Abstract
Purpose
Skin and soft tissue aging has been an important topic of discussion among plastic surgeons and their patients. While botulinum toxin, facial fillers, chemical peels, and surgical lifts preside as the mainstay of treatment to restore appearance of youth, emergent technologies, such as CRISPR-Cas9, proteostasis, flap biology, and stem cell therapies, have gained traction in addressing the aging process of skin and soft tissue. Several studies have introduced these advancements, but it remains unclear how safe and effective these therapeutics are in facial rejuvenation, and how they may fit in the existent treatment workflow for soft tissue aging.
Materials/Methods
A systematic literature review was conducted to identify and assess therapeutics utilized in addressing skin and soft tissue aging. Variables collected included year of publication, journal, article title, organization of study, patient sample, treatment modality, associated outcomes. In addition, we performed a market analysis of companies involved in promoting technologies and therapeutics within this space. PitchBook (Seattle, WA), a public market database, was utilized to classify companies, and record the amount of venture capital funding allocated to these categories.
Results
Initial review yielded four hundred and two papers. Of these, thirty-five were extracted after applying inclusion and exclusion criteria. Though previous literature regards CRISPR-Cas9 technology as the most favorable anti-aging innovation, after reviewing the current literature, stem cell therapies utilizing recipient chimerism appeared to be the superior skin anti-aging technique when accounting for possible disadvantages of various techniques. The psychosocial and cosmetic outcomes from using cell therapy to modulate allograft survival and tolerance may confer more long-term proposed benefits than the technologies in CRISPR-Cas9, flap biology innovations, and autologous platelet-rich plasma use. Market analysis yielded a total of 87 companies, which promoted innovations in technology, biotechnology, biopharmaceuticals, cell-based therapies, and genetic therapy.
Conclusion
This review provides physicians and patients with relevant, usable information regarding how therapeutics can impact treatment regimen for facial aesthetics and skin rejuvenation. Furthermore, the goal of this research is to elucidate the varying therapeutics to restore appearance of youth, present associated outcomes, and in doing so, present plastic surgeons and their colleagues with greater insight on the role of these therapeutics and technologies in clinical practice. Future studies can further assess the safety and efficacy of these innovations and discuss how these may fit within surgical plans among patients seeking rejuvenation procedures.
Level of Evidence III
This journal requires that authors assign a level of evidence to each article. For a full description of these Evidence-Based Medicine ratings, please refer to the Table of Contents or the online Instructions to Authors www.springer.com/00266.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Skin and soft tissue aging has presided as an important topic of discussion among plastic surgeons and their patients. Cutaneous aging has been cited to occur via intrinsic and extrinsic processes. Moreover, intrinsic aging occurs due to decreased proliferation of cells including keratinocytes, fibroblasts, and melanocytes, via a process termed cellular senescence [1]. During such processes, degeneration of fibrous extracellular matrix components such as elastin, fibrillin, and collagen, and decreased in vascularity occur. Collagen fibrils become disorganized, fragmented, and reduced in number and diameter. Extrinsic aging is primarily driven by exposure to ultra-violet (UV) radiation, which impairs differentiation process of epidermal keratinocytes and promotes accumulation of abnormal elastic tissue within the dermis [2,3,4]. Moreover, multiple molecular mechanisms have been proposed to mitigate the processes of skin and soft tissue aging; these include genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, and altered intercellular communication [1,2,3,4,5,6].
Plastic surgery as a specialty has incorporated both noninvasive interventions, such as botulinum toxin, soft tissue fillers, bio-stimulants, chemical peels, platelet-rich plasma (PRP), and lasers, as well as surgical procedures, such as fat grafting, threading lifts, face and neck lift, blepharoplasty, and rhinoplasty, in efforts to promote a more youthful appearance among their patients. However, the application of newer technologies, such as genetic therapies, flap biology, and stem cell-based treatments, has gained traction among venture capitalists; the longevity economy has fielded $7.6 billion in hopes to promote healthspan and restoration of youth within the ever-growing aging population. Moreover, these advancements are yet to be integrated within the practice of most plastic surgeons and dermatologists. In addition, it remains unclear how safe and effective these therapeutics are in skin rejuvenation, and how they may fit in the existent treatment workflow for skin and soft tissue aging.
