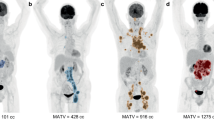
Abstract
Chimeric antigen receptor T-cell therapy (CART) can be administered outpatient yet requires management of potential side effects such as cytokine release syndrome (CRS) and immune effector cell-associated neurotoxicity syndrome (ICANS). The pre-infusion tumor burden is associated with CRS, yet there is no data on the relevance of pre-infusion tumor growth rate (TGR). Our objective was to investigate TGR for the occurrence and severity of CRS and ICANS. Consecutive patients with available pre-baseline and baseline (BL) imaging before CART were included. TGR was determined as both absolute (abs) and percentage change (%) of Lugano criteria-based tumor burden in relation to days between exams. CRS and ICANS were graded according to ASTCT consensus criteria. Clinical metadata was collected including the international prognostic index (IPI), patient age, ECOG performance status, and LDH. Sixty-two patients were included (median age: 62 years, 40% female). The median pre-BL TGR [abs] and pre-BL TGR [%] was 7.5 mm2/d and 30.9%/d. Pre-BL TGR [abs] and pre-BL TGR [%] displayed a very weak positive correlation with the grade of CRS (r[abs] = 0.14 and r[%] = 0.13) and no correlation with ICANS (r[abs] = − 0.06 and r[%] = − 0.07). There was a weak positive correlation between grade of CRS and grade of ICANS (r = 0.35; p = 0.005) whereas there was no significant correlation of CRS or ICANS to any other of the examined parameters. The pre-infusion TGR before CART was weakly associated with the occurrence of CRS, but not the severity, whereas there were no significant differences in the prediction of ICANS. There was no added information when compared to pre-infusion tumor burden alone. Outpatient planning and toxicity management should not be influenced by the pre-infusion TGR.






Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- BL :
-
Baseline
- CART :
-
Chimeric antigen receptor T-cell therapy
- CRS :
-
Cytokine release syndrome
- ECOG :
-
Eastern Cooperative Oncology Group
- FDG :
-
Fluorodeoxyglucose
- FL :
-
Follicular lymphoma
- ICANS :
-
Immune effector cell-associated neurotoxicity syndrome
- IPI :
-
International prognostic index
- IQR :
-
Interquartile range
- LBCL :
-
Large B-cell lymphoma
- LDH :
-
Lactate dehydrogenase
- MCL :
-
Mantle-cell lymphoma
- OS :
-
Overall survival
- PET/CT :
-
Positron emission tomography-computed tomography
- PFS :
-
Progression-free survival
- r/r :
-
Relapsed or refractory
- SPD :
-
Sum of the product diameters
- TB :
-
Tumor burden
- TGR :
-
Tumor growth rate
- TL :
-
Target lesion
References
June CH, Sadelain M (2018) Chimeric antigen receptor therapy. N Engl J Med 379:64–73. https://doi.org/10.1056/NEJMra1706169
Schuster SJ, Bishop MR, Tam CS, Waller EK, Borchmann P, McGuirk JP, Jager U, Jaglowski S, Andreadis C, Westin JR et al (2019) Tisagenlecleucel in adult relapsed or refractory diffuse large B-cell lymphoma. N Engl J Med 380:45–56. https://doi.org/10.1056/NEJMoa1804980
Schuster SJ, Svoboda J, Chong EA, Nasta SD, Mato AR, Anak O, Brogdon JL, Pruteanu-Malinici I, Bhoj V, Landsburg D et al (2017) Chimeric antigen receptor T cells in refractory B-cell lymphomas. N Engl J Med 377:2545–2554. https://doi.org/10.1056/NEJMoa1708566
Neelapu SS, Locke FL, Bartlett NL, Lekakis LJ, Miklos DB, Jacobson CA, Braunschweig I, Oluwole OO, Siddiqi T, Lin Y et al (2017) Axicabtagene ciloleucel CAR T-cell therapy in refractory large B-cell lymphoma. N Engl J Med 377:2531–2544. https://doi.org/10.1056/NEJMoa1707447
Wang M, Munoz J, Goy A, Locke FL, Jacobson CA, Hill BT, Timmerman JM, Holmes H, Jaglowski S, Flinn IW et al (2020) KTE-X19 CAR T-cell therapy in relapsed or refractory mantle-cell lymphoma. N Engl J Med 382:1331–1342. https://doi.org/10.1056/NEJMoa1914347
