Abstract
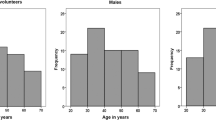
T-large granular lymphocytes (T-LGL) characterized by dim CD5 staining, although not completely understood, have unique roles in the immune system. Expansion of peripheral blood (PB) clonal T-LGL populations is associated with various entities in adults. We have previously demonstrated clonal T-LGL proliferations in pediatric immune dysregulation/inflammatory/proliferative conditions. However, T-LGL populations have not been studied in broader spectrum pathologies. In this study we evaluated sizes and correlates of T-LGL populations in the pediatric and young adult populations with various disease states. Lymphocytes including T-LGL were investigated retrospectively by reviewing PB multiparameter flow cytometric data with various indications over a 4-year period. Associations with clinical, laboratory findings, and T-LGL population sizes were sought. Among 520 cases reviewed, 240 were females and 280 males with a mean age of 9 years (0–33 years); mean T-LGL population constituted 14% (1–67%) in PB T cells. There were significant differences between T-LGL and CD5-bright, regular T cells. T-LGL correlated with CD8 + /DR + (R = 0.570; P < 0.01) and CD8 + /CD11b + (R = 0.597; P < 0.01) expression, indicating activated cytotoxic phenotype. The highest average T-LGL were seen in bone marrow transplant recipients (23.7%), Evans syndrome (23.7%), lymphoma (20.6%), and acute EBV infection (20.4%) cases, all with underlying immune dysregulation pathologies. In pediatric and young adult patients with different clinical conditions, PB T-LGL constitute an average of 14% of the T cells and have a predominantly activated cytotoxic T cell phenotype. Higher relative presence was seen in cases with an immune dysregulation background. These results may serve as a reference for T-LGL research efforts.


Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Lamy T, Moignet A, Loughran TP Jr (2017) LGL leukemia: from pathogenesis to treatment. Blood 129(9):1082–1094
Gorczyca W (2022) Flow cytometry in neoplastic hematology: morphologic-immunophenotypic-genetic correlation. 4th ed. CRC Press Taylor & Francis Group. p. 75
Fattizzo B, Bellani V, Pasquale R, Giannotta JA, Barcellini W (2021) Large granular lymphocyte expansion in myeloid diseases and bone marrow failure syndromes: whoever seeks finds. Front Oncol 11:748610
Semenzato G, Teramo A, Calabretto G, Gasparini VR, Zambello R (2022) All that glitters is not LGL Leukemia. Leukemia 36(11):2551–2557
Lamy T, Loughran TP Jr (2003) Clinical features of large granular lymphocyte leukemia. Semin Hematol 40(3):185–195
Sun H, Wei S, Yang L (2019) Dysfunction of immune system in the development of large granular lymphocyte leukemia. Hematology 24(1):139–147
Moignet A, Lamy T (2018) Latest advances in the diagnosis and treatment of large granular lymphocytic leukemia. Am Soc Clin Oncol Educ Book 38:616–625
Savaşan S, Al-Qanber B, Buck S, Wakeling E, Gadgeel M (2020) Clonal T-cell large granular lymphocyte proliferations in childhood and young adult immune dysregulation conditions. Pediatr Blood Cancer 67(5):e28231
Savaşan S, Wakeling E, Knight T, Buck S, Gadgeel M (2019) Different clonal t-large granular lymphocyte proliferations in SCID. J Clin Immunol 39(3):245–248
Heuser M, Thol F, Ganser A (2016) Clonal hematopoiesis of indeterminate potential. Dtsch Arztebl Int 113(18):317–322
Rajpurkar M, Buck S, Lafferty J, Wakeling E, Ravindranath Y, Savaşan S (2019) Acquired pure red cell aplasia and acquired amegakaryocytic thrombocytopenia associated with clonal expansion of T-cell large granular lymphocytes in a patient with lipopolysaccharide-responsive beige-like anchor (LRBA) protein deficiency. J Pediatr Hematol Oncol 41(8):e542–e545
Kondoh K, Morimoto M, Keino D et al (2013) T-cell large granular lymphocyte leukemia in a child with anemia and Crohn’s disease. Pediatr Int 55(1):111–114
Kitchen BJ, Boxer LA (2008) Large granular lymphocyte leukemia (LGL) in a child with hyper IgM syndrome and autoimmune hemolytic anemia. Pediatr Blood Cancer 50(1):142–145
Blanchong CA, Olshefski R, Kahwash S (2001) Large granular lymphocyte leukemia: case report of chronic neutropenia and rheumatoid arthritis-like symptoms in a child. Pediatr Dev Pathol 4(1):94–99