The purpose of this study was multi-fold: 1) to conduct a systematic literature review of therapeutics and technologies used to address skin and soft tissue aging processes; 2) to perform a market and trend analysis of venture-backed companies operating in the aforementioned indications; 3) to understand the safety and efficacy profiles of these respective innovations; 4) to discuss the implications for plastic surgeons when considering to implement them into their clinical practice. We hypothesized that cosmetic and aesthetic supplements would be the most common offering marketed by companies, given lower production costs and thus, lower barrier to entry, while innovations in stem-cell, proteostasis, and genetic therapy would be fewer yet the most lucrative in acquiring funding.
Methods
A systematic literature review was conducted with National Center for Biotechnology Information, Medline, ASPS Tracking Operations and Outcomes for Plastic Surgeons, Cochrane, Web of Science, Scopus, and PubMed databases to identify articles that discussed therapeutics to address skin and soft tissue aging. A combination of the following keywords was utilized to conduct the literature review: “anti-aging,” “facial aesthetics,” “soft tissue aging,” “facial ageing,” “aesthetics,” “innovation,” and “skin ageing.” Variables collected included year of publication, journal, article title, organization of study, patient sample, and treatment modality, and level of evidence. Articles were excluded if data were published before 2000, yielded non-significant findings, or discussed already established treatments to mitigate skin and soft tissue aging processes (Table 1). Articles were then categorized by the level of evidence as set forth by the American Society of Plastic Surgeons (ASPS). Any conflicts were resolved through discussion and full text review among D.G. and S.K. Of note, the search was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. No funding was required to conduct this review of the literature.
In addition, a formal analysis of Pitchbook (Seattle, WA), a platform used to provide private market data, was conducted in order to identify companies operating within the fields of aesthetic aging and longevity. Companies were isolated using the following search terms: “Longevity Biotech,” “VC-backed,” “Accelerator/Incubator-backed,” “Anti-Aging,” “Soft Tissue Aging,” “Aesthetic Surgery,” “Facial Aesthetics,” “Anti-Aging Skin,” “Autologous,” and “Longevity.” Companies were excluded from analysis if 1) not operating within the healthcare or life sciences sector, or 2) not addressing issues directly within skin and soft tissue aging processes. Companies were then classified into the following categories: personal products, biologic, platform technology, genetic therapy, and cell-based therapy. Company descriptions and aggregate funding amount to date were collected via respective company websites, Pitchbook, as well as Crunchbase (San Francisco, CA) [7].
Results
Literature Review
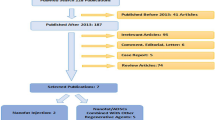
Initial review yielded a total of four hundred and two articles. After application of inclusion and exclusion criteria, a total of thirty-five studies were analyzed for the purposes of this publication. A flow-chart demonstrating the isolation of these studies is detailed in Fig. 1.
Genetic-based technologies, namely via maintaining cellular proteostasis, manipulating the process of alternative splicing for specific genes, and leveraging adenovirus vectors, were discussed in three studies. The majority of cited studies discussed stem cell-based innovations, specifically within adipose-derived mesenchymal stem cells (ADSCs), in combination with hyaluronic acid, fat grafting, PRP, and fibrin derived compounds, were additionally discussed as viable methods to address the sequela of photoaging and wrinkles in the aging process. Flap biology via genetic modification was also discussed as an option to mitigate the natural aging process within vascular composite allotransplantation, for example. Furthermore, oral collagen and hyaluronic acid (HA) supplements were also described in numerous studies. A summary of analyzed articles can be viewed in Table 1.
