Summary
Background
Mobilization of haematopoietic stem cells for autologous transplantation in lymphoma patients currently lacks optimal and universally accepted protocol guidelines. Available mobilization procedures use either cytokines alone, predominantly granulocyte colony-stimulating factor, or cytokines in combination with chemotherapy. Chemomobilization may increase the CD34+ cell yield, and some studies indicate that a higher number of CD34+ cells reinfused is associated with better outcome after autologous haematopoietic stem cells transplantation. We aimed to compare the peak of CD34+ cells in peripheral blood between two alternative mobilization therapies in a retrospective cohort of patients affected by lymphoma.
Methods
Between 2001–2012, 125 retrospectively observed lymphoma patients were treated with either cytarabine (\(n = 36\)) or cyclophosphamide (\(n = 89\)), both combined with granulocyte colony-stimulating factor. The difference in the peak of circulating CD34+ cells was the study primary endpoint.
Results
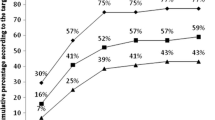
The peak of circulating CD34+ cells were significantly higher in the cytarabine group compared with the cyclophosphamide group (median 129/μL vs 77/μL; \(p < 0.05\)), even when accounting for the timing at collection (169% higher in the cytarabine group, \(p < 0.01\)). Haematopoietic stem cells median time collection started 3 days later in the cytarabine group (16 vs 13 days; \(p < 0.01\)). A leukaphaeresis single session was adequate to collect an optimal number of CD34+ cells (greater than \(5 \times 10^{6}/\text{kg}\)) in 69% patients in the cytarabine group compared with 57% in the cyclophosphamide group (\(p = 0.21\)).
Conclusions
Cytarabine gives a higher peak of circulating CD34+ cells in comparison with cyclophosphamide in a retrospective observational cohort of lymphoma patients submitted to chemomobilization.
Riassunto
Premesse
Attualmente la mobilizzazione di cellule staminali emopoietiche autologhe in pazienti affetti da linfoma manca di linee guida ottimali e universalmente riconosciute. Le procedure di mobilizzazione disponibili si basano sulla sola stimolazione con citochine, prevalentemente fattore stimolante le colonie granulocitarie, o una combinazione di citochine e chemioterapia. La chemiomobilizzazione può incrementare il numero di cellule CD34+ raccolte, inoltre alcuni studi indicano come una maggior dose di CD34+ reinfuse si associ a un migliore outcome dopo il trapianto autologo di cellule staminali emopoietiche. Scopo del nostro studio è stato confrontare il picco di cellule CD34+ periferiche ottenuto con due differenti regimi di chemiomobilizzazione in una coorte retrospettiva di pazienti affetti da linfoma.
Metodi
Tra il 2001 e il 2012, 125 pazienti affetti da linfoma sono stati mobilizzati con citarabina (\(n = 36\)) o ciclofosfamide (\(n = 89\)), entrambe associate con fattore stimolante le colonie granulocitarie. La differenza nel picco delle cellule CD34+ circolanti è stato l’obiettivo primario dello studio.
Risultati
Il picco di cellule CD34+ circolanti era significativamente più elevato nel gruppo trattato con citarabina rispetto a quello trattato con ciclofosfamide (mediana 129/μL vs 77/μL; \(p < 0{,}05\)), anche considerando il giorno di raccolta (169% più elevato nel gruppo citarabina, \(p < 0{,}01\)). La raccolta di cellule staminali emopoietiche iniziava con una mediana di 3 giorni di ritardo nel gruppo citarabina (16 vs 13 giorni; \(p < 0{,}01\)). Una singola staminaferesi permetteva la raccolta di un numero ottimale di cellule CD34+ (\(> 5 \times 10^{6}/\text{kg}\)) nel 69% dei pazienti trattati con citarabina rispetto al 57% di quelli con ciclofosfamide \((p = 0{,}21)\).
Conclusioni
Citarabina fornisce un picco di cellule CD34+ circolanti più elevato rispetto a ciclofosfamide in una coorte retrospettiva di pazienti affetti da linfomi sottoposti a chemiomobilizzazione.