Ketterl TG, Wu D, Fromm JR et al (2018) Donor derived T-cell large granular lymphocyte leukemia after cord blood transplant for pediatric T-cell lymphoblastic leukemia. Bone Marrow Transplant 53(3):352–355
Boeckx N, Uyttebroeck A, Langerak AW, Brusselmans C, Goossens W, Bossuyt X (2004) Clonal proliferation of T-Cell large granular lymphocytes. Pediatr Blood Cancer 42(3):275–277
Nann-Rütti S, Tzankov A, Cantoni N et al (2012) Large granular lymphocyte expansion after allogeneic hematopoietic stem cell transplant is associated with a cytomegalovirus reactivation and shows an indolent outcome. Biol Blood Marrow Transplant 18(11):1765–1770
Kim D, Al-Dawsari G, Chang H et al (2013) Large granular lymphocytosis and its impact on long-term clinical outcomes following allo-SCT. Bone Marrow Transplant 48(8):1104–1111
Dolstra H, Preijers F, Van de Wiel-van KE, Schattenberg A, Galama J, de Witte T (1995) Expansion of CD8+CD57+ T cells after allogeneic BMT is related with a low incidence of relapse and with cytomegalovirus infection. Br J Haematol 90(2):300–307
Jaime-Pérez JC, Aguilar-Calderón PE, Salazar-Cavazos L, Gómez-Almaguer D (2018) Evans syndrome: clinical perspectives, biological insights and treatment modalities. J Blood Med 9:171–184
Viny AD, Lichtin A, Pohlman B, Loughran T, Maciejewski J (2008) Chronic B-cell dyscrasias are an important clinical feature of T-LGL leukemia. Leuk Lymphoma 49(5):932–938
Goyal T, Thakral B, Wang SA et al (2018) T-cell large granular lymphocytic leukemia and coexisting b-cell lymphomas: a study from the bone marrow pathology group. Am J Clin Pathol 149(2):164–171
Yang J, Epling-Burnette PK, Painter JS et al (2008) Antigen activation and impaired Fas-induced death-inducing signaling complex formation in T-large-granular lymphocyte leukemia. Blood 111(3):1610–1616
Wang RC, Chang ST, Hsieh YC et al (2014) Spectrum of Epstein-Barr virus-associated T-cell lymphoproliferative disorder in adolescents and young adults in Taiwan. Int J Clin Exp Pathol 7(5):2430–2437
Yang R, Ai Y, Liu C, Lu X (2022) Aggressive Natural Killer Cell Leukemia in an Adolescent Patient: A Case Report and Literature Review. Front Pediatr 10:829927
Song YL, Wang BF, Jiang NG, Jin YM, Zeng TT (2020) CD8dimCD3+ lymphocytes in fever patients might be biomarkers of active EBV infection and exclusion indicator of T-LGLL. Biomark Med 14(18):1703–1715
Callan MF, Steven N, Krausa P et al (1996) Large clonal expansions of CD8+ T cells in acute infectious mononucleosis. Nat Med 2(8):906–911
Funding
This study is supported partially by funds from Children’s Foundation and Kids Without Cancer Foundation.
Author information
Authors and Affiliations
Contributions
M.G. performed flow cytometry data acquisition and wrote the manuscript; I.A. collected flow cytometry and clinical data and performed statistical analysis; B.A. collected flow cytometry and clinical data; S.B. performed flow cytometry data acquisition; S.S. planned the study, analyzed the results, revised the manuscript, and approved the final version.
Corresponding author
Ethics declarations
Ethics approval
This research study was conducted retrospectively from data obtained for clinical purposes. We consulted extensively with the IRB of Wayne State University who determined that our study did not need ethical approval. An IRB official waiver of ethical approval was granted from the IRB of Wayne State University.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Accepted in abstract form titled “T-large granular lymphocyte frequencies and correlates in disease states detected by flowcytometry” for the American Society of Pediatric Hematology/Oncology (ASPHO) May 2023 annual meeting (Poster #305) and published in the Pediatric Blood and Cancer in June 2023 issue (Volume 70, Supplement 3).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gadgeel, M., Al Kooheji, I., Al-Qanber, B. et al. T-large granular lymphocyte frequencies and correlates in disease states detected by multiparameter flow cytometry in pediatric and young adult population. Ann Hematol 103, 133–140 (2024). https://doi.org/10.1007/s00277-023-05449-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-023-05449-2