Abstract
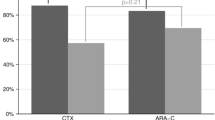
Between June 2003 and November 2004, we collected mobilized peripheral blood units from 29 patients with non-Hodgkin’s lymphoma and multiple myeloma for autologous peripheral blood stem cell transplantation. They received granulocyte colony-stimulating factor (G-CSF) (16 μg/kg/day) for a total of 5 days. Immediately before and 3 h after the fourth and fifth dose of G-CSF, we performed flow cytometry analysis to quantify: T cells (CD3+CD4+, CD3+CD8+), B cells (CD19+), NK cells (CD3-CD16+CD56+), NKT cells (CD3+CD16+CD56+), type 1 dendritic cells (DC1) (lin-HLA-DR+CD11c+), type 2 dendritic cells (DC2) (lin-HLA-DR+CD123+), regulatory T cells (Tregs) (CD4+CD25+), and activated T cells (CD3+HLA-DR+). All cell subsets were mobilized after G-CSF treatment with the exception of B, NK, and NKT lymphocytes. The median number of Treg cells before and after G-CSF was statistically different (29±14.9×106/l vs 70.1±46.1×106/l, P<0.02). DCs were mobilized significantly with a 5.9-fold increase in DC2 (15.1±30.3×106/l vs 89.8±81.0×106/l, P<0.02) and a 2.6-fold increase for DC1 (41±42.5×106/l vs 109.5±58.0×106/l, P<0.04). Patients received a mean of 3.1±1.2×107/kg NK cells, 1.3±0.9×107/kg NKT cells, 0.41±0.29×107/kg DC1, 0.2±0.22×107/kg DC2, and 1.8±1.9×107/kg Tregs. In conclusion, intermediate doses of G-CSF induce mobilization of different lymphocyte subsets, with the exception of B, NK, and NKT cells. The mobilization of certain suppressive populations (DC2 and Treg) could be in theory deleterious, at least in patients with cancer.



Similar content being viewed by others
References
Bensinger WI, Weaver CH, Appelbaum FR (1995) Transplantation of allogeneic peripheral blood stem cells mobilized by recombinant human granulocyte colony-stimulating factor. Blood 85:1655–1658
Bolwell B, Sobecks R, Pohlman B, Andresen S, Theil K, Serafino S, Rybicki L, Kalaycio M (2003) Etoposide (VP-16) plus G-CSF mobilizes different dendritic cell subsets than does G-CSF alone. Bone Marrow Transplant 31:95–98
Chin-Yee I, Keeney M, Anderson L, Nayar R, Sutherland DR (1997) Current status of CD34+ cell analysis by flow cytometry. The ISHAGE Guidelines. Clin Immunol 17:21–29
Damiani D, Fanin R, Silvestri F (1997) Randomized trial of autologous filgrastim-primed bone marrow transplantation versus filgrastim-mobilized peripheral blood stem cell transplantation in lymphoma patients. Blood 90:36–42
Dhodapkar MV, Geller MD, Chang DH, Shimizu K, Fujii SI, Dhodapkar KM, Krasovsky J (2003) A reversible defect in natural killer T cell function characterizes the progression of premalignant to malignant multiple myeloma. J Exp Med 197:1667–1676
Dreger P, Haferlach T, Eckstein V (1994) G-CSF mobilized peripheral blood progenitor cells for allogeneic transplantation: safety, kinetics of mobilization, and composition of the graft. Br J Haematol 87:609–613
Fontenot JD, Gavin MA, Rudensky AY (2003) Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol 4:330–336
Gasparetto C, Gasparetto M, Morse M, Rooney B, Vredenburgh JJ, Long GD, Rizzieri DA, Loftis J, Chao NJ, Smith C (2002) Mobilization of dendritic cells from patients with breast cancer into peripheral blood stem cell leukapheresis samples using Flt-3-ligand and G-CSF or GM-CSF. Cytokine 18:8–19
Gavin MA (2002) Homeostasis and anergy of CD4+CD25+ suppressor T cells in vivo. Nat Immunol 3:33–41
Lanza F, Healy J, Sutherland DR (2001) Structural and functional features of the CD34 antigen: an update. J Biol Regul Homeost Agents 15:1–13
Levings MK, Sangregorio R, Galbiati F, Squadrone S, De Waal Malefyt R, Roncarolo MG (2001) IFN-α and IL-10 induce the differentiation of human type 1 T regulatory cells. J Immunol 166:5530–5539
Liso A, Stockerl-Goldstein K, Aufferman-Gretziner S, Benike C, Reichardt V, Van Beckhoven A, Rajapaksa R, Engelman E, Blume K, Levy R (2000) Idiotype vaccination using dendritic cells after autologous peripheral blood progenitor cell transplantation for multiple myeloma. Biol Blood Marrow Transplant 6:621–627
Lonial S, Hicks M, Rosenthal H, Langston A, Redei I, Torre C, Duenzl M, Feinstein B, Cherry J, Waller E (2004) A randomized trial comparing the combination of granulocyte–macrophage colony-stimulating factor plus granulocyte colony-stimulating factor versus granulocyte colony-stimulating factor for mobilization of dendritic cell subsets in hematopoietic progenitor cell products. Biol Blood Marrow Transplant 10:848–857
McHugh RS (2002) CD4+CD25+ immunoregulatory T cells: gene expression analysis reveals a functional role of the glucocorticoid-induced TNF receptor. Immunity 16:311–323
Miller JS, Prosper F, McCullar V (1997) Natural killer (NK) cells are functionally abnormal and NK cells progenitors are diminished in granulocyte colony-stimulating factor-mobilized peripheral blood progenitor cell collections. Blood 90:3098–3105
Morse M, Zhou L, Tedder T, Lyerly H, Smith C (1997) Generation of dendritic cells in vitro from peripheral blood mononuclear cells with GM-CSF, IL-4 and TNF-α. Ann Surg 226:6–16
