Abstract
Purpose
Peripheral arterial calcification is an important predictor of outcomes after both conservative and endovascular treatment. Digital subtraction angiography (DSA)-based calcification scores are limited by low sensitivity and inter-observer agreement. The Peripheral Arterial Calcium Scoring System (PACSS) assesses the severity of target lesion calcification. The newly introduced modified PACSS (mPACSS) also evaluates target vessel calcification. This study aimed to assess the inter- and intra-observer reliability of PACSS and mPACSS on computed tomography angiography (CTA) in (infra)popliteal endovascular interventions.
Methods
A random sample of 50 limbs from the prospective multicenter Dutch Chronic Lower Limb-Threatening Ischemia Registry (THRILLER) were included. Three experienced independent raters scored PACSS on CTA. Three months later, one blinded rater assessed the same 50 CTA scans, keeping track of assessment time. The reliability of the original 5-step PACSS, a simplified binary PACSS (0–2 vs 3–4) and the 7-step mPACSS were tested using Cohen’s and Fleiss’ kappa statistics.
Results
In total, 50 limbs (mean age 70.1 ± 11.0, 29 men) with 41 popliteal and 40 infrapopliteal lesions were scored. Inter-observer agreement of PACSS and binary PACSS were moderate (κ = 0.60) and substantial (κ = 0.72), respectively, while intra-observer agreement was almost perfect in both scores (κ = 0.86). Inter- and intra-observer agreement of mPACSS were moderate (κ = 0.48) and substantial (κ = 0.77), respectively. Mean assessment time for an experienced rater was 3.43 ± 0.93 min per CTA scan.
Conclusion
Both the semi-quantitative PACSS and mPACSS scores for (infra)popliteal arteries can be performed reliably on pre-operative CTA.
Graphic Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The global prevalence of peripheral arterial disease (PAD) is increasing, with a particular rise in the proportion of patients with chronic limb-threatening ischemia (CLTI) [1]. This has resulted in an increasing demand for below the knee (BTK) revascularizations, which can be challenging due to long lesion length, small vessel diameter and severe calcification [2].
In the field of PAD, severe calcification complicates endovascular interventions because it causes difficulties to cross the lesion, leads to early elastic recoil and incomplete expansion of stents, and forms a mechanical barrier for drug penetration when using drug-eluting devices [3]. In previous studies, severe calcification was associated with an increased risk of technical failure, loss of patency, clinically driven target lesion revascularization (CD-TLR), major adverse limb events (MALE), amputation and mortality in both conservative patients and after endovascular intervention [4,5,6,7,8,9,10,11,12,13,14,15].
Multiple quantitative and semiquantitative peripheral calcification scores have been developed and used in clinical studies and trials [4,5,6,7,8,9,10,11,12,13,14,15]. The most commonly used scores, the semiquantitative peripheral arterial calcium scoring system (PACSS) and peripheral academic research consortium (PARC), are scored on two-dimensional digital subtraction angiography (DSA) imaging [3, 16]. The accuracy and reliability of these scores are important for the interpretation of trial outcomes that depend on the severity of calcification, such as device trials. However, previous studies concluded that DSA is limited in its ability to identify calcium and the inter-observer agreement of both PACSS and PARC was minimal [16, 17]. These studies also mention that methods using cross-sectional imaging are warranted and that computed tomography angiography (CTA)-based systems may outperform two-dimensional DSA-based systems.
Lastly, it is relevant to include the calcification of the entire target vessel (TV) rather than just target lesion (TL) calcification [6, 15]. Therefore, we developed the modified PACSS (mPACSS), which integrates calcification of the TL and calcification of the entire TV into one scoring method.
The aim of this study was to assess the inter- and intra-observer reliability of the original and a modified PACSS score based on CTA imaging in popliteal and infrapopliteal endovascular interventions.
