Abstract
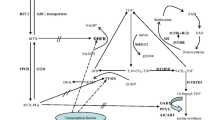
Methylenetetrahydrofolate reductase (MTHFR) is one of the most critical enzyme in folic acid metabolism, and it converts 5,10-MTHF to 5-MTHF. 5,10-MTHF is required for conversion of uridilate to thymidylate. On the other side, MTHFR enzyme causes methylation of homocysteine into methionine, leading to methylation of DNA. Chemotherapeutic agents have different effects, but DNA is the target for most of them. Because folate is the cornerstone in DNA synthesis, we analysed herein if the polymorphisms in MTHFR gene can alter the susceptibility of lymphoproliferative disease risk and if it has an effect on chemotherapy response. One hundred fifty-six patients with lymphoid malignancies and 82 healthy controls were included into the study. Neither gene frequencies nor allel frequencies were found to increase lymphoproliferative disease risk significantly in both overall group and subgroups. Although it was not statistically significant, we found a 2.7-fold increased risk in acute lymphocytic leukaemia (ALL)/Burkitt lymphoma patients with TT genotype [odds ratio (OR), 2.7; 95% confidence interval (CI), 0.88–8.2] than CC genotype but a 1.7-fold decreased risk with TT genotype in diffuse large B-cell lymphoma (DLBCL; OR, 0.58; 95% CI, 0.17–1.88) and a 1.8-fold decreased risk in Hodgkin’s lymphoma with TT genotype (OR, 0.55; 95% CI, 0.10–2.87) than CC genotype. The chemotherapy response was analysed in DLBCL, Hodgkin’s lymphoma and ALL/Burkitt’s lymphoma because these patients received standard chemotherapy protocols. No significant difference was detected between responder and non-responders according to MTHFR T677C polymorphism, but the patients who had TT genotype respond 1.75-fold worse than CC (OR, 0.57; 95% CI, 0.07–4.64) in ALL patients (p=0.59), and in DLBCL, CT genotype revealed a 1.8-fold worse response than CC genotype (OR, 0.54; 95% CI, 0.17–1.7), but TT genotype revealed 2.6-fold better response rates than patients with CC genotype (OR, 2.6; 95% CI, 0.26–26.8). As a conclusion, MTHFR C677T polymorphism does not increase the risk of lymphoproliferative disease, and it does not have an effect on chemotherapy response significantly; however, the patients with TT genotype have a slightly increased risk for ALL, and they also respond worse than CC genotype. TT genotype slightly decreases the risk of DLBCL, and the patients have much favorable response rates.
Similar content being viewed by others
References
Ames BN (2001) DNA damage from micronutrient deficiencies is likely to be a major cause of cancer. Mutat Res 475:7–20
Aplenc R, Thompson J, Han P, La M, Zhao H, Lange B, Rebbeck T (2005) Methylenetetrahydofolate reductase polymorphism and therapy response in pediatric acute lymphoblast leukemia. Cancer Res 65:2482–2487
Aslanidis C, Schmitz G (2001) High speed methylenetetrahydrofolate reductase C→T 677 mutation detection on Light Cycler. In: Meuer S, Wittwer C, Nakagawara K (eds) Rapid cycle real-time PCR methods and applications. Springer, Berlin Heidelberg New York, pp 83–89
Blount BC, Mack MM, Wehr CM, MacGregor JT, Hiatt RA, Wang G, Wickramasinghe SH, Everson RB, Ames BN (1997) Folate deficiency causes uracil misincorporation into human DNA and chromosome breakage: implication for cancer and neuronal damage. Proc Natl Acad Sci USA 94:3290–3295
