Abstract
Electrochemotherapy (ECT) is a novel non-thermal ablative technique that combines chemotherapy and the application of electric pulses for reversible cell membrane electroporation. This method was recently performed in the treatment of deep-seated liver tumors during open surgery but experience about percutaneous ECT is rare and further developments like combination of percutaneous ECT with stereotactic navigated devices may be very promising. We report on a case of a 4.7 × 4.5 × 3.5 cm unresectable HCC at the hepatic hilum adjacent to the major vessels and the bile duct that was successfully treated using percutaneous ECT in combination with stereotactic navigation. Follow-up imaging 6 weeks and 6 months after ECT showed complete response.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, local ablative procedures have become a curative treatment option even for patients with increasingly large hepatocellular carcinoma (HCC). This is due to technical advances in (1) thermal ablative procedures such as multiantenna microwave ablation (MWA) [1, 2], (2) combination of radiofrequency ablation (RFA) or MWA with transarterial chemoembolization (TACE) [3] and (3) the use of stereotactic navigation devices [4,5,6]. Irreversible electroporation (IRE) is a non-thermal treatment method, which allows treatment of HCC lesions in close proximity to major blood vessels without the disadvantages of thermal ablation methods like heat sink effect and without the risk of injury of central vessels and bile ducts [7]. This renders IRE a good treatment option for tumor lesions close to the liver hilum. However, this method is limited to tumors with a maximum diameter of about 3 cm.
Resulting from the abovementioned limitations percutaneous ablation can be very challenging, especially in case of a large central tumors.
Electrochemotherapy (ECT) is a non-thermal ablative technique that combines chemotherapy and the application of electric pulses for reversible cell membrane electroporation. Consequently, mitotic tumor cell death results through high intracellular concentrations of the chemotherapeutic agent. In contrast to ECT, IRE uses irreversible cell membrane electroporation (without the need of additional drugs) that results in damage to the cell membrane and apoptosis. ECT has proven to be a safe and effective treatment for cutaneous tumors/metastases and first case series state, that ECT is also an effective treatment method for liver lesions in intraoperative settings [8,9,10].
We report a case of ECT for treatment of a large central HCC using a stereotactically navigated percutaneous approach.
Patient and Technique
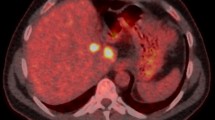
A 75-years old male patient with Child A liver cirrhosis presented with a 4.7 × 4.5 × 3.5 cm large central HCC involving liver segments IV/VIII and a satellite HCC (1.3 × 1.3 × 1.2 cm, segment V). The portal vein showed thrombosis resulting in cavernous transformation while the hepatic artery and the central bile ducts were located in close proximity to the HCC resulting in local cholestasis (Fig. 1). Alpha-fetoprotein showed a slight elevation with 9.3 ng/ml. The case was reviewed in our multidisciplinary tumor board (consisting of hepatologists, oncologists, liver transplant surgeons, interventional radiologists, radiation therapists, nuclear medicine physicians and pathologists). The decision to perform percutaneous ECT of the central lesion was made because of the large size and the proximity to the hepatic artery and central bile duct. MWA of the satellite tumor was scheduled because of its size of less than 3 cm and a more peripheral location.
75-years old male patient with Child A liver cirrhosis and a 4.7 × 4.5 × 3.5 cm large central HCC BCLC B involving liver segments IV/VIII (asterisks). A Contrast-enhanced CT in arterial phase. B Contrast-enhanced MRI, T1 vibe 3d fs in arterial phase. Arrows mark close proximity of the tumor to the liver artery; Arrowheads mark close proximity of the tumor to the central bile ducts, resulting in local cholestasis
Technique of Stereotactic Percutaneous Electrochemotherapy
The procedure was performed under general anesthesia and deep muscle relaxation in the supine position with the right side slightly lifted. A Somatom Definition Edge CT-scanner (Siemens, Forchheim, Germany) was used in combination with a stereotactic navigation system (CAS-ONE IR, CAScination AG, Bern, Switzerland) for optimized percutaneous placement of ECT electrodes (IGEA Cliniporator VITAE, IGEA S.p.A., Carpi, Italy). Induction of general anesthesia, patient preparation and setup of the stereotactic navigation system took 37 min. Six electrodes with 4.0 cm active tip were placed within and around the tumor with the distance between the electrode-tips ranging from 2.1 to 3.0 cm (Fig. 2). Stereotactic placement of the 6 ECT-electrodes took 59 min, including planning of the trajectories. Bleomycin was selected as chemotherapeutic agent because, unlike other drugs for ECT, it can be administered intravenously [11]. 30000 IU of Bleomycin diluted in 10 ml NaCl (15000 IU/m2 body surface area) was injected intravenously (as bolus); 8 min after Bleomycin injection, 8 electric pulses with a frequency of 1000 Hz, impulse length of 100 µs, and voltages ranging from 1.4 to 3 kV were delivered between each electrode pairing over a timespan of 16 min. Stereotactic MWA of the satellite tumor was performed during the same session. A control-scan in arterial and portal-venous contrast phase showed complete devascularization of both, tumor and satellite. No periinterventional adverse events occurred. Total procedural time (including patient preparation, time under anesthesia, ECT and MWA of the satellite tumor) took 3 h and 24 min.