Shimabukuro-Vornhagen A, Gödel P, Subklewe M, Stemmler HJ, Schlößer HA, Schlaak M, Kochanek M, Böll B, von Bergwelt-Baildon MS (2018) Cytokine release syndrome. J Immunother Cancer 6:56. https://doi.org/10.1186/s40425-018-0343-9
Hay KA, Hanafi LA, Li D, Gust J, Liles WC, Wurfel MM, Lopez JA, Chen J, Chung D, Harju-Baker S et al (2017) Kinetics and biomarkers of severe cytokine release syndrome after CD19 chimeric antigen receptor-modified T-cell therapy. Blood 130:2295–2306. https://doi.org/10.1182/blood-2017-06-793141
Lee DW, Santomasso BD, Locke FL, Ghobadi A, Turtle CJ, Brudno JN, Maus MV, Park JH, Mead E, Pavletic S et al (2019) ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol Blood Marrow Transplant 25:625–638. https://doi.org/10.1016/j.bbmt.2018.12.758
Greenbaum U, Strati P, Saliba RM, Torres J, Rondon G, Nieto Y, Hosing C, Srour SA, Westin J, Fayad LE et al (2021) CRP and ferritin in addition to the EASIX score predict CAR-T-related toxicity. Blood Adv 5:2799–2806. https://doi.org/10.1182/bloodadvances.2021004575
Pennisi M, Sanchez-Escamilla M, Flynn JR, Shouval R, Alarcon Tomas A, Silverberg ML, Batlevi C, Brentjens RJ, Dahi PB, Devlin SM et al (2021) Modified EASIX predicts severe cytokine release syndrome and neurotoxicity after chimeric antigen receptor T cells. Blood Adv 5:3397–3406. https://doi.org/10.1182/bloodadvances.2020003885
Pasquini MC, Hu ZH, Curran K, Laetsch T, Locke F, Rouce R, Pulsipher MA, Phillips CL, Keating A, Frigault MJ et al (2020) Real-world evidence of tisagenlecleucel for pediatric acute lymphoblastic leukemia and non-Hodgkin lymphoma. Blood Adv 4:5414–5424. https://doi.org/10.1182/bloodadvances.2020003092
Karschnia P, Jordan JT, Forst DA, Arrillaga-Romany IC, Batchelor TT, Baehring JM, Clement NF, Gonzalez Castro LN, Herlopian A, Maus MV et al (2019) Clinical presentation, management, and biomarkers of neurotoxicity after adoptive immunotherapy with CAR T cells. Blood 133:2212–2221. https://doi.org/10.1182/blood-2018-12-893396
Santomasso BD, Park JH, Salloum D, Riviere I, Flynn J, Mead E, Halton E, Wang X, Senechal B, Purdon T et al (2018) Clinical and biological correlates of neurotoxicity associated with CAR T-cell therapy in patients with B-cell acute lymphoblastic leukemia. Cancer Discov 8:958–971. https://doi.org/10.1158/2159-8290.CD-17-1319
Morris EC, Neelapu SS, Giavridis T, Sadelain M (2022) Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol 22:85–96. https://doi.org/10.1038/s41577-021-00547-6
Bansal R, Paludo J, Hathcock MA, Spychalla MT, Khurana A, Hampel PJ, Durani U, Alkhateeb HB, Dingli D, Hayman SR et al (2022) Outpatient practice pattern for recently approved CAR-T in patients with lymphoma and multiple myeloma. Blood 140:2399–2401. https://doi.org/10.1182/blood-2022-167187
Borogovac A, Keruakous A, Bycko M, HolterChakrabarty J, Ibrahimi S, Khawandanah M, Selby GB, Yuen C, Schmidt S, Autry MT et al (2022) Safety and feasibility of outpatient chimeric antigen receptor (CAR) T-cell therapy: experience from a tertiary care center. Bone Marrow Transplant 57:1025–1027. https://doi.org/10.1038/s41409-022-01664-z
Meignan M, Cottereau AS, Specht L, Mikhaeel NG (2021) Total tumor burden in lymphoma - an evolving strong prognostic parameter. Br J Radiol 94:20210448. https://doi.org/10.1259/bjr.20210448
Gobbi PG (2014) Tumor burden in Hodgkin’s lymphoma: much more than the best prognostic factor. Crit Rev Oncol Hematol 90:17–23. https://doi.org/10.1016/j.critrevonc.2013.11.002
Gomez-Roca C, Koscielny S, Ribrag V, Dromain C, Marzouk I, Bidault F, Bahleda R, Ferte C, Massard C, Soria JC (2011) Tumour growth rates and RECIST criteria in early drug development. Eur J Cancer 47:2512–2516. https://doi.org/10.1016/j.ejca.2011.06.012
Ferte C, Fernandez M, Hollebecque A, Koscielny S, Levy A, Massard C, Balheda R, Bot B, Gomez-Roca C, Dromain C et al (2014) Tumor growth rate is an early indicator of antitumor drug activity in phase I clinical trials. Clin Cancer Res : An Off J Am Assoc Cancer Res 20:246–252. https://doi.org/10.1158/1078-0432.CCR-13-2098