Company-Level Analysis
Initial search yielded a total of six hundred and seven companies, among which three hundred and fifty-seven did not operate within healthcare and the life sciences. The remaining two hundred and fifty companies were included in the analysis; these companies were classified into one of five categories, including platform technology, personal products, biologic, cell-based therapy, and genetic therapy. Our analysis demonstrated that the majority, 41% of companies hailed from the biologic space, followed by 24% from the genetic therapy space, 20% from personal products, 8% from cell-based therapies, and 7% from platform technologies. However, in terms of aggregated funding amount, personal products have gained the highest amount of funding, with an estimated $378,675,000 funding in the personal products space, closely followed by the biologic space with $347,531,000. The number of companies, as well as aggregated funding amount, for each category can be viewed in Fig. 2.
Discussion
This study presents various mechanisms to address skin and soft tissue aging via literature and industry-level analyses. The literature review was largely comprised of articles that built upon already well-established methods for aesthetic rejuvenation, such as hyaluronic acid fillers, PRP, fat grafting, while the company-level analysis was dominated by personal products, such as supplements and skin care remedies.
Cell-Based Therapies
The vast majority of articles included in the literature review alluded to the fundamental role that adipose-derived stem cells (ADSCs) have in tissue regeneration and remodeling processes. Moreover, in an analysis by Gentile et al, it was found that ADSCs directly activate dermal fibroblast proliferation, an antioxidant effect, and matrix metalloproteinases (MMPs) reduction [8, 9]. In combination with mainstay methods for facial rejuvenation, such as fat grafting, PRP, and soft tissue fillers, ADSCs have a multiplicative effect in refining skin texture and quality. Our market analysis drew attention to several companies building cell-based technologies, via exosomes as well as stem cells derived from adults and plants, to address skin and soft tissue aging [7, 10]. Future studies should further explore how ADSCs may be incorporated into the practice of plastic surgeons from a financial and logistical perspective [11].
Autologous Methods
Our review of the literature also revealed multiple methods for leveraging use of autologous tissue to address skin and soft tissue aging. Moreover, several studies discuss the important role of platelet-rich plasma (PRP) within skin rejuvenation, through mechanisms of angiogenesis and mitogenesis [11,12,13,14,15,16]. Others have revealed opportunity for other autologous growth factors, fibroblasts, as well as combinations of dermis, fat, and fascia in the creation of various derivatives for facial rejuvenation [17,18,19,20,21,22,23]. Future investigations may explore long-term clinical outcomes, including safety and efficacy, associated with these respective therapeutics and additionally discuss the logistics entailed in utilizing these innovations in real clinical practice with patients.
Genetic Therapies
Our investigation highlights the emergent role of genetic therapies, through multiple modalities, including alternative splicing, viral vectors, and epigenetics. Bramwell et al. leveraged the process of alternative splicing to regulate responsible for extra-cellular matrix remodeling, restoration of collagen and elastin frameworks, and reversal of adipose tissue atrophy [24]. In doing so, there exists an opportunity for moderators of alternative splicing to be incorporated into injectable fillers or topical creams for patients. Moreover, Giatstidis et al. proposed the notion of “mature” gene therapy, wherein adenovirus vectors can be utilized to manipulate epidermal growth factor (EGF), fibroblast growth factor (FGF), insulin-like growth factor (IGF), keratinocyte growth factor (KFG), platelet-derived growth factor (PDGF), transforming growth factor-β (TGF-β), and vascular endothelial growth factor (VEGF) [25]. Sabath et al. especially fixated on the role of genetic therapies preserving the process of cellular proteostasis [26,27,28,29,30,31]. Finally, companies, such as Ponce de Leon, have proposed that epigenetic modification (specifically DNA methylation) may be used to reverse aging processes [32,33,34,35,36,37].