Similar content being viewed by others
References
Passweg JR, Baldomero H, Peters C et al. (2014) Hematopoietic SCT in Europe: data and trends in 2012 with special consideration of pediatric transplantation. Bone Marrow Transplant 49:744–750
Siena S, Schiavo R, Pedrazzoli P et al. (2000) Therapeutic relevance of CD34 cell dose in blood cell transplantation for cancer therapy. J Clin Oncol 18:1360–1377
Bolwell BJ, Pohlman B, Rybicki L et al. (2007) Patients mobilizing large numbers of CD34+ cells (‘super mobilizers’) have improved survival in autologous stem cell transplantation for lymphoid malignancies. Bone Marrow Transplant 40:437–441
Jantunen E, Fruehauf S (2011) Importance of blood graft characteristics in auto-SCT: implications for optimizing mobilization regimens. Bone Marrow Transplant 46:627–635
Meldgaard Knudsen L, Jensen L, Gaarsdal E et al. (2000) A comparative study of sequential priming and mobilisation of progenitor cells with rhG-CSF alone and high-dose cyclophosphamide plus rhG-CSF. Bone Marrow Transplant 26:717–722
Narayanasami U, Kanteti R, Morelli J et al. (2001) Randomized trial of filgrastim versus chemotherapy and filgrastim mobilization of hematopoietic progenitor cells for rescue in autologous transplantation. Blood 98:2059–2064
Toor AA, van Burik JA, Weisdorf DJ (2001) Infections during mobilizing chemotherapy and following autologous stem cell transplantation. Bone Marrow Transplant 28:1129–1134
Kuittinen T, Nousiainen T, Halonen P et al. (2004) Prediction of mobilisation failure in patients with non-Hodgkin’s lymphoma. Bone Marrow Transplant 33:907–912
Perseghin P, Terruzzi E, Dassi M et al. (2009) Management of poor peripheral blood stem cell mobilization: incidence, predictive factors, alternative strategies and outcome. A retrospective analysis on 2177 patients from three major Italian institutions. Transfus Apher Sci 41:33–37
Hosing C, Saliba RM, Ahlawat S et al. (2009) Poor hematopoietic stem cell mobilizers: a single institution study of incidence and risk factors in patients with recurrent or relapsed lymphoma. Am J Hematol 84:335–337
Wuchter P, Ran D, Bruckner T et al. (2010) Poor mobilization of hematopoietic stem cells-definitions, incidence, risk factors, and impact on outcome of autologous transplantation. Biol Blood Marrow Transplant 16:490–499
Gidron A, Singh V, Egan K et al. (2008) Significance of low peripheral blood CD34+ cell numbers prior to leukapheresis: what should the threshold required for apheresis be? Bone Marrow Transplant 42:439–442
Pavone V, Gaudio F, Console G et al. (2006) Poor mobilization is an independent prognostic factor in patients with malignant lymphomas treated by peripheral blood stem cell transplantation. Bone Marrow Transplant 37:719–724
Gambell P, Herbert K, Dickinson M et al. (2012) Peripheral blood CD34+ cell enumeration as a predictor of apheresis yield: an analysis of more than 1,000 collections. Biol Blood Marrow Transplant 18:763–772
Olivieri A, Marchetti M, Lemoli R et al. (2012) Proposed definition of ‘poor mobilizer’ in lymphoma and multiple myeloma: an analytic hierarchy process by ad hoc working group Gruppo ItalianoTrapianto di Midollo Osseo. Bone Marrow Transplant 47:342–351
Hübel K, Fresen MM, Apperley JF et al. (2012) European data on stem cell mobilization with plerixafor in non-Hodgkin’s lymphoma, Hodgkin’s lymphoma and multiple myeloma patients. A subgroup analysis of the European Consortium of stem cell mobilization. Bone Marrow Transplant 47:1046–1050
Fitoussi O, Perreau V, Boiron JM et al. (2001) A comparison of toxicity following two different doses of cyclophosphamide for mobilization of peripheral blood progenitor cells in 116 multiple myeloma patients. Bone Marrow Transplant 27:837–842
Hiwase DK, Bollard G, Hiwase S et al. (2007) Intermediate-dose CY and G-CSF more efficiently mobilize adequate numbers of PBSC for tandem autologous PBSC transplantation compared with low-dose CY in patients with multiple myeloma. Cytotherapy 9:539–547
Giebel S, Kruzel T, Czerw T et al. (2013) Intermediate-dose Ara-C plus G-CSF for stem cell mobilization in patients with lymphoid malignancies, including predicted poor mobilizers. Bone Marrow Transplant 48:915–921
Di Nicola M, Siena S, Bregni M et al. (1993) Quantization of CD34+ peripheral blood hematopoietic progenitors for autografting in cancer patients. Int J Artif Organs 5:80–82
Sheppard D, Bredeson C, Allan D et al. (2012) Systematic review of randomized controlled trials of hematopoietic stem cell mobilization strategies for autologous transplantation for hematologic malignancies. Biol Blood Marrow Transplant 18:1191–1203
Giralt S, Costa L, Schriber J et al. (2014) Optimizing autologous stem cell mobilization strategies to improve patien outcomes: consensus guidelines and recommendations. Biol Blood Marrow Transplant 20:295–308
Bensinger W, DiPersio JF, McCarty JM (2009) Improving stem cell mobilization strategies: future directions. Bone Marrow Transplant 43:181–195
Tarella C, Di Nicola M, Caracciolo D et al. (2002) High-dose ara-C with autologous peripheral blood progenitor cell support induces a marked progenitor cell mobilization: an indication for patients at risk for low mobilization. Bone Marrow Transplant 30:725–732
Montillo M, Tedeschi A, Rossi V et al. (2004) Successful CD34+ cell mobilization by intermediate-dose Ara-C in chronic lymphocytic leukemia patients treated with sequential fludarabine and Campath-1H. Leukemia 18:57–62
Kruzel T, Sadus-Wojciechowska M, Najda J et al. (2012) Very high efficacy of intermediate-dose cytarabine in combination with G-CSF as a second-line mobilization of hematopoietic stem cells. Int J Hematol 96:287–289
Perseghin P, Marchetti M, Pierelli L et al. (2014) A policy for the disposal of autologous hematopoietic progenitor cells: report from an Italian consensus panel. Transfusion 54:2353–2360
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Human and animal rights
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Salvadori, U., Melotti, R., Al-Khaffaf, A. et al. Cytarabine assures a high concentration of circulating CD34+ cells during haematopoietic stem cells mobilization in lymphoma patients. Riv Ital Med Lab 13, 173–179 (2017). https://doi.org/10.1007/s13631-017-0165-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13631-017-0165-7
Keywords
- Haematopoietic stem cell mobilization
- Peripheral blood stem cell transplantation
- Cytarabine
- Cyclophosphamide