Abstract
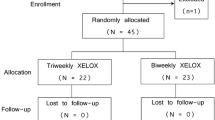
Oxaliplatin 100 mg/m2 iv on day 1, and capecitabine 1,000 mg/m2 orally bid from day 1 (evening) to day 11 (morning) were administered every 2 weeks (OXXEL regimen) to 38 patients as first-line treatment for metastatic colorectal carcinoma. A total of 318 cycles were administered, with a median of 8 (range, 4–12) cycles per patient. Response rate (RR) was 45% (95% confidence interval (CI), 29%–62%), with 7 complete responses and 10 partial responses; furthermore, 12 patients showed a stable disease, so that a disease control was achieved in 29 (76%) patients. RR was greater among patients with performance status 0 (52%), without weight loss (52%), younger than 65 years (50%), and previously unexposed to adjuvant chemotherapy (48%), while no correlation was found with the actually delivered oxaliplatin dose intensity. Overall, haematological side effects were negligible, with no case of grade 4 toxicity, and only one patient suffering from an episode of grade 3 neutropenic fever. Severe anaemia occurred in 4 (11%) patients, and grade 3 neuropathy affected 9 (24%) patients. Median progression-free survival was 7.9 (95% CI, 6.2–9.6) months, and median overall survival has not been reached yet. In conclusion, the OXXEL regimen resulted safe and active, and it deserves further evaluation in metastatic colorectal cancer patients.
Similar content being viewed by others
References
Borner MM, Dietrich D, Stupp R et al (2002) Phase II study of capecitabine and oxaliplatin in first- and second-line treatment of advanced or metastatic colorectal cancer. J Clin Oncol 20:1759–1766
Cassidy J, Tabernero J, Twelves C, et al (2004) XELOX (capecitabine plus oxaliplatin): active first-line therapy for patients with metastatic colorectal cancer. J Clin Onc 22:2084–2091
Comella P, Massidda B, Filippelli G, et al (2004) Oxaliplatin (OXA) plus levo-folinic acid (l-FA) and 5-fluorouacil (FU) i.v. bolus (OXAFAFU) is more active than irinotecan (IRI) plus levo-folinic acid (l-FA) and 5-fluorouacil (FU) i.v. bolus (IRIFAFU) in advanced colorectal carcinoma (ACC): Final results of the Southern Italy Cooperative Oncology Group (SICOG) trial 0103. Ann Oncol 15 (Supplement 3): iii72, abstract# 271PD
Cox DR (1970) The analysis of binary data. Methnem, London
De Gramont A, Cervantes A, André T et al (2004) OPTIMOX study: FOLFOX7/LV5FU2 compared to FOLFOX4 in patients with advanced colorectal cancer. Proc Am Soc Clin Oncol 251s, abstract# 3525
Fisher GA, Kuo T, Cho CD et al (2004) A phase II study of Gefitinib in combination with FOLFOX-4 (IFOX) in patients with metastatic colorectal cancer. Proc Am Soc Clin Oncol 248s, abstract# 3514
Goldberg RM, Sargent DJ, Morton RF et al (2004) A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol 22:23–30
Grothey A, Jordan K, Kellner O et al (2003) Capecitabine plus irinotecan (CAPIRI) vs capecitabine plus oxaliplatin (CAPOX) as first-line therapy of advanced colorectal cancer (ACRC): updated results of a randomized phase II trial. Eur J Cancer S90, abstract# 295
Hoff PM, Ansari R. Batisti G et al (2001) Comparison of oral capecitabine versus intravenous fluorouracil plus leucovorin as first-line treatment in 605 patients with metastatic colorectal cancer: results of a randomized phase III study. J Clin Oncol 19:2282–2292
Kaplan EL, Meier P (1958) Non-parametric estimation from incomplete observations. J Am Stat Assoc 53:457–481
Maindrault-Gœbel F, de Gramont A, Louvet C et al (2000) Evaluation of oxaliplatin dose intensity in bimonthly leucovorin and 48-hour 5-fluorouracil continuous infusion regimens (FOLFOX) in pretreated metastatic colorectal cancer. Oncology Multidisciplinary Research Group (GERCOR). Ann Oncol 11:1477–1483
Maindrault-Gœbel F, de Gramont A, Louvet C et al (2001) High-dose intensity oxaliplatin added to the simplified bimonthly leucovorin and 5-fluorouracil regimen as second-line therapy for metastatic colorectal cancer (FOLFOX 7). Eur J Cancer 37:1000–1005
Miller AB, Hoogstraten B, Staquet M, Winkler V (1981) Reporting results of cancer treatment. Cancer 47:207–214
Scheithauer WS, Kornek GV, Raderer M, et al (2003) Randomized multicenter phase II trial of two different schedules of capecitabine plus oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 21:1307–1312
Simon R (1989) Optimal two stage design for phase II clinical trials. Contr Clin Trial 10:1–10
Tabernero JM, Van Cutsem E, Sastre J et al (2004) An international phase II study of cetuximab in combination with oxaliplatin/5-fluorouracil (5-FU)/folinic acid (FA) (FOLOFOX-4) in the first-line treatment of patients with metastatic colorectal cancer (CRC) expressing Epidermal Growth Factor Receptor (EGFR): Preliminary results. Proc Am Soc Clin Oncol 248s, abstract# 3512
Twelves C, on behalf of the Xeloda Colorectal Cancer Group (2002) Capecitabine as first-line treatment in colorectal cancer: pooled data from two large, phase III trials. Eur J Cancer 38:S15–S20
Van Cutsem E, Twelves C, Cassidy J et al (2001) Oral capecitabine compared with intravenous 5-fluorouracil plus leucovorin (Mayo Clinic regimen) in patients with metastatic colorectal cancer: results of a large phase III study. J Clin Oncol 19:4097–4106
Zeuli M, Nardoni C, Pino MS et al (2003) Phase II study of capecitabine and oxaliplatin as first-line treatment in advanced colorectal cancer. Ann Oncol 14:1378–1382
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Comella, P., Massidda, B., Palmeri, S. et al. Biweekly oxaliplatin combined with oral capecitabine (OXXEL regimen) as first-line treatment of metastatic colorectal cancer patients: a Southern Italy Cooperative Oncology Group phase II study. Cancer Chemother Pharmacol 56, 481–486 (2005). https://doi.org/10.1007/s00280-005-1003-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-005-1003-6