Chen J, Giovannucci EL, Hunter DJ (1999) MTHFR polymorphism, methyl-replete diets and the risk of colorectal carcinoma and adenoma among US men and women: an example of gene environment interactions in colorectal tumorigenesis. J Nutr 129:560–564
Cheson BD, Horning SJ, Coiffier B, Shipp MA, Fisher RI, Connors JM, Lister TA, Vose J, Grillo-Lopez A, Hagenbeek A, Cabanillas F, Klippensten D, Hiddemann W, Castellino R, Harris NL, Armitage JO, Carter W, Hoppe R, Canellos GP (1999) Report of an international workshop to standardize response criteria for non-Hodgkin’s lymphomas. NCI Sponsored International Working Group. J Clin Oncol 18:1244
Cheson BD, Bennet JM, Kopecky KJ, Büchner T, Willman CL, Estey EH, Schiffer CA, Doehner H, Tallman MS, Lister TA, Lo-Coco F, Willemze R, Biondi A, Hiddemann W, Larson RA, Löwenberg B, Sanz MA, Head DR, Ohno R, Bloomfield CD (2003) Revised recommendations of the international working group for diagnosis, standardization of response criteria, treatment outcomes, and reporting standards for therapeutic trials in acute myeloid leukemia. J Clin Oncol 21:4642–4649
De Jonge R, Hooijberg JH, van Zelst BD, Jansen G, vanZantwijk CH, Kaspers GJL, Peters FGJ, Ravindranath Y, Pieters R, Lindemans J (2005) Effect of polymorphisms in folate-related genes on in vitro methotrexate sensitivity in pediatric acute lymphoblastic leukemia. Blood 106:717–720
Duthie SJ (1999) Folic acid deficiency and cancer: mechanisms of DNA instability. Br Med Bull 55:578–592
Ergül E, Sazcı A, Utkan Z, Cantürk NZ (2003) Polymorphisms in the MTHFR gene are associated with breast cancer. Tumor Biol 24:286–290
Esteller M, Garcia A, Martinez-Palones JM, Xercavins J, Reventos J (1997) Germ line polymorphisms in cytochrome-P450 1A1 (C4887 CYP1A1) and methylenetetrahydrofolate reductase (MTHFR) genes and endometrial cancer susceptibility. Carcinogenesis 18:2307–2311
Friso S, Choi SW, Girelli D, Mason JB, Dolnikowski GG, Bagley PJ, Olivieri O, Jacques PF, Rosenberg IH, Correcher R, Selhub J (2002) A common mutation in the 5,10-methylenetetrahydrofolate reductase gene affects genomic DNA methylation through an interaction with folate status. Proc Natl Acad Sci USA 99:5606–5611
Frosst P, Blom HJ, Milos R, Goyett P, Sheppard CA, Matthews RG, Boers GJ, den Heijer M, Kluijtmans LA, van den Heuve LP, Rozen R (1995) A candidate genetic risk factor for vascular disease. A common mutation in methylenetetrahydrofolate reductase. Nat Genet 10:111–113
Gemmati D, Ongaro A, Scapoli GL, Della Porta M, Tognazzo S, Serino ML, Di Bona E, Rodeghiero F, Gilli G, Reverberi R, Caruso A, Pasello M, Pellati A, De Mattei M (2004) Common gene polymorphisms in the metabolic folate and methylation pathway and the risk of acute lymphoblastic leukemia and non-Hodgkin’s lymphoma in adults. Cancer Epidemiol Biomarkers Prev 13:787–794
Goyette P, Sumner JS, Milos R, Duncan AM, Rosenblatt DS, Matthews RG, Rozen R (1994) Human methylenetetrahydrofolate reductase: isolation of cDNA mapping and mutation identification. Nat Genet 7:195–200
Hande KR (2004) Principles and pharmacology of chemotherapy. In: Greer JP, Foerster J, Lukens JN, Rodgers GM, Paraskevas F, Glader B (eds) Wintrobe’s clinical hematology, vol 2. Lippincott Williams &Wilkins, Philadelphia, pp 1945–1969
Jolivet J, Cowan KH, Curt GA (1983) The pharmacology and clinical use of methotrexate. N Engl J Med 309:1094–1104
Kim YI (2004) Folate and DNA methylation: a mechanistic link between folate deficiency and colorectal cancer? Cancer Epidemiol Biomarkers Prev 13:511–519