Methods
A random sample of 50 limbs was used from two hospitals that participate in the DuTcH chRonIc Lower Limb-threatening ischEmia Registry (THRILLER). THRILLER is an ongoing national multicenter prospective registry that includes all consecutive patients that undergo a popliteal or infrapopliteal endovascular intervention in 7 Dutch hospitals between February 2021 and December 2023. The protocol was approved by the local ethics committees and was previously published [18]. Informed consent was obtained from all patients. Despite the target population being patients with CLTI, patients were included on the basis of the treatment location (popliteal and the first 2/3 of the infrapopliteal arteries) and not on their clinical manifestation. Exclusion criteria were patients with acute limb ischemia, (infra)popliteal interventions as a result of distal embolization and patients unable to give informed consent.
For this retrospective analysis of prospectively collected data, patients were extracted who underwent a CTA scan before the index procedure. Whether a CTA scan had been performed depended on center and physician preference, indication and the presence of recent imaging. Data regarding patient demographics, comorbidities, medical examinations and imaging, lesion characteristics and procedural characteristics were collected from electronic medical records and entered into an online data capture software.
Peripheral Arterial Calcium Scoring System
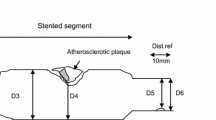
PACSS was originally designed to be scored on the basis of DSA [3]. However, in this study, all lesions were scored according to the PACSS score based on the preoperative CTA scan between 1 and 1.5 mm slice thickness reconstructions. Bone window settings were used as reference window by all raters and adjusted individually for optimal visualization. Our CTA protocol used a 120 kV with a reference mAs between 100 and 150 depending on patients weight. Solitary lesions in the most distal one-third of the infrapopliteal arteries were not scored, because assessment of circumference in this segments could not be performed reliably. The PACSS score is a semi-quantitative score, meaning that quantitative data is transformed into numerical data to allow for more robust group comparisons. PACSS grades circumference (unilateral vs. bilateral or circumferential) and length (5 cm cut-off) of TL calcification, grading lesions from PACSS 0 to 4 (Figs. 1 and 2). In addition, the original 5-step PACSS score was simplified into a binary score; non-severe calcification was assigned to patients with PACSS grade 0–2 and classified as 0, severe calcification was assigned to patients with PACSS grade 3 and 4 and classified as 1 [9,10,11,12,13, 16].
The peripheral arterial calcium scoring system (PACSS) and its modification (mPACSS). The original peripheral arterial calcium scoring system (PACSS) score consists of 5 grades based on the target lesion (TL) calcification. In the mPACSS score, total target vessel (TV) calcification is also scored, resulting in a TV score of 0–2. This score is then added to the existing PACSS score, ultimately giving patients a mPACSS score of 0–6. The TV is the popliteal (POP), tibial anterior (TAA), tibial posterior (TPA) or peroneal artery (PA). In the case the TL is located in both the popliteal and an infrapopliteal artery, both arteries are considered the TV. In the case the TL is located solely in the tibioperoneal trunk (TPT), the TV includes the TPT and the TPA or PA, depending on the target arterial path
Modified PACSS
Because calcification of the entire TV is relevant to predict outcomes after peripheral intervention, an addition was made to the original PACSS score (Fig. 1). In this mPACSS score, total TV calcification is also scored, resulting in a TV score of 0–2. This score is then added to the existing PACSS score, ultimately giving patients a mPACSS score of 0–6. Possible TVs are the popliteal, tibial anterior, tibial posterior (TPA) and peroneal artery (PA). In cases where the TL is located solely in the tibioperoneal trunk (TPT), the TV includes the TPT and the TPA or PA, depending on the target arterial path (TAP).
Reliability Scoring
Three experienced and independent raters (a vascular researcher and two interventional radiologists) assessed the CTA scans of the included patients (M.N., M.S., H.J.S.). The raters received information regarding the treated limb and lesions, but were blind to all other patient data, procedural information and outcomes. The segments to be scored per lesion on CTA were provided in advance and separately scored according to the PACSS and mPACSS scores. In advance, scoring arrangements were made to improve interobserver agreement (Table 1).
To test the intra-observer reliability, one rater (M.N.) assessed the same 50 CTA scans three months after the initial assessment. The rater was blinded to patient data and the initial scores.
Outcomes
The primary outcomes were the inter- and intra-observer reliability of the original 5-step PACSS score, the binary PACSS score, the TV calcification score and the mPACSS score.