Biologics
Biologics, defined as small molecules targeting cellular pathways, have an established role in the anti-aging aesthetics industry. Furthermore, in the Pitchbook analysis, many companies, such as Dorian Therapeutics developed small molecules (senoblockers), selectively inhibit processes leading up to senescence, a process defined by cellular aging to the point of non-division [32,33,34,35,36,37]. Other companies developed molecules to directly target collagen scaffolds, ECM, and other cellular processes such as autophagy [38,39,40,41,42,43,44]. It remains unclear how these therapeutics compare to existent modalities for skin rejuvenation. Moreover, prior studies have yet to find definitive data as to where these therapeutics may preside in the treatment regimen proposed by plastic surgeons, dermatologists, and their colleagues.
Personal Products and Natural Remedies
Natural ingredients, in the formulation of topical creams, lotions and preparations, have been used for anti-aging effects. Fowler et al describe ingredients, such as colloidal oatmeal, aloe vera, green tea, niacinamide and feverfew confer anti-inflammatory properties [45]. For hyperpigmentation and antioxidative capabilities, licorice, green tea, arbutin, soy, acai berry, turmeric and pomegranate have been described as efficacious. In our market analysis, multiple supplements that comprised of carotenoids, shark cartilage, plant derivatives have been taken by consumers for anti-aging effects [46,47,48,49,50,51]. In addition, collagen and hyaluronic acid (HA) supplements have conferred improvements in facial wrinkling and texture, as well as increases in skin elasticity and hydration [52,53,54,55,56,57]. Beyond aesthetic appearance, some supplements, namely those with nicotinamide, have been proposed to decrease risks of developing skin cancer [58]. Additional research is needed to elucidate and confirm the benefits of these ingredients in the management of skin and soft tissue aging, and possible skin cancer prevention.
Flap Biology and Tissue Engineering
In a study done by Ghali et al, it was established that creating genetically modified flaps to enhance the recipient site may preside as a new anti-aging therapy and mechanism [59]. Researchers further state that CRISPR/Cas-9 technology would further facilitate concept by making this process more cost-effective, efficient, and multiplexed [60,61,62,63]. Moreover, this technology could be specifically applied to patients undergoing microsurgical reconstruction (head and neck, breast), and those undergoing vascular composite allotransplantation [10, 63,64,65]. The application of this innovation would ultimately refine aesthetic results in a way that promotes healthy skin quality and texture over time [66,67,68,69]. The precise application of genetically modified flaps allows engineering in anti-aging to be personalized and allow for limited scarring, proven time and time again to be of vital importance to patients [70,71,72,73,74]. Moreover, studies such as Dempsey et al have demonstrated genetically modified free flaps can be used without eliciting toxic systemic effects, through delivering IL-12 directly into the local environment of a skin tumor and suppress its growth [74]. The remarkable strides in research in flap biology and tissue engineering are currently being heavily applied to tumor suppression, but numerous studies have proved these effects would translate seamlessly to anti-aging applications [75,76,77,78,79]. Nonetheless, genetic modification of flaps may pose risks of increased rejection among patients. Future studies should explore these possibilities in pre-clinical studies to affirm the safety and efficacy of such ideas.
Limitations
There are several limitations of this study that warrant consideration. First, the process of isolating and classifying companies within the Pitchbook database may be subject to inherent selection bias. In order to minimize such bias, the authors implemented standardized data collection forms, and additionally, consulted with one another in the event of a conflicting opinion. Second, our analysis was limited to companies within exclusively the Pitchbook database; thus, does not incorporate technologies from companies currently amid formation and raising capital. Third, while the investigation was able to elucidate the various methods toward addressing skin and soft tissue aging, measurement and comparison of outcomes (such as skin elasticity, dermal collagen density) were not incorporated within the analysis. Future studies may directly compare objective metrics among the cited therapeutics and technologies for the aging process. Despite these limitations, this study is the first to present a multi-level analysis, from academic and industry perspectives, to further explore the anti-aging landscape of innovation.