Winkelmann M, Blumenberg V, Rejeski K, Quell C, Bucklein VL, Ingenerf M, Unterrainer M, Schmidt C, Dekorsy FJ, Bartenstein P et al (2023) Prognostic value of pre-infusion tumor growth rate for patients with lymphoma receiving chimeric antigen receptor T-cell therapy. Cytotherapy. https://doi.org/10.1016/j.jcyt.2023.03.007
Cheson BD, Fisher RI, Barrington SF, Cavalli F, Schwartz LH, Zucca E, Lister TA, Alliance AL, Lymphoma G, Eastern Cooperative Oncology G et al (2014) Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol 32:3059–3068. https://doi.org/10.1200/JCO.2013.54.8800
International Non-Hodgkin’s Lymphoma Prognostic Factors P (1993) A predictive model for aggressive non-Hodgkin’s lymphoma. N Engl J Med 329:987–994. https://doi.org/10.1056/NEJM199309303291402
Dos Santos DMC, Rejeski K, Winkelmann M, Liu L, Trinkner P, Gunther S, Bucklein VL, Blumenberg V, Schmidt C, Kunz WG et al (2022) Increased visceral fat distribution and body composition impact cytokine release syndrome onset and severity after CD19 CAR-T in advanced B-cell malignancies. Haematologica. https://doi.org/10.3324/haematol.2021.280189
Vercellino L, Di Blasi R, Kanoun S, Tessoulin B, Rossi C, D’Aveni-Piney M, Oberic L, Bodet-Milin C, Bories P, Olivier P et al (2020) Predictive factors of early progression after CAR T-cell therapy in relapsed/refractory diffuse large B-cell lymphoma. Blood Adv 4:5607–5615. https://doi.org/10.1182/bloodadvances.2020003001
Wang J, Hu Y, Yang S, Wei G, Zhao X, Wu W, Zhang Y, Zhang Y, Chen D, Wu Z et al (2019) Role of fluorodeoxyglucose positron emission tomography/computed tomography in predicting the adverse effects of chimeric antigen receptor T cell therapy in patients with non-Hodgkin lymphoma. Biol Blood Marrow Transplant 25:1092–1098. https://doi.org/10.1016/j.bbmt.2019.02.008
Dean EA, Mhaskar RS, Lu H, Mousa MS, Krivenko GS, Lazaryan A, Bachmeier CA, Chavez JC, Nishihori T, Davila ML et al (2020) High metabolic tumor volume is associated with decreased efficacy of axicabtagene ciloleucel in large B-cell lymphoma. Blood Adv 4:3268–3276. https://doi.org/10.1182/bloodadvances.2020001900
Iacoboni G, Simo M, Villacampa G, Catala E, Carpio C, Diaz-Lagares C, Vidal-Jordana A, Bobillo S, Marin-Niebla A, Perez A et al (2021) Prognostic impact of total metabolic tumor volume in large B-cell lymphoma patients receiving CAR T-cell therapy. Ann Hematol 100:2303–2310. https://doi.org/10.1007/s00277-021-04560-6
Titov A, Petukhov A, Staliarova A, Motorin D, Bulatov E, Shuvalov O, Soond SM, Piacentini M, Melino G, Zaritskey A et al (2018) The biological basis and clinical symptoms of CAR-T therapy-associated toxicites. Cell Death Dis 9:897. https://doi.org/10.1038/s41419-018-0918-x
Champiat S, Ferrara R, Massard C, Besse B, Marabelle A, Soria JC, Ferte C (2018) Hyperprogressive disease: recognizing a novel pattern to improve patient management. Nat Rev Clin Oncol 15:748–762. https://doi.org/10.1038/s41571-018-0111-2
Champiat S, Dercle L, Ammari S, Massard C, Hollebecque A, Postel-Vinay S, Chaput N, Eggermont A, Marabelle A, Soria JC et al (2017) Hyperprogressive disease is a new pattern of progression in cancer patients treated by anti-PD-1/PD-L1. Clin Cancer Res : An Off J Am Assoc Cancer Res 23:1920–1928. https://doi.org/10.1158/1078-0432.CCR-16-1741
Wagner NB, Lenders MM, Kuhl K, Reinhardt L, Andre F, Dudda M, Ring N, Ebel C, Stager R, Zellweger C et al (2021) Pretreatment metastatic growth rate determines clinical outcome of advanced melanoma patients treated with anti-PD-1 antibodies: a multicenter cohort study. J Immunother Cancer 9(5):e002350. https://doi.org/10.1136/jitc-2021-002350