The secondary outcome was the mean time needed to assess one CTA scan for an experienced rater, i.e. the rater that assessed the same 50 CTA scans three months after the initial assessment.
Statistical analysis
Continuous variables are presented as mean ± standard deviation and categorical variables as absolute number and proportion of the study population. The Cohen’s Kappa (κ) statistic was used to test the intra-observer reliability of the PACSS and mPACSS scores. The Fleiss’ Kappa (κ) statistic was used to test the inter-observer reliability of both scores. The scale recommended by Landis and Koch was used to interpret the degree of agreement of Kappa results [19]. All statistical analyses were performed using SPSS.
Results
A total of 44 patients with 50 treated limbs with an available CTA scan prior to an endovascular procedure were included in this study. In total, 41 popliteal and 40 infrapopliteal lesions were treated.
Baseline and lesion characteristics are summarized in Table 2. Diabetes mellitus was present in 54.5% of the patients. A previous target limb revascularization was performed in 58.0% of the limbs. Rutherford categories 3–6 were present in 6.0%, 18.0%, 70.0% and 6.0% of the limbs, respectively. Mean lesion length was 60.9 ± 51.8 mm for popliteal and 150.1 ± 130.6 mm for infrapopliteal lesions, respectively. GLASS 4 was present in 65.9% and 50.0% of the popliteal and infrapopliteal lesions, respectively.
PACSS Scores
The inter-observer agreement of the original PACSS and binary PACSS scores were moderate (κ = 0.60 and substantial (κ = 0.72), respectively. The intra-observer agreement of both scores was almost perfect (both: κ = 0.86). The inter- and intra-observer agreement of the PACSS score was not significantly different for infrapopliteal lesions compared with popliteal lesions (Table 3).
Modified PACSS Scores
The inter- and intra-observer agreement of the TV calcification score were substantial (κ = 0.64) and almost perfect (κ = 0.87), respectively. The inter- and intra-observer agreement of the mPACSS score were moderate (κ = 0.48) and substantial (κ = 0.77). The inter-observer agreement of mPACSS was significantly lower for popliteal lesions (k = 0.42, 95% CI 0.34–0.50) compared to infrapopliteal lesions (k = 0.52, 95% CI 0.43–0.60). No other significant differences were observed between popliteal and infrapopliteal lesions (Table 3).
Assessment Time
The mean assessment time for an experienced rater was 3.43 ± 0.93 min per CTA scan. The time for scans with one lesion versus scans with multiple lesions was not significantly different (3.29 ± 0.67 vs. 3.56 ± 1.20).
Discussion
This study showed that scoring PACSS on pre-operative CTA imaging in popliteal and infrapopliteal lesions can be performed reliably if prior scoring arrangements are made. In addition, scoring the calcification of the entire TV appeared to be reliable.
One previous study tested the inter- and intra-observer reliability of semi-quantitative peripheral calcification scores [16]. The referred study scored the PACSS and PARC on DSA and the Fanelli scoring system on both DSA and CTA imaging. All scoring systems were dichotomized and tested on femoropopliteal lesions. The inter- and intra-observer agreement of the binary PACSS were fair (κ = 0.32) and moderate to substantial (κ = 0.52–0.62) according to the scale recommended by Landis and Koch, respectively. The inter- and intra-observer agreement in the other calcification scores were fair (κ = 0.38–0.40) and highly variable (κ = 0.36–0.92), respectively.
The PACSS score has been studied primarily in femoropopliteal lesions, in which higher PACSS grades were significantly associated with loss of primary patency, increased target lesion revascularization (TLR), increased MALE and increased mortality [8,9,10,11,12]. Regarding infrapopliteal disease, one retrospective study found that PACSS grade 4 was associated with failed guidewire crossing of below-the-knee chronic total occlusions in univariate analysis [20]. All these studies scored the grade of calcification on two-dimensional DSA instead of CTA imaging. DSA is superior in assessing vessel patency, the absence of which lowers the likelihood of achieving lesion revascularization. However, CTA imaging has the advantages of three-dimensionality and non-invasiveness, and can be performed preoperatively for risk stratification. In addition, previous studies concluded that DSA is limited in its ability to identify calcium as compared with intravascular ultrasound (IVUS). Most importantly, the inter- and intra-observer agreement of PACSS on DSA were unacceptably low [16, 17]. Therefore, these studies raised serious concerns regarding the accuracy and reliability of the currently most commonly used angiographic calcification scores for PAD.
