Abstract
Purpose
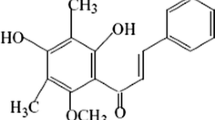
In the following, the cellular and molecular mechanism of cytotoxicity induced by prodrug dacarbazine toward the isolated rat hepatocytes was studied.
Method
Accelerated cytotoxicity screening technique (ACMS) was used to perform this study.
Result
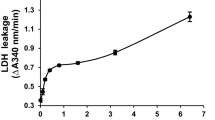
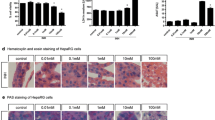
Addition of dacarbazine to isolated rat hepatocytes resulted in reactive oxygen species (ROS) formation, and lysosomal membrane leakiness before hepatocyte lysis occurred. Hepatocyte ROS generation was inhibited by desferoxamine (a ferric chelator). Cytotoxicity was prevented by antioxidants or ROS scavengers (mannitol or dimethylsulfoxide), cytochorome P450 inhibitors (phenylimidazole, diphenyliodonium chloride, 4-methylpyrazole, and benzylimidazole). In addition to lysosomal damage, dacarbazine caused hepatocyte protease activation and cell proteolysis.
Conclusion
Dacarbazine cytotoxicity is associated with ROS (H2O2, O •−2 ) generation. It is suggested that H2O2 could cross the lysosomal membrane, react with lysosomal Fe2+ to form hydroxyl radical (Haber-Weiss reaction) which is the major cause of lysosomal membrane leakiness, proteases, and other digestive enzymes' release and finally the cell death.



Similar content being viewed by others
Abbreviations
- DTIC:
-
5-(3, 3-Dimethyl-1-triazeno)-imidazole-4-carboxamide
- MTIC:
-
5-(3-Monomethyl-1-triazeno)-imidazole-4-carboxamide
- AIC:
-
Aminoimidazole carboxamide
- ROS:
-
Reactive oxygen species
- SD:
-
Standard deviation
- ANOVA:
-
Analysis of variance
- DMSO:
-
Dimethylsulfoxide
- DCF:
-
Dichlorofluorescein
- HEPES:
-
(2-Hydroxyethyl)-1-piperazine-ethansulfonic acid)
- h:
-
Hour
- BHA:
-
Butylated hydroxyanisole
- BHT:
-
Butylated hydroxy toluene
References
Beretta G, Bonadonna G, Bajetta E, Tancini G, De Lena M, Azzarelli A, Veronesi U (1976) Combination chemotherapy with DTIC (NSC-45388) in advanced malignant melanoma, soft tissue sarcomas, and Hodgkin’s disease. Cancer Treat Rep 60(2):205–211
Johnson RO, Metter G, Wilson W, Hill G, Krementz E (1976) Phase I evaluation of DTIC (NSC-45388) and other studies in malignant melanoma in the Central Oncology Group. Cancer Treat Rep 60(2):183–187
Costanzi JJ (1976) DTIC (NSC-45388) studies in the southwest oncology group. Cancer Treat Rep 60(2):189–192
Yamagata S, Ohmori S, Suzuki N, Yoshino M, Hino M, Ishii I, Kitada M (1998) Metabolism of dacarbazine by rat liver microsomes contribution of CYP1A enzymes to dacarbazine N-demethylation. J Pharmacol Exp Ther 26(4):379–382
Abraham DJ (2003) Burger’s medicinal chemistry drug discovery, vol 5, Wiley, New Jersey, pp 1–105
Lev DC, Ruiz M, Mills L, McGary EC, Price JE, Bar-Eli M (2003) Dacarbazine causes transcriptional up-regulation of interleukin 8 and vascular endothelial growth factor in melanoma cells: a possible escape mechanism from chemotherapy. Mol Cancer Ther 2(8):753–763
Saunders PP, Schultz GA (1970) Studies of the mechanism of action of the antitumor agent 5(4)-(3,3-dimethyl-1-triazeno) imidazole-4(5)-carboxamide in Bacillus subtilis. Biochem Pharmacol 19(3):911–919
http://www.rxlist.com/cgi/generic2/dacarbazine_cp.htm. Accessed 26 Nov 2005)
http://www.drugs.com/pdr/DACARBAZINE.html. Accessed 26 Nov 2005)
Skibba JL, Beal DD, Ramirez G, Bryan GT (1970) N-demethylation the antineoplastic agent 4(5)-(3,3-dimethyl-1-triazeno) imidazole-5(4)-carboxamide by rats and man. Cancer Res 30(1):147–150
Skibba JL, Bryan GT (1971) Methylation of nucleic acids and urinary excretion of 14 C-labeled 7-methylguanine by rats and man after administration of 4(5)-(3,3-dimethyl-1-triazeno)-imidazole5(4)-carboxomide. Toxicol Appl Pharmacol 18(3):707–719
Mizuno NS, Humphry EW (1972) Metabolism of 5-(3,3-dimethyl-1-triazeno) imidazole-4-carboxamide (NSC-45388) in human and animal tumor tissue. Cancer Chemother Rep 56(4):465–472
Mizuno NS, Decker RW, Zakis B (1975) Effects of 5-(3-methyl-1-triazeno)imidazole-4-carboxamide (NSC-407347), an alkylating agent derived from 5-(3,3-dimethyl-1-triazeno)imidazole-4-carboxamide (NSC-45388). Biochem Pharmacol 24(5):615–619