Morse MA, Nair S, Fernandez-Casal M, Deng Y, St Peter M, Williams R, Hobeika A, Mosca P, Clay T, Ian Cumming R, Fisher E, Clavien P, Proia AD, Niedzwiecki D, Caron D, Lyerly H (2000) Preoperative mobilization of circulating dendritic cells by Flt3 ligand administration to patients with metastatic colon cancer. J Clin Oncol 18(23):3883–3893
Peron JM, Esche C, Subbotin VM, Maliszewski C, Lotze MT, Shurin MR (1998) Flt3-ligand administration inhibits liver metastases: role of NK cells. J Immunol 161:6164–6170
Rutella S, Pierelli L, Bonanno G, Sica S, Ameglio F, Capoluongo E, Mariotti A, Scambia G, d’Onofrio G, Leone G (2002) Role for granulocyte colony-stimulating factor in the generation of human T regulatory type I cells. Blood 100(7):2562–2571
Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M (1995) Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol 155(3):1151–1164
Sheridan WP, Begley CG, Juttner CA (1992) Effect of peripheral-blood progenitor cells mobilized by filgrastim (G-CSF) on platelet recovery after high-dose chemotherapy. Lancet 339:640–644
Shier LR, Schultz KR, Imren S, Regan J, Issekutz A, Sadek I, Gilman A, Luo Z, Panzarella T, Eaves CJ, Couban S (2004) Differential effects of granulocyte colony-stimulating factor on marrow- and blood-derived hematopoietic and immune cell populations in healthy human donors. Biol Blood Marrow Transplant 10:624–634
Shimizu J, Yamazaki S, Sakaguchi S (1999) Induction of tumor immunity by removing CD25+CD4+ T cells: a common basis between tumor immunity and autoimmunity. J Immunol 162:5211–5218
Shpall EJ, Wheeler CA, Turner SA (1999) A randomized phase 3 study of peripheral blood progenitor cell mobilization with stem cell factor and filgrastim in high-risk breast cancer patients. Blood 93:2491–2501
Siena S, Bregni M, Brando B, Ravagnani F, Bonadona G, Gianni AM (1989) Circulation of CD34+ hematopoietic stem cells in the peripheral blood of high-dose cyclophosphamide-treated patients: enhancement by intravenous recombinant human granulocyte–macrophage colony-stimulating factor. Blood 74:1905–1914
Simmet SJ, Stewart LA, Sweetenham J, Morgan G, Johnson PWM (2000) Autologous stem cell transplantation for malignancy: a systematic review of the literature. Clin Lab Haematol 22(2):61–72
Sosman JA, Stiff P, Moss SM, Sorokin P, Martone B, Bayer R, Van Besien K, Devine S, Stock W, Peace D, Chen Y, Long C, Gustin D, Viana M, Hoffman R (2001) Pilot trial of interleukin-2 with granulocyte colony-stimulating factor for the mobilization of progenitor cells in advanced breast cancer patients undergoing high-dose chemotherapy: expansion of immune effectors within the stem-cell graft and post-stem-cell infusion. J Clin Oncol 19:634–644
Sutherland DR, Anderson L, Keeney M, Nayar R, Chin-yee I (1996) The ISHAGE guidelines for CD34+ cell determination by flow cytometry. J Hematother 5:213–226
Theilgaard-Mönch K, Raaschou-Jensen K, Palm H, Schjodt K, Heilmann C, Vindelov L, Jaconsen N, Dickmeiss E (2001) Flow cytometry assessment of lymphocyte subsets, lymphoid progenitors, and hematopoietic stem cells in allogeneic stem cell grafts. Bone Marrow Transplant 28:1073–1082
Timmerman JM, Levy R (1999) Dendritic cell vaccination for cancer immunotherapy. Annu Rev Med 50:507–529
To LB, Shepperd KM, Haylock DN (1990) Single high doses of cyclophosphamide enable the collection of high numbers of hemopoietic stem cells from the peripheral blood. Exp Hematol 18:442–449
Van Maurik A, Herber M, Wood KJ, Jones ND (2002) Cutting edge: CD4+CD25+ alloantigen-specific immunoregulatory cells that can prevent CD8+ T cell-mediated graft rejection: implications for anti-CD154 immunotherapy. J Immunol 169(10):5401–5404
Weaver CH, Birch R, Greco FA (1998) Mobilisation and harvesting of peripheral blood stem cells: randomized evaluations of different doses of filgrastim. Br J Haematol 100:338–347
Woo EY, Chi CS, Goletz TJ, Schlienger K, Yeh H, Coukos G (2001) Regulatory CD4+CD25+ T cells in tumors from patients with early-stage non-small lung cancer and late stage ovarian cancer. Cancer Res 61:4766–4772
Acknowledgement
This work was partially supported by grant No. 2004-123 from the FOFOI (Fondo de Fomento a la Investigación, Dirección de Prestaciones Médicas, Coordinación de Investigación en Salud, Instituto Mexicano del Seguro Social, México). The experiments performed in the present paper complied with the current laws of Mexico and were approved by the Ethical Committee in La Raza Medical Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vela-Ojeda, J., García-Ruiz Esparza, M.A., Reyes-Maldonado, E. et al. Peripheral blood mobilization of different lymphocyte and dendritic cell subsets with the use of intermediate doses of G-CSF in patients with non-Hodgkin’s lymphoma and multiple myeloma. Ann Hematol 85, 308–314 (2006). https://doi.org/10.1007/s00277-006-0090-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-006-0090-8