Conclusions
Our study sought to explore the current modalities to address skin and soft tissue aging via literature and industry-level analyses. We concluded that there are multiple promising technologies and therapeutics within the anti-aging aesthetic industry, with innovations in genetic therapy, cell-based therapy, platform technologies, biologics, and personal products. The literature review was largely comprised of articles and journal pieces that built upon already well-established methods for aesthetic rejuvenation, such as hyaluronic acid fillers and fat grafting. In contrast, the company-level analysis was dominated by personal products, such as supplements and skin care remedies. In conducting this investigation, we present plastic surgeons and their colleagues with greater insight on the role of these therapeutics and technologies in clinical practice. Moreover, we provide investors relevant data on the growing industry of skin and soft tissue aging, detailing the most prominent categories of research. Future studies can further assess the safety and efficacy of these innovations and discuss how these may fit within surgical plans among patients seeking rejuvenation procedures.
References
Zang S, Duan E (2018) Fighting against skin aging. Cell Transplant 27(5):729–738
Gerth D (2015) Structural and volumetric changes in the ageing face. Facial Plast Surg 31(1):3–9
Elson ML (1995) Soft tissue augmentation a review. Dermatol Surg. 21(6):491–500
Zins JE, Moreira-Gonzalez A (2006) Cosmetic procedures for the ageing face. Clin Geriatr Med 22(3):709–728
Farkas JP, Pessa JE, Hubbard B, Rohrich RJ (2013) The science and theory behind facial aging. Plast Reconstr Surg Glob Open 7(1):e8–e15
Roh DS, Panayi A, Bhasin S, Orgill D, Sinha I (2019) Implications of aging in plastic surgery. Plast Reconstr Surg Glob Open 7(1):e2085
Crunchbase. https://www.crunchbase.com/. Last updated 2022
Gentile R (2019) Renuvion/J-plasma for subdermal skin tightening facial contouring and skin rejuvenation of the face and neck. Fac Plast Surg Clin North Amer. 27(3):273–290
Gentile P, Garcovich S (2021) Adipose-derived mesenchymal stem cells (AD-MSCs) against ultraviolet (UV) radiation effects and the skin photoaging. Biomedicines. 9(5):532
Schweizer R, Gorantla VS, Plock JA (2015) Premise and promise of mesenchymal stem cell-based therapies in clinical vascularized composite allotransplantation. Curr Opin Organ Transplant 20:608614
Matsumura H, Mohri Y, Binh NT et al (2016) Hair follicle aging is driven by transepidermal elimination of stem cells via COL17A1 proteolysis. Science 351:4395
Buzalaf MAR, Levy FM (2022) Autologous platelet concentrates for facial rejuvenation. J Appl Oral Sci 30:e20220020
Elghblawi E (2018) Platelet-rich plasma, the ultimate secret for youthful skin elixir and hair growth triggering. J Cosmet Dermatol 17(3):423–430
Abuaf OK, Yildiz H, Baloglu H (2016) Histologic evidence of new collagen formulation using platelet rich plasma in skin rejuvenation: a prospective controlled clinical study. Ann Dermatol 28(6):718–724
Rigotti G, Charles-de-Sá L, Gontijo-de-Amorim NF et al (2016) Expanded stem cells, stromal-vascular fraction, and platelet-rich plasma enriched fat: comparing results of different facial rejuvenation: approaches in a clinical trial. Aesthet Surg J 36(3):261–270
Choi YJ, Kim HS, Min JH et al (2017) A clinical study on the usefulness of autologous plasma filler in the treatment of nasolabial fold wrinkles. J Cosmet Laser Ther 19(3):174–180