This study demonstrates that the original 5-step PACSS can be scored reliably on CTA imaging. Besides the original PACSS score, we also tested the reliability of a binary PACSS score. Semi-quantitative scores with more than two categories are suitable for determining the prognosis, however, in clinical practice the interventionalist is confronted with a binary question, namely is the calcification present severe enough to choose for an additional calcium modifying treatment such as atherectomy or specialty balloons[16, 21, 22]. The inter-observer agreement of the binary score was slightly higher than the original 5-step PACSS score, while the intra-observer agreement was similarly strong.
In addition, we decided to add the calcification grade of the entire TV, as it might be a more accurate representation of total limb calcification than TL calcification alone, as measured in the original PACSS score. Two studies already demonstrated that TV calcification of infrapopliteal arteries is a significant predictor of technical success, limb salvage amputation-free survival (AFS) and major adverse cardiovascular events (MACE) [6, 15].
The inter-observer agreement of the trichotomic TV calcification and 7-step mPACSS score was substantial (κ = 0.64) and moderate (κ = 0.48), respectively, while the intra-observer agreement was almost perfect (κ = 0.87) and substantial (κ = 0.77), respectively. The inter-observer reliability in all scores was calculated with Fleiss’ kappa, which is suitable for calculating the absolute agreement of nominal scores without any intrinsic ordering or ranking [19]. On the other hand, in the case of true ordinal scores, weighted kappa values should be calculated. Regarding PACSS and mPACSS, it is debatable whether these scores should be considered nominal or truly ordinal. In case both scores are considered truly ordinal, the weighted kappa values of inter-observer agreement would be substantially higher (PACSS: κ = 0.71–0.76, mPACSS: κ = 0.72–0.77). Supplementary Table 1 summarizes all weighted kappa values of the PACSS score and its modifications.
Previous studies using CTA imaging to score the grade of infrapopliteal calcification used the quantitative tibial artery calcification (TAC) score and found associations between severe TAC and lower technical success, limb salvage, AFS and survival free of MACE [8, 17]. The inter-observer agreement was high for the quantitative TAC score, which makes quantitative calcium scoring a promising method that deserves further evaluation in future research. On the other hand, quantitative scores are not widely available, time consuming and dependent on a certain level of expertise. This study has shown that semi-quantitative scoring of PACSS on CTA is reliable and fast in popliteal and infrapopliteal disease. Next steps are to test the reliability in femoropopliteal disease and to validate this calcium score on a patient cohort with treatment outcomes, preferably a large prospective multi-center cohort. The use of virtual non-contrast techniques may help improve calcium scoring. Ideally, the scoring system should aid the clinician’s decision in the approach for revascularization, thereby improving revascularization success.
Limitations of this retrospective study are the biased study population, small differences in imaging protocols and limited data regarding intra-observer reliability. In this study, 94% of the limbs were included for CLTI and only popliteal and infrapopliteal lesions were included because the study population was extracted from the THRILLER registry. This may affect the generalizability of the findings of this study to the overall PAD population. Secondly, the protocols for CTA imaging were generally consistent, but minor differences existed. For example, slice thickness varied from 1 to 1.5 mm, which made scoring of calcium bilaterality slightly more difficult in the scans with thicker slices, particularly in the infrapopliteal arteries. Thirdly, only one rater scored PACSS twice, which resulted in limited data regarding intra-observer reliability. However, for this rater, substantial to almost perfect agreement was achieved in all scoring components.
Finally, prior scoring arrangements were essential to improve inter-observer agreement, but may have conflicted with the original assignment of PACSS grades. For example, lesions smaller than 5 cm could not be scored in grades 2 and 4. Nevertheless, the exact interpretation of the different PACSS grades remains unclear and is subject for future scientific research [3].