Rejeski K, Perez A, Iacoboni G, Penack O, Bucklein V, Jentzsch L, Mougiakakos D, Johnson G, Arciola B, Carpio C et al (2022) The CAR-HEMATOTOX risk-stratifies patients for severe infections and disease progression after CD19 CAR-T in R/R LBCL. J Immunother Cancer 10(5):e004475. https://doi.org/10.1136/jitc-2021-004475
Rejeski K, Perez A, Sesques P, Hoster E, Berger C, Jentzsch L, Mougiakakos D, Frolich L, Ackermann J, Bucklein V et al (2021) CAR-HEMATOTOX: a model for CAR T-cell-related hematologic toxicity in relapsed/refractory large B-cell lymphoma. Blood 138:2499–2513. https://doi.org/10.1182/blood.2020010543
Funding
The work was supported by funding from the research program “Förderung für Forschung und Lehre (FöFoLe) project number 1147” of the Medical Faculty of Ludwig Maximilian University (LMU) Munich and the Bavarian Cancer Research Center (BZKF) to M.W. The work was further supported by the Else-Kröner-Fresenius Stiftung (to V.B.) and the German Cancer Consortium DKTK (to V.B.).
Author information
Authors and Affiliations
Contributions
M.W. and W.G.K. conceived and designed the study; V.B., K.R., C.Q., V.L.B., M.I., M.U., and C.S. collected the data; M.W., V.B., K.R., C.Q., V.L.B., and W.G.K. analyzed and interpreted the data; M.W. and W.G.K. drafted the manuscript; and all authors revised the manuscript.
Corresponding author
Ethics declarations
Ethics approval
All medical records and imaging studies were reviewed with the approval of the LMU Munich Institutional Review Board (LMU Ethics Committee, project number 19–817).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
V.B.: BMS/Celgene: research funding; Kite/Gilead: consultancy, honoraria, research funding; Janssen: research funding, honoraria; Novartis: research funding, honoraria; Roche: consultancy, research funding; Takeda: research funding. K.R.: Kite/Gilead: research funding; Kite/Gilead: travel support; Novartis: honoraria. V.L.B.: Amgen: honoraria; Celgene/BMS: research funding; Kite/Gilead: research funding, honoraria; Novartis: honoraria; Pfizer: honoraria. C.S.: Kite/Gilead: travel support. M.v.B.: Astellas: consultancy, research funding, and honoraria; BMS: consultancy, research funding, and honoraria; Kite/Gilead: consultancy, research funding, and honoraria; Miltenyi: consultancy, research funding, and honoraria; Mologen: consultancy, research funding, and honoraria; MSD Sharp & Dohme: consultancy, research funding, and honoraria; Novartis: consultancy, research funding, and honoraria; Roche: consultancy, research funding, and honoraria. M.S.: Amgen: research funding, speakers bureau; Astra Zeneca: speakers bureau; Aven Cell: consultancy; BMS/Celgene: research funding, speakers bureau; CDR-Life: consultancy; Gilead: research funding, speakers bureau; GSK: speakers bureau; Ichnos Sciences: consultancy; Incyte Biosciences: consultancy; Janssen: research funding, consultancy, speakers bureau; Miltenyi Biotec: research funding, consultancy; Morphosys: research funding; Molecular Partners: consultancy; Novartis: research funding, consultancy, speakers bureau; Pfizer: consultancy, speakers bureau; Roche: research funding, speakers bureau; Seattle Genetics: research funding; Takeda: research funding, consultancy, speakers bureau. W.G.K.: Bristol Myers Squibb: advisor; mint Medical: advisor; Need, Inc: advisor; Boehringer Ingelheim, advisor. The remaining authors declare no competing financial interests. None of the mentioned conflicts of interest were related to financing of the content of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Winkelmann, M., Blumenberg, V., Rejeski, K. et al. Predictive value of pre-infusion tumor growth rate for the occurrence and severity of CRS and ICANS in lymphoma under CAR T-cell therapy. Ann Hematol 103, 259–268 (2024). https://doi.org/10.1007/s00277-023-05507-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-023-05507-9