Smith SR, Munavalli G, Weiss R (2012) A multicenter, double-blind, placebo-controlled trial of autologous fibroblast therapy for the treatment of nasolabial fold wrinkles. Dermatol Surg 38(7):1234–1243
Weiss RA (2013) Autologous cell therapy: will it replace dermal fillers? Facial Plast Surg Clin North Am 21(2):299–304
Chandrashekar BS, Sarangi K, Mastim MA et al (2022) A prospective multicenter study to evaluate the safety and efficacy of the topical application of MYOWNN, an autologous growth factor concentrate (AGFC) Serum, in anti-aging. Cureus. 14(5):e25190
Rossi M, Roda B, Zia S (2020) Characterization of the tissue and stromal cell components of micro-superficial enhanced fluid fat injection (micro-SEFFI) for facial aging. Aesthet Surg J 40(6):679–690
Erol OO (2000) Facial autologous soft-tissue contouring by adjunction of tissue cocktail injection (micrograft and minigraft mixture of dermis, fascia, and fat). Plast Reconstr Surg 106(6):1375
Guyuron B, Son JH (2019) String fat/dermis graft for correction of wrinkles and scars. Plast Reconstr Surg 144(1):93–96
Charles-de-Sá L, Gontijo-de-Amorim NF, Takiya CM et al (2015) Antiaging treatment of the facial skin by fat graft and adipose-derived stem cells. Plast Reconstr Surg 135(4):999–1009
Bramwell LR, Harries LW (2021) Targeting alternative splicing for reversal of cellular senescence in the context of aesthetic aging. Plast Reconstr Surg 147(1S–2):25S-32S. https://doi.org/10.1097/PRS.0000000000007618. (PMID: 33347071)
Giatsidis G, Dalla Venezia E, Bassetto F (2013) The role of gene therapy in regenerative surgery: updated insights. Plast Reconstr Surg 131:14251435
Sabath N, Levy-Adam F, Younis A, Rozales K, Meller A, Hadar S, Soueid-Baumgarten S, Shalgi R (2020) Cellular proteostasis decline in human senescence. Proc Natl Acad Sci USA 117:31902–31913
Höhn A, Weber D, Jung T, Ott C, Hugo M, Kochlik B, Kehm R, König J, Grune T, Castro JP (2017) Happily (n)ever after: aging in the context of oxidative stress, proteostasis loss and cellular senescence. Redox Biol 11:482–501
Korovila I, Hugo M, Castro JP, Weber D, Höhn A, Grune T, Jung T (2017) Proteostasis, oxidative stress and aging. Redox Biol 13:550–567
Hipp MS, Park S-H, Hartl FU (2014) Proteostasis impairment in protein-misfolding and -aggregation diseases. Trends Cell Biol 24:506–514
Kaushik S, Cuervo AM (2015) Proteostasis and aging. Nat Med 21:1406–1415
Labbadia J, Morimoto RI (2015) The biology of proteostasis in aging and disease. Annu Rev Biochem 84:435–464
Li H, Yang Y, Hong W et al (2020) Applications of genome editing technology in the targeted therapy of human diseases: mechanisms, advances and prospects. Sig Transduct Target Ther 5:1
Csekes E, Račková L (2021) Skin aging, cellular senescence and natural polyphenols. Int J Mol Sci 22(23):12641
Roh DS, Li Edward B-H, Liao EC (2018) CRISPR Craft: DNA editing the reconstructive ladder. Plast Reconstr Surg 142(5):1355–1364. https://doi.org/10.1097/PRS.0000000000004863
Ghali S, Dempsey MP, Jones DM, Grogan RH, Butler PE, Gurtner GC (2008) Plastic surgical delivery systems for targeted gene therapy. Ann Plast Surg 60:323332
Roman S, Lindeman R, O’Toole G, Poole MD (2005) Gene therapy in plastic and reconstructive surgery. Curr Gene Ther 5:8199
Shenaq SM, Rabinovsky ED (1996) Gene therapy for plastic and reconstructive surgery. Clin Plast Surg 23:157171
Rhee SM, You HJ, Han SK (2014) Injectable tissue-engineered soft tissue for tissue augmentation. J Korean Med Sci 29(Suppl 3):S170-175