Conclusion
This study showed that both the semi-quantitative PACSS and mPACSS scores for the popliteal and infrapopliteal arteries can be scored reliably on CTA. Future studies should focus on the validation of this calcium scoring system in relation to outcomes, possibly leading to improved patient selection and thereby improved revascularization outcomes.
Abbreviations
- AFS:
-
Amputation-free survival
- BTK:
-
Below the knee
- CAC:
-
Coronary artery calcium
- CAD:
-
Coronary artery disease
- CD-TLR:
-
Clinically driven target lesion revascularization
- CLTI:
-
Chronic limb-threatening ischemia
- CTA:
-
Computed tomography angiography
- DSA:
-
Digital subtraction angiography
- GLASS:
-
Global limb anatomic staging system
- IVUS:
-
Intravascular ultrasound
- MACE:
-
Major adverse cardiovascular events
- MALE:
-
Major adverse limb events
- (m)PACSS:
-
(Modified) Peripheral arterial calcium scoring system
- PA:
-
Peroneal artery
- PAD:
-
Peripheral arterial disease
- PARC:
-
Peripheral academic research consortium
- PCI:
-
Percutaneous coronary intervention
- TAC:
-
Tibial artery calcification
- TAP:
-
Target arterial path
- TPA:
-
Tibial posterior artery
- TPT:
-
Tibioperoneal trunk
- THRILLER:
-
Dutch chronic lower limb-threatening ischemia registry
- TL:
-
Target lesion
- TV:
-
Target vessel
- WIfI:
-
Wound, Ischemia, foot Infection
References
Song P, Rudan D, Zhu Y, Fowkes FJI, Rahimi K, Fowkes FGR, et al. Global, regional, and national prevalence and risk factors for peripheral artery disease in 2015: an updated systematic review and analysis. Lancet Glob Health. 2019;7:e1020–30.
Baumann F, Engelberger RP, Willenberg T, Do D. Infrapopliteal lesion morphology in patients with critical limb ischemia: implications for the development of anti-restenosis technologies. J Endovasc Ther. 2013;20(2):149–56.
Rocha-Singh KJ, Zeller T, Jaff MR. Peripheral arterial calcification: prevalence, mechanism, detection, and clinical implications. Catheter Cardiovasc Interv. 2014;83:E212–20.
Zetterval SL, Marshall AP, Fleser P, Guzman RJ. Association of arterial calcification with chronic limb ischemia in patients with peripheral artery disease. J Vasc Surg. 2018;67:507–13.
Huang C-L, Wu I-H, Wu Y-W, Hwang J-J, Wang S-S, Chen W-J, et al. Association of lower extremity arterial calcification with amputation and mortality in patients with symptomatic peripheral artery disease. PLoS ONE. 2014;9:e90201.
Kang IS, Lee W, Choi BW, Choi D, Hong M-K, Jang Y, et al. Semiquantitative assessment of tibial artery calcification by computed tomography angiography and its ability to predict infrapopliteal angioplasty outcomes. J Vasc Surg. 2016;64:1335–43.
Konijn LCD, Takx RAP, de Jong PA, Spreen MI, Veger HTC, Mali WPTM, et al. Arterial calcification and long-term outcome in chronic limb-threatening ischemia patients. Eur J Radiol. 2020;132:109305.
Tokuda T, Oba Y, Koshida R, Suzuki Y, Murata A, Ito T. The impact of femoropopliteal artery calcium score after endovascular treatment. Ann Vasc Surg. 2020;66:543–53.
Ichihashi S, Shibata T, Fujimura N, Nagatomi S, Yamamoto H, Kyuragi R, et al. Vessel calcification as a risk factor for in-stent restenosis in complex femoropopliteal lesions after zilver PTX paclitaxel-coated stent placement. J Endovasc Ther. 2019;26(5):613–20.