Technical details of the procedure. A Treatment setting in the interventional CT suite using stereotactic navigation (Cascination CAS-ONE IR). B Planning and validation of electrode placement, using the 3D model, generated by the navigation software. C Percutaneous placement of 6 ECT-electrodes (IGEA Cliniporator VITAE). D MPR of the electrode tips in a native control scan demonstrates spacing of the electrode-pairings between 2.1 and 3 cm
Early Postinterventional Course
Magnetic resonance imaging (MRI) performed 24 h after the procedure confirmed complete devascularization of tumor and satellite. The patient was discharged two days after the procedure in good condition and without complications. Alpha-fetoprotein decreased to 7.55 ng/ml.
Follow-up
Contrast-enhanced MRI 6 weeks and 6 months after ECT showed complete response with no residual or recurrent HCC and without any signs of damage of the central bile ducts or affection of the liver artery (Fig. 3).
Follow-up imaging. A Contrast-enhanced CT scan in arterial phase at the end of intervention shows complete devascularization of the tumor (arrow). B Contrast-enhanced MRI in arterial phase, T1 Vibe 3d fs, 24 h after ablation confirms complete tumor devascularization (arrow). C and D Contrast-enhanced MRI in arterial phase, T1 Vibe 3d fs, 6 weeks (C) and 6 months (D) after ablation show complete response according to mRECIST and shrinking of the ablation defect to 3.1 × 2.8 × 2.8 cm without affection of the liver arteries and with almost complete restoration of the local cholestasis (arrow)
Discussion
During open surgery, ECT has proven to be a potential method for treatment of HCC in 10 patients [10] as well as for treatment of colorectal liver metastases in 16 patients [9]. As of today, very little is known about percutaneous ECT of deep-seated tumors: only one case report on percutaneous ECT of a HCC lesion has been described [12]. In contrary to our case, Djocic et al. performed cone-beam computed tomography guided ECT on a small (18 mm), peripherally located HCC in a patient with multifocal HCC. That patient had already undergone TACE and MWA prior to ECT.
In our case ECT was performed using a primary stereotactic percutaneous approach for treatment of a large (4.7 × 4.5 × 3.5 cm) central unresectable HCC involving the major vessels and the main bile ducts. Multiantenna MWA was not suitable due to the central localization of the tumor with the associated risk of incomplete ablation due to heat sink effect from the hepatic hilar vessels and the risk of thermal damage to the central bile ducts [7]. IRE is more suitable to treat such central liver lesions but this technique is still limited to smaller lesions [7]. However, ECT allows the treatment of larger lesions, because the maximum distance between electrode pairs may be greater and still generate a sufficiently strong electric field (maximum distance of 3.0 cm per electrode pair for IGEA Cliniporator VITAE).
So far, ECT has never been combined with stereotactic navigation. A stereotactic percutaneous approach for ablation of liver lesions results in more precise positioning of the probes and the possibility to predict the exact ablation zones without extension of the procedure time [6]. By using a stereotactic percutaneous approach, we were able to position all probes for ECT precisely with an optimal distance between the electrode-tips ranging from 2.1 to 3.0 cm. Accordingly, the possibility to navigate the percutaneous transhepatic approach in accordance to the individual anatomy and to control it at any time using a stereotactic navigation system and computed tomography may be technically superior to ECT during open surgery or percutaneous techniques using fluoroscopy or cone-beam CT.
The short hospitalization in this case reflects the advantages of minimal-invasive percutaneous ablation therapy: median length of hospital stay after ECT during open surgery for liver metastases was 14 days [9].
Conclusion
Stereotactic percutaneous ECT has the potential to be used as a curative treatment method for HCC with diameters of more than 4 cm, even in close proximity to critical structures like major blood vessels and central bile ducts.