Kım YI (2004) Folate and carcinogenesis: evidence, mechanisms, and implications. J Nutr Biochem 10:66–68
Krajinovic M, Lamothe S, Labuda D, Lemieux-Blanchard E, Theoret Y, Moghrabi A, Sinnet D (2004) Role of MTHFR genetic polymorphisms in the susceptibility to childhood acute lymphoblastic leukemia. Blood 103:252–257
Lincz LF, Scorgie FE, Kerridge I, Potts R, Spencer A, Enno A (2003) Methionine synthase genetic polymorphism MS A2756G alters susceptibility to follicular but not diffuse large B-cell non-Hodgkin’s lymphoma or multiple myeloma. Br J Haematol 120:1051–1054
Matsuo K, Suzuki R, Hamajima N, Ogura M, Kagami Y, Taji H, Kondoh E, Maeda S, Asakura S, Kaba S, Nakamura S, Seto M, Morishima Y, Tajima K (2001) Association between polymorphisms of folate- and methionine-metabolizing enzymes and susceptibility to malignant lymphoma. Blood 97:3205–3209
Niclot S, Pruvot Q, Besson C, Savoy D, Macintyre E, Salles G, Brousse N, Varet B, Landais P, Taupin P, Junien C, Baudry-Bluteau D (2006) Implication of the folate–methionine metabolism pathways in susceptibility to follicular lymphomas. Blood 108:278–285
Skibola CF, Smith MT, Kane E, Roman E, Rollinson S, Cartwright RA, Morgan G (1999) Polymorphisms in the methylenetetrahydrofolate reductase gene are associated with susceptibility to acute leukemia in adults. Proc Natl Acad Sci USA 96:12810–12815
Skibola C, Forrest SM, Coppede F, Agana L, Hubbard A, Smith MT, Bracci PM, Holly EA (2004) Polymorphisms and haplotypes in folate-metabolizing genes and risk of non-Hodgkin lymphoma. Blood 104:2155–2162
Slattery ML, Potter JD, Samowitz W, Schaffer D, Leppert M (1999) methylenetetrahydrofolate reductase, diet, and risk of colon cancer. Cancer Epidemiol Biomarkers Prev 8:513–518
Ulrich CM, Yasui Y, Storb R, Schubert MM, Wagner JL, Bigler J, Ariail SK, Keener CL, Li S, Liu H, Farin FM, Potter JD (2001) Pharmacogenetic of methotrexate: toxicity among marrow transplantation patients varies with the methylenetetrahydrofolate reductase C677T polymorphism. Blood 98:231–234
Wiemels JL, Smith RN, Taylor GM, Eden OB, Alexander FE, Greaves MF, United Kingdom Childhood Cancer Study Investigators (2001) Methylenetetrahydrofolate reductase (MTHFR) polymorphisms and risk of molecularly defined subtypes of childhood acute leukemia. Proc Natl Acad Sci USA 98:4004–4009
Wisberg I, Tran P, Christensen B, Sibani S, Rozen R (1998) A second genetic polymorphism in methylenetetrahydrofolate reductase (MTHFR) associated with decreased enzyme activity. Mol Genet Metab 64:169–172
Zanrosso CW, Hatagima A, Emerenciano M, Ramos F, Figueiredo A, Felix TM, Segal SL, Guigliani R, Muniz MTC, Pombo-de-Oliveira MS (2006) The role of methylenetetrahydrofolate reductase in acute lymphoblastic leukemia in a Brazilian mixed population. Leukemia Res 2006 30:477–481
Zingg JM, Jones PA (1997) Genetic and epigenetic aspects of DNA methylation on genome expression, evolution, mutation and carcinogenesis. Carcinogenesis 18:869–882
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Timuragaoglu, A., Dizlek, S., Uysalgil, N. et al. Methylenetetrahydrofolate reductase C677T polymorphism in adult patients with lymphoproliferative disorders and its effect on chemotherapy. Ann Hematol 85, 863–868 (2006). https://doi.org/10.1007/s00277-006-0175-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-006-0175-4