Abstract
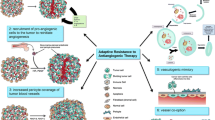
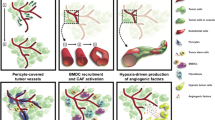
Central to the process of brain tumor development is angiogenesis, which involves a host of molecules and receptors. In recent years, antiangiogenic therapies have been developed and tested for their effectiveness against these tumors. Among them are inhibitors against vascular endothelial growth factor and its receptors, as well as inhibitors targeting the platelet-derived growth factor family, integrins, and histone deacetylase. While many have been shown to be effective with limited toxicity, some tumors are able to adopt escape mechanisms. Further research is needed in the development of effective multi-targeted agents to reduce these effects.

Similar content being viewed by others
References
Fidler IJ (1999) Critical determinants of cancer metastasis: rationale for therapy. Cancer Chemother Pharmaco 43:S3–S10
Jiménez B, Volpert OV (2001) Mechanistic insights on the inhibition of tumor angiogenesis. J Mol Med 78(12):663–672
Hicklin DJ, Ellis LM (2005) Role of the vascular endothelial growth factor pathway in tumor growth and angiogenesis. J Clin Oncol 23(5):1011–1027
Holmes K, Roberts OL, Thomas AM, Cross MJ (2007) Vascular endothelial growth factor receptor-2: structure, function, intracellular signalling and therapeutic inhibition. Cell Signal 19(10):2003–2012
Bautch VL, Redick SD, Scalia A, Harmaty M, Carmeliet P, Rapoport R (2000) Characterization of the vasculogenic block in the absence of vascular endothelial growth factor-A. Blood 95(6):1979–1987
Fong G-H, Rossant J, Gertsenstein M, Breitman ML (1995) Role of the Flt-1 receptor tyrosine kinase in regulating the assembly of vascular endothelium. Nature 376(6535):66–70
Shalaby F, Rossant J, Yamaguchi TP et al (1995) Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 376(6535):62–66
Zagzag D, Amirnovin R, Greco MA et al (2000) Vascular apoptosis and involution in gliomas precede neovascularization: a novel concept for glioma growth and angiogenesis. Lab Invest 80(6):837–849
Zagzag D, Lukyanov Y, Lan L et al (2006) Hypoxia-inducible factor 1 and VEGF upregulate CXCR4 in glioblastoma: implications for angiogenesis and glioma cell invasion. Lab Invest 86(12):1221–1232
Vredenburgh J, Desjardins A, Herndon J et al (2007) Bevacizumab plus irinotecan in recurrent glioblastoma multiforme. J Clin Oncol 25:4722–4729
Friedman HS, Prados MD, Wen PY et al (2009) Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol 27(28):4733–4740
Nghiemphu PL, Liu W et al (2009) Bevacizumab and chemotherapy for recurrent glioblastoma. Neurology 72(14):1217–1222
Kerber M, Reiss Y et al (2008) Flt-1 signaling in macrophages promotes glioma growth in vivo. Cancer Res 68(18):7342–7351
Geng L, Donnelly E et al (2001) Inhibition of vascular endothelial growth factor receptor signaling leads to reversal of tumor resistance to radiotherapy. Cancer Res 61(6):2413–2419
Pietsch T, Valter MM, Wolf HK et al (1997) Expression and distribution of vascular endothelial growth factor protein in human brain tumors. Acta Neuropathol 93(2):109–117
Caye-Thomasen P et al (2005) VEGF and VEGF receptor-1 concentration in vestibular schwannoma homogenates correlates to tumor growth rate. [Miscellaneous Article]. Otol Neurotol 26(1):98–101
Hoch RV, Soriano P (2003) Roles of PDGF in animal development. Development 130(20):4769–4784
Andrae J, Gallini R et al (2008) Role of platelet-derived growth factors in physiology and medicine. Genes Dev 22(10):1276–1312
Charles N, Ozawa T et al (2010) Perivascular nitric oxide activates notch signaling and promotes stem-like character in PDGF-induced glioma cells. Cell Stem Cell 6(2):141–152
Lokker NA, Sullivan CM et al (2002) Platelet-derived growth factor (PDGF) autocrine signaling regulates survival and mitogenic pathways in glioblastoma cells. Cancer Res 62(13):3729–3735
Hynes RO (2002) Integrins: bidirectional, allosteric signaling machines. Cell 110(6):673–687
Zhong J, Paul A et al (2010) Mesenchymal migration as a therapeutic target in glioblastoma. J Oncol 2010(21):430142
Kanamori M, Kawaguchi T et al (2006) Intracranial microenvironment reveals independent opposing functions of host αVβ3 expression on glioma growth and angiogenesis. J Biol Chem 281(48):37256–37264
Schnell O, Krebs B et al (2008) Expression of integrin αvβ3 in gliomas correlates with tumor grade and is not restricted to tumor vasculature. Brain Patho 18(3):378–386
Puduvalli V (2001) Brain metastases: biology and the role of the brain microenvironment. Curr Oncol Rep 3(6):467–475