References
Wright AS, Lee FT, Mahvi DM. Hepatic microwave ablation with multiple antennae results in synergistically larger zones of coagulation necrosis. Ann Surg Oncol. 2003;10:275–83. https://doi.org/10.1245/aso.2003.03.045.
Brace CL, Laeseke PF, Sampson LA, Frey TM, van der Weide DW, Lee FT. Microwave ablation with multiple simultaneously powered small-gauge triaxial antennas: results from an in vivo swine liver model. Radiol. 2007;244:151–6. https://doi.org/10.1148/radiol.2441052054.
Zhang T-Q, Huang Z-M, Shen J-X, Chen G-Q, Shen L-J, Ai F, et al. Safety and effectiveness of multi-antenna microwave ablation-oriented combined therapy for large hepatocellular carcinoma. Therap Adv Gastroenterol. 2019;12:1756284819862966. https://doi.org/10.1177/1756284819862966.
Mbalisike EC, Vogl TJ, Zangos S, Eichler K, Balakrishnan P, Paul J. Image-guided microwave thermoablation of hepatic tumours using novel robotic guidance: an early experience. Eur Radiol. 2015;25:454–62. https://doi.org/10.1007/s00330-014-3398-0.
Engstrand J, Toporek G, Harbut P, Jonas E, Nilsson H, Freedman J. Stereotactic CT-guided percutaneous microwave ablation of liver tumors with the use of high-frequency jet ventilation: an accuracy and procedural safety study. AJR Am J Roentgenol. 2017;208:193–200. https://doi.org/10.2214/AJR.15.15803.
Beyer LP, Lürken L, Verloh N, Haimerl M, Michalik K, Schaible J, et al. Stereotactically navigated percutaneous microwave ablation (MWA) compared to conventional MWA: a matched pair analysis. Int J Comput Assist Radiol Surg. 2018;13:1991–7. https://doi.org/10.1007/s11548-018-1778-7.
Silk MT, Wimmer T, Lee KS, Srimathveeravalli G, Brown KT, Kingham PT, et al. Percutaneous ablation of peribiliary tumors with irreversible electroporation. J Vasc Interv Radiol. 2014;25:112–8. https://doi.org/10.1016/j.jvir.2013.10.012.
Edhemovic I, Gadzijev EM, Brecelj E, Miklavcic D, Kos B, Zupanic A, et al. Electrochemotherapy: a new technological approach in treatment of metastases in the liver. Technol Cancer Res Treat. 2011;10:475–85. https://doi.org/10.7785/tcrt.2012.500224.
Edhemovic I, Brecelj E, Gasljevic G, Marolt Music M, Gorjup V, Mali B, et al. Intraoperative electrochemotherapy of colorectal liver metastases. J Surg Oncol. 2014;110:320–7. https://doi.org/10.1002/jso.23625.
Djokic M, Cemazar M, Popovic P, Kos B, Dezman R, Bosnjak M, et al. Electrochemotherapy as treatment option for hepatocellular carcinoma, a prospective pilot study. Eur J Surg Oncol. 2018;44:651–7. https://doi.org/10.1016/j.ejso.2018.01.090.
Mir LM, Gehl J, Sersa G, Collins CG, Garbay J-R, Billard V, et al. Standard operating procedures of the electrochemotherapy: Instructions for the use of bleomycin or cisplatin administered either systemically or locally and electric pulses delivered by the CliniporatorTM by means of invasive or non-invasive electrodes. Eur J Cancer Suppl. 2006;4:14–25. https://doi.org/10.1016/j.ejcsup.2006.08.003.
Djokic M, Dezman R, Cemazar M, Stabuc M, Petric M, Smid LM, et al. Percutaneous image guided electrochemotherapy of hepatocellular carcinoma: technological advancement. Radiol Oncol. 2020;54:347–52. https://doi.org/10.2478/raon-2020-0038.
Funding
Open Access funding enabled and organized by Projekt DEAL. This study was not supported by any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
For this type of study (retrospective study) formal consent is not required and was waived by the IRB of the University of Regensburg.
Informed Consent
This study has obtained IRB approval (University of Regensburg) and the need for informed consent was waived.
Consent for Publication
For this type of study consent for publication is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luerken, L., Doppler, M., Brunner, S.M. et al. Stereotactic Percutaneous Electrochemotherapy as Primary Approach for Unresectable Large HCC at the Hepatic Hilum. Cardiovasc Intervent Radiol 44, 1462–1466 (2021). https://doi.org/10.1007/s00270-021-02841-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00270-021-02841-1