Altman AM, Khalek FJA, Seidensticker M (2010) Human tissue-resident stem cells combined with hyaluronic acid gel provide fibrovascular-integrated soft-tissue augmentation in a murine photoaged skin model. Plast Reconstr Surg 125(1):63–73
Wei H, Gu SX, Linang YD (2017) Nanofat-derived stem cells with platelet-rich fibrin improve facial contour remodeling and skin rejuvenation after autologous structural fat transplantation. Oncotarget 8(40):68542–68556
Lequeux C, Oni G, Wong C et al (2012) Subcutaneous fat tissue engineering using autologous adipose-derived stem cells seeded onto a collagen scaffold. Plast Reconstr Surg 130(6):1208–1217
Ozer K, Colak O (2019) Micro-autologous fat transplantation combined with platelet-rich plasma for facial filling and regeneration: a clinical perspective in the shadow of evidence-based medicine. J Cranio Surg. 30(3):672–677
Cavallini M, De Luca C, Prussia G et al (2022) PN-HPT ® (polynucleotides highly purified technology) in facial middle third rejuvenation. Explor Potential. J Cosmet Dermatol. 21(2):615–624
Solakoglu S, Tiryaki T, Ciloglu SE (2008) The effect of cultured autologous fibroblasts on longevity of cross-linked hyaluronic acid used as a filler. Aesthet Surg J 28(4):412–416
Fowler JF Jr, Woolery-Lloyd H, Waldorf H, Saini R (2010) Innovations in natural ingredients and their use in skin care. J Drugs Dermatol 9(6 Suppl):S72-81 (PMID: 20626172)
Bowe WP, Pugliese S (2014) Cosmetic benefits of natural ingredients. J Drugs Dermatol 13(9):1021–1025 (PMID: 25226001)
Baumann L, Woolery-Lloyd H, Friedman A (2009) “Natural” ingredients in cosmetic dermatology. J Drugs Dermatol 8(6 Suppl):s5-9 (PMID: 19562883)
Wu J (2006) Treatment of rosacea with herbal ingredients. J Drugs Dermatol 5(1):29–32 (PMID: 16468289)
Chanchal D, Swarnlata S (2008) Novel approaches in herbal cosmetics. J Cosmet Dermatol 7(2):89–95. https://doi.org/10.1111/j.1473-2165.2008.00369.x. (PMID: 18482010)
Baumann LS (2007) Less-known botanical cosmeceuticals. Dermatol Ther 20(5):330–342. https://doi.org/10.1111/j.1529-8019.2007.00147.x. (PMID: 18045358)
Jhawar N, Wang JV, Saedi N (2020) Oral collagen supplementation for skin aging: a fad or the future. J Cosmet Dermatol 19(2):910–912
De Miranda RB, Weimer P, Rossi RC (2021) Effects of hydrolyzed collagen supplementation on skin aging: a systematic review and meta-analysis. Int J Dermatol 60(12):1449–1461
Asserin J, Lati E, Shioya T et al (2015) The effect of oral collagen peptide supplementation on skin moisture and the dermal collagen network: evidence from an ex vivo model and randomized, placebo-controlled clinical trials. J Cosmet Dermatol 14(4):291–301
Inoue N, Sugihara F, Wang X (2016) Ingestion of bioactive collagen hydrolysates enhance facial skin moisture and elasticity and reduce facial ageing signs in a randomised double-blind placebo-controlled clinical study. J Sci Food Agric 96(12):4077–4081
Hsu TF, Su ZR, Hsieh YH et al (2021) Oral hyaluronan relieves wrinkles and improves dry Skin: a 12-week double-blinded, placebo-controlled study. Nutrients 13(7):2220
Gollner I, Voss W, von Hehn U et al (2017) Ingestion of an oral hyaluronan solution improves skin hydration, wrinkle reduction, elasticity, and skin roughness: results of a clinical study. J Evid Based Complement Altern Med 22(4):816–823
Schwartz SR, Park J (2012) Ingestion of BioCell Collagen(®), a novel hydrolyzed chicken sternal cartilage extract; enhanced blood microcirculation and reduced facial aging signs. Clin Interv Aging 7:267–273
