):2022–2034
Hammond LA, Eckardt JR, Ganapathi R, Burris HA, Rodriguez GA, Eckhardt SG, Rothenberg ML, Weiss GR, Kuhn JG, Hodges S, Von Hoff DD, Rowinsky EK (1998) A phase I and translational study of sequential administration of the topoisomerase I and II inhibitors topotecan and etoposide. Clin Cancer Res 4(6):1459–1467
Saraiya B, Gounder M, Dutta J, Saleem A, Collazo C, Zimmerman L, Nazar A, Gharibo M, Schaar D, Lin Y, Shih W, Aisner J, Strair RK, Rubin EH (2008) Sequential topoisomerase targeting and analysis of mechanisms of resistance to topotecan in patients with acute myelogenous leukemia. Anticancer Drugs 19(4):411–420. doi:10.1097/CAD.0b013e3282f5218b
Acknowledgments
This study was conducted by the Eastern Cooperative Oncology Group (Robert L. Comis, M.D., Chair) and supported in part by Public Health Service Grants CA23318, CA66636, CA21115, CA107868, CA16116, CA13650 and from the National Cancer Institute, National Institutes of Health and the Department of Health and Human Services. Its content is solely the responsibility of the authors and does not necessarily represent the official views of the National Cancer Institute.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Owonikoko, T.K., Aisner, J., Wang, X.V. et al. E5501: phase II study of topotecan sequenced with etoposide/cisplatin, and irinotecan/cisplatin sequenced with etoposide for extensive-stage small-cell lung cancer. Cancer Chemother Pharmacol 73, 171–180 (2014). https://doi.org/10.1007/s00280-013-2338-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-013-2338-z