Long L, Dolan ME (2001) Role of cytochrome P450 isoenzymes in metabolism of O (6)-benzylguanine: implications for dacarbazine activation. Clin Cancer Res 7(12):4239–4244
http://redpoll.pharmacy.ualberta.ca/drugbank/. Accessed 1 Jan 2006
Farquhar D, Benvenuto J (1984) 1-Aryl-3,3-dimethyltriazenes: potential central nervous system active analogues of 5-(3,3-dimethyl-1-triazeno) imidazole-4-carboxamide (DTIC). J Med Chem 27(12):1723–1727
Catapano CV, Broggini M, Erba E, Ponti M, Mariani L, Citti L, D’ Incalci M (1987) In vitro and in vivo methazolastone-induced DNA damage and repair in L-1210 leukemia sensitive and resistant to chloroethylnitrosoureas. Cancer Res 47(18):4884–4889
Daidone G, Maggio B, Raffa D, Plescia S, Schillaci D, Raimondi MV (2004) Synthesis and in vitro antileukemic activity of new 4-triazenopyrazole derivatives. Farmaco 59(5):413–417
Pourahmad J, O’Brien PJ (2000) A comparison of hepatocyte cytotoxic mechanisms for Cu2+ and Cd2+. Toxicology 143:263–273
Pourahmad J, O’Brien PJ, Jokar F, Daraei B (2003) Carcinogenic metal induced sites of reactive oxygen species formation in hepatocytes. Toxico In Vitro 17:803–810
Shen HM, Shic Y, Sheny A, Ong CN (1996) Detection of elevated reactive oxygen species level in cultured rat hepatocytes treated with aflatoxin B1. Free Radical Biol Med 21:139–146
Pourahmad J, Ross S, O’Brien PJ (2001) Lysosomal involvement in hepatocyte cytotoxicity induced by Cu2+ but not Cd2+. Free Rad Biol Med 30:89–97
Smith MT, Thor H, Hartizell P, Orrenius S (1982) The measurement of lipid peroxidation in isolated hepatocytes. Biochem Pharmacol 31:19–26
Siraki AG, Pourahmad J, Chan TS, Khan S, O’Brien PJ (2002) Endogenous and endobiotic induced reactive oxygen species formation by isolated hepatocytes. Free Rad Biol Med 32:2–10
Hallinan T, Gor J, Rice-Evans CA, Stanley R, O’Reilly R, Brown D (1991) Lipid peroxidation in electroporated hepatocytes occurs much more readily than does hydroxyl-radical formation. Biochem J 277:767–771
Moridani MY, Scobie H, Jamshidzadeh A, Salehi P, O’Brien PJ (2001) Caffeic acid, chlorogenic acid, and dihydrocaffeic acid metabolism: glutathione conjugate formation. Drug Metab Dispos 29(11):1432–1439
Khan S, Sood C, O’Brien PJ (1993) Molecular mechanisms of dibromoalkane cytotoxicity in isolated rat hepatocytes. Biochem Pharmacol 45(2):439–447
Graham RM, Morgan EH, Baker E (1998) Characterisation of citrate and iron citrate uptake by cultured rat hepatocytes. J Hepatol 29:603–613
Luiken JJ, Aerts JM, Meijer AJ (1996) The role of the intralysosomal pH in the control of autophagic proteolytic flux in rat hepatocytes. Eur J Biochem 235:564–573
Brunk UT, Zhang H, Dalen H, Ollinger K (1995) Exposure of cells to nonlethal concentrations of hydrogen peroxide induces degeneration-repair mechanisms involving lysosomal destabilization. Free Rad Biol Med 19:813–822
Seglen PO (1997) DNA ploidy and autophagic protein degradation as determinants of hepatocellular growth and survival. Cell Biol Toxicol 13:301–315
Pourahmad J, Khan S, O’Brien PJ (2001) Lysosomal oxidative stress cytotoxicity induced by nitrofurantoin redox cycling in hepatocytes. Adv Exp Med Biol 500:261–265
Pourahmad J, O’Brien PJ (2001) Biological reactive intermediates that mediate chromium (VI) toxicity. Adv Exp Med Biol 500:203–207
Pourahmad J, Ross S, O’Brien PJ (2001) Lysosomal involvement in hepatocyte cytotoxicity induced by Cu(2+) but not Cd(2+). Free Radic Biol Med 30(1):89–97
Kuhn SH, Gemperli MB, De Beer FC (1985) Effect of two gold compounds on human polymorphonuclear leukocyte lysosomal function and phagocytosis. Inflammation 9:39–44
Olbricht CJ, Fink M, Guljahr E (1991) Alterations in lysosomal enzymes of the proximal tubule in gentamicin nephrotoxicity. Kidney Int 39:639–646
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pourahmad, J., Amirmostofian, M., Kobarfard, F. et al. Biological reactive intermediates that mediate dacarbazine cytotoxicity. Cancer Chemother Pharmacol 65, 89–96 (2009). https://doi.org/10.1007/s00280-009-1007-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-009-1007-8