Chen A, Martin AJ, Choy B (2015) A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. New Eng J Med 373:1618–1626
Ghali S, Bhatt KA, Dempsey MP et al (2009) Treating chronic wound infections with genetically modified free flaps. Plast Reconstr Surg 123:11571168
Guitart JR Jr, Johnson JL, Chien WW (2016) Research techniques made simple: the application of CRISPR-Cas9 and genome editing in investigative dermatology. J Invest Dermatol 136:e87–e93
Cyranoski D (2016) CRISPR gene-editing tested in a person for the first time. Nature 539:479
Zhang XH, Tee LY, Wang XG, Huang QS, Yang SH (2015) Off-target effects in CRISPR/Cas9-mediated genome engineering. Mol Ther Nucleic Acids 4:e264. https://doi.org/10.1038/mtna.2015.37
Niu D, Wei HJ, Lin L et al (2017) Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9. Science 357:13031307
Cetrulo CL Jr, Ng ZY, Winograd JM, Eberlin KR (2017) The advent of vascularized composite allotransplantation. Clin Plast Surg 44:425429
Cowan PJ, Tector AJ (2017) The resurgence of xenotransplantation. Am J Transplant 17:25312536
Zimmermann R, Jakubietz R, Jakubietz M, Strasser E, Schlegel A, Wiltfang J et al (2001) Different preparation methods to obtain platelet components as a source of growth factors for local application. Transfusion 41:1217–1224. https://doi.org/10.1046/j.1537-2995.2001.41101217.x
Marx RE (2004) Platelet-rich plasma: evidence to support its use. J Oral Maxillofac Surg 62:489–496. https://doi.org/10.1016/j.joms.2003.12.003
Freymiller EG (2004) Platelet-rich plasma: evidence to support its use. J Oral Maxillofac Surg 62:1046–1047. https://doi.org/10.1016/j.joms.2004.05.205
Wrotniak M, Bielecki T, Gaździk TS (2007) Current opinion about using the platelet-rich gel in orthopaedics and trauma surgery. Ortop Traumatol Rehabil 9:227–238
Kim DH, Je YJ, Kim CD, Lee YH, Seo YJ, Lee JH et al (2011) Can platelet-rich plasma be used for skin rejuvenation? Evaluation of effects of platelet-rich plasma on human dermal fibroblast. Ann Dermatol 23:424–431
Krasna M, Domanovic D, Tomsic A, Svajger U, Jeras M (2007) Platelet gel stimulates proliferation of human dermal fibroblasts in vitro. Acta Dermatovenerol Alp Panonica Adriat 16:105–110
Lucarelli E, Beccheroni A, Donati D, Sangiorgi L, Cenacchi A, Del Vento AM et al (2003) Platelet-derived growth factors enhance proliferation of human stromal stem cells. Biomaterials 24:3095–3100. https://doi.org/10.1016/S0142-9612(03)00114-5
Kanno T, Takahashi T, Tsujisawa T, Ariyoshi W, Nishihara T (2005) Platelet-rich plasma enhances human osteoblast-like cell proliferation and differentiation. J Oral Maxillofac Surg 63:362–369. https://doi.org/10.1016/j.joms.2004.07.016
Dempsey MP, Hamou C, Michaels J IV et al (2008) Using genetically modified microvascular free flaps to deliver local cancer immunotherapy with minimal systemic toxicity. Plast Reconstr Surg 121:15411553
Boismal F, Serror K, Dobos G, Zuelgaray E, Bensussan A, Michel L (2020) Vieillissement cutané - physiopathologie et thérapies innovantes [Skin aging: pathophysiology and innovative therapies]. Med Sci (Paris) 36(12):1163–1172. https://doi.org/10.1051/medsci/2020232. (Epub 2020 Dec 9. PMID: 33296633)
Menicacci B, Cipriani C, Margheri F, Mocali A, Giovannelli L (2017) Modulation of the senescence-associated inflammatory phenotype in human fibroblasts by olive phenols. Int J Mol Sci 18:2275
Seth R, Khan AA, Pencavel TD et al (2015) Adenovirally delivered enzyme prodrug therapy with herpes simplex virus-thymidine kinase in composite tissue free flaps shows therapeutic efficacy in rat models of