Weber GF, Ashkar S (2000) Molecular mechanisms of tumor dissemination in primary and metastatic brain cancers. Brain Res Bull 53(4):421–424
Ouaissi M, Ouaissi A (2006) Histone deacetylase enzymes as potential drug targets in cancer and parasitic diseases. J Biomed Biotechnol 2006:13474
Campos B, Bermejo JL et al (2011) Expression of nuclear receptor corepressors and class I histone deacetylases in astrocytic gliomas. Cancer Sci 102(2):387–392
Sharma V, Koul N et al (2010) HDAC inhibitor, scriptaid, induces glioma cell apoptosis through JNK activation and inhibits telomerase activity. J Cell Mol Med 14(8):2151–2161
Thudi NK, Shu ST et al (2011) Development of a brain metastatic canine prostate cancer cell line. Prostate 71(12):1251–1263
Vredenburgh JJ, Desjardins A et al (2007) Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res 13(4):1253–1259
Kreisl TN, Kim L et al (2009) Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol 27(5):740–745
Narayana A, Kelly P et al (2009) Antiangiogenic therapy using bevacizumab in recurrent high-grade glioma: impact on local control and patient survival. J Neurosurgery 110(1):173–180
Norden AD, Young GS et al (2008) Bevacizumab for recurrent malignant gliomas. Neurology 70(10):779–787
Lai A, Tran A et al (2011) Phase II study of bevacizumab plus temozolomide during and after radiation therapy for patients with newly diagnosed glioblastoma multiforme. J Clin Oncol 29(2):142–148
Narayana A, Golfinos JG et al (2008) Feasibility of using bevacizumab with radiation therapy and temozolomide in newly diagnosed high-grade glioma. Int J Rad Oncol Biol Phys 72(2):383–389
Vredenburgh JJ, Desjardins A et al (2012) Addition of bevacizumab to standard radiation therapy and daily temozolomide is associated with minimal toxicity in newly diagnosed glioblastoma multiforme. Int J Rad Oncol Biol Phys 82(1):58–66
Narayana A, Gruber D et al (2012) A clinical trial of bevacizumab, temozolomide, and radiation for newly diagnosed glioblastoma. J Neurosurgery 116(2):341–345
Chamberlain MC, Johnston S (2009) Bevacizumab for recurrent alkylator-refractory anaplastic oligodendroglioma. Cancer 115(8):1734–1743
Mautner V-F, Nguyen R et al (2010) Bevacizumab induces regression of vestibular schwannomas in patients with neurofibromatosis type 2. Neuro-Oncol 12(1):14–18
Plotkin SR, Stemmer-Rachamimov AO et al (2009) Hearing improvement after bevacizumab in patients with neurofibromatosis type 2. N Engl J Med 361(4):358–367
Mautner V-F, Nguyen R et al (2010) Radiographic regression of vestibular schwannomas induced by bevacizumab treatment: sustain under continuous drug application and rebound after drug discontinuation. Ann Oncol 21(11):2294–2295
Puchner MJA, Hans VH et al (2010) Bevacizumab-induced regression of anaplastic meningioma. Ann Oncol 21(12):2445–2446
Goutagny S, Raymond E et al (2011) Radiographic regression of cranial meningioma in a NF2 patient treated by bevacizumab. Ann Oncol 22(4):990–991
Socinski MA, Langer CJ et al (2009) Safety of bevacizumab in patients with non–small-cell lung cancer and brain metastases. J Clin Oncol 27(31):5255–5261
Labidi SI, Bachelot T et al (2009) Bevacizumab and paclitaxel for breast cancer patients with central nervous system metastases: a case series. Clin Breast Cancer 9(2):118–121
Wong ET, Huberman M et al (2008) Bevacizumab reverses cerebral radiation necrosis. J Clin Oncol 26(34):5649–5650
Torcuator R, Zuniga R et al (2009) Initial experience with bevacizumab treatment for biopsy confirmed cerebral radiation necrosis. J Neuro-Oncol 94(1):63–68
Spratlin JL, Mulder KE et al (2010) Ramucirumab (IMC-1121B): a novel attack on angiogenesis. Fut Oncol 6(7):1085–1094
Spratlin JL, Cohen RB et al (2010) Phase I pharmacologic and biologic study of ramucirumab (IMC-1121B), a fully human immunoglobulin G1 monoclonal antibody targeting the vascular endothelial growth factor receptor-2. J Clin Oncol 28(5):780–787
Farrar CT, Kamoun WS et al (2011) Sensitivity of MRI tumor biomarkers to VEGFR inhibitor therapy in an orthotopic mouse glioma model. PLoS One 6(3):e17228
Dietrich J, Wang D et al (2009) Cediranib: profile of a novel anti-angiogenic agent in patients with glioblastoma. Expert Opin Investig Drugs 18(10):1549–1557
Batchelor TT, Duda DG et al (2010) Phase II study of cediranib, an oral pan–vascular endothelial growth factor receptor tyrosine kinase inhibitor, in patients with recurrent glioblastoma. J Clin Oncol 28(17):2817–2823
Wachsberger PR, Lawrence YR et al (2012) Epidermal growth factor receptor expression modulates antitumor efficacy of vandetanib or cediranib combined with radiotherapy in human glioblastoma xenografts. Int J Rad Oncol Biol Phys 82(1):483–491