Okuno S, Iida O, Shiraki T, Fujita M, Masuda M, Okamoto S, et al. Impact of calcification on clinical outcomes after endovascular therapy for superficial femoral artery disease: assessment using the peripheral artery calcification scoring system. J Endovasc Ther. 2016;23:731–7.
de Athayde SR, Matielo MF, Brochado Neto FC, Pires APM, de Almeida RD, de Jesus MM, et al. Impact of calcification and infrapopliteal outflow on the outcome of endovascular treatment of femoropopliteal occlusive disease. JRSM Cardiovasc Dis. 2019;8:2048004019828941.
Yoshioka N, Tokuda T, Koyama A, Yamada T, Nishikawa R, Shimamura K, et al. Clinical outcomes and predictors of restenosis in patients with femoropopliteal artery disease treated using polymer-coated paclitaxel-eluting stents or drug-coated balloons. Heart vessel. 2022;37:555–66.
Tepe G, Beschorner U, Ruether C, Fischer I, Pfaffinger P, Noory E, et al. Drug-eluting balloon therapy for femoropopliteal occlusive disease: predictors of outcome With a special emphasis on calcium. J Endovasc Ther. 2015;22:727–33.
He H, Weng J, Zhao Y, Cai S, Zhang X, Yin H. Impact of plaque calcification and stent oversizing on clinical outcomes of atherosclerotic femoropopliteal arterial occlusive disease following stent angioplasty. Eur J Vasc Endovasc Surg. 2019;58:215–22.
Guzman RJ, Brinkley DM, Schumacher PM, Donahue RMJ, Beavers H, Qin X. Tibial artery calcification as a marker of amputation risk in patients with peripheral arterial disease. J Am Coll Cardiol. 2008;51:1967–74.
Allan RB, Wise NC, Wong YT, Delaney CL. Accuracy and reliability of peripheral artery calcium scoring systems using an intravascular ultrasound reference standard. Ann Vasc Surg. 2023;91:233–41.
Yin D, Maehara A, Shimshak TM, Ricotta JJ, Ramaiah V, Foster MT, et al. Intravascular ultrasound validation of contemporary angiographic scores evaluating the severity of calcification in peripheral arteries. J Endovasc Ther. 2017;24:478–87.
Nugteren MJ, Hazenberg CEVB, Akkersdijk GP, Bakker OJ, Dinkelman MK, Fioole B, et al. The Dutch chronic lower limb-threatening ischemia registry (THRILLER): a study protocol for popliteal and infrapopliteal endovascular interventions. PLoS One. 2023;18:e0288912.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977;33:159–74.
Tan M, Ueshima D, Urasawa N, Hayakawa N, Dannoura Y, Itoh T, et al. Prediction of successful guidewire crossing of below-the-knee chronic total occlusions using a Japanese scoring system. J Vasc Surg. 2021;74:506–13.
Nugteren MJ, Welling RHA, Bakker OJ, Ünlü Ç, Hazenberg CEVB. Vessel preparation in infrapopliteal arterial disease: a systematic review and meta-analysis. J Endovasc Ther. 2022;31:191.
Nugteren MJ, Hazenberg CEVB, Akkersdijk GP, van den Heuvel DAF, Schreve MA, Ünlü Ç. Twelve-month outcomes of intravascular lithotripsy for treatment of calcified popliteal and infrapopliteal lesions in patients with chronic limb-threatening ischemia. J Endovasc Ther. 2023. https://doi.org/10.1177/15266028231205421.
Acknowledgements
We would like to thank everyone who helped organize the center-specific logistics in the participating medical centers of THRILLER, such as the inclusion and follow-up. Furthermore, we would like to thank Prof. dr. M.L. Bots for his kindly provided statistical advice for this manuscript.
Funding
THRILLER was supported by unrestricted grants by Bayer and Abbott. The funders did not have a role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
Institutional Review Board approval was obtained. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Consent for Publication
For this type of study consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nugteren, M.J., Ünlü, Ç., Samim, M. et al. Inter- and Intra-observer Agreement of the Peripheral Arterial Calcium Scoring System in Patients Undergoing (Infra)Popliteal Endovascular Interventions. Cardiovasc Intervent Radiol (2024). https://doi.org/10.1007/s00270-024-03839-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00270-024-03839-1