Siegelin MD, Raskett CM et al (2010) Sorafenib exerts anti-glioma activity in vitro and in vivo. Neurosci Lett 478(3):165–170
Navis AC, Hamans BC et al (2011) Effects of targeting the VEGF and PDGF pathways in diffuse orthotopic glioma models. The J Pathol 223(5):626–634
Neyns B, Sadones J et al (2011) Phase II study of sunitinib malate in patients with recurrent high-grade glioma. J Neuro-Oncol 103(3):491–501
Hainsworth JD, Ervin T et al (2010) Concurrent radiotherapy and temozolomide followed by temozolomide and sorafenib in the first-line treatment of patients with glioblastoma multiforme. Cancer 116(15):3663–3669
Yoshikawa A, Nakada M et al (2012) Recurrent anaplastic meningioma treated by sunitinib based on results from quantitative proteomics. Neuropathol Appl Neurobiol 38(1):105–110
Amaravadi RK, Schuchter LM et al (2009) Phase II trial of temozolomide and sorafenib in advanced melanoma patients with or without brain metastases. Clin Cancer Res 15(24):7711–7718
Gore ME, Hariharan S et al (2011) Sunitinib in metastatic renal cell carcinoma patients with brain metastases. Cancer 117(3):501–509
Wuthrick EJ, Kamrava M et al (2011) A phase 1b trial of the combination of the antiangiogenic agent sunitinib and radiation therapy for patients with primary and metastatic central nervous system malignancies. Cancer 117(24):5548–5559
Maurer GD, Tritschler I et al (2009) Cilengitide modulates attachment and viability of human glioma cells, but not sensitivity to irradiation or temozolomide in vitro. Neuro-Oncol 11(6):747–756
Reardon DA, Fink KL et al (2008) Randomized phase II study of cilengitide, an integrin-targeting arginine-glycine-aspartic acid peptide, in recurrent glioblastoma multiforme. J Clin Oncol 26(34):5610–5617
Stupp R, Hegi ME et al (2010) Phase I/IIa study of cilengitide and temozolomide with concomitant radiotherapy followed by cilengitide and temozolomide maintenance therapy in patients with newly diagnosed glioblastoma. J Clin Oncol 28(16):2712–2718
Sawa H, Murakami H et al (2004) Histone deacetylase inhibitor, FK228, induces apoptosis and suppresses cell proliferation of human glioblastoma cells in vitro and in vivo. Acta Neuropathol 107(6):523–531
Iwamoto FM, Lamborn KR et al (2011) A phase I/II trial of the histone deacetylase inhibitor romidepsin for adults with recurrent malignant glioma: North American brain tumor consortium study 03–03. Neuro-Oncol 13(5):509–516
Berg SL, Stone J et al (2004) Plasma and cerebrospinal fluid pharmacokinetics of depsipeptide (FR901228) in nonhuman primates. Cancer Chemother Pharmacol 54(1):85–88
Hariharan S, Gustafson D et al (2007) Assessment of the biological and pharmacological effects of the ανβ3 and ανβ5 integrin receptor antagonist, cilengitide (EMD 121974), in patients with advanced solid tumors. Ann Oncol 18(8):1400–1407
Shah MH, Binkley P et al (2006) Cardio toxicity of histone deacetylase inhibitor depsipeptide in patients with metastatic neuroendocrine tumors. Clin Cancer Res 12(13):3997–4003
Rini BI, Choueiri TK et al (2008) Sunitinib-induced macrocytosis in patients with metastatic renal cell carcinoma. Cancer 113(6):1309–1314
Brunello A, Saia G et al (2009) Worsening of osteonecrosis of the jaw during treatment with sunitinib in a patient with metastatic renal cell carcinoma. Bone 44(1):173–175
Bergers G, Hanahan D (2008) Modes of resistance to anti-angiogenic therapy. Nat Rev Cancer 8(8):592–603
Lucio-Eterovic AK, Piao Y et al (2009) Mediators of glioblastoma resistance and invasion during antivascular endothelial growth factor therapy. Clin Cancer Res 15(14):4589–4599
de Groot JF, Fuller G et al (2010) Tumor invasion after treatment of glioblastoma with bevacizumab: radiographic and pathologic correlation in humans and mice. Neuro-Oncol 12(3):233–242
Iwamoto FM, Abrey LE et al (2009) Patterns of relapse and prognosis after bevacizumab failure in recurrent glioblastoma. Neurology 73(15):1200–1206
Narayana A, Kunnakkat SD et al (2012) Change in pattern of relapse after antiangiogenic therapy in high-grade glioma. Int J Rad Oncol Biol Phys 82(1):77–82
Wen PY, Macdonald DR et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28(11):1963–1972
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kunnakkat, S., Mathew, M. & Narayana, A. Antiangiogenic therapy in the management of brain tumors: a clinical overview. Cancer Chemother Pharmacol 70, 353–363 (2012). https://doi.org/10.1007/s00280-012-1926-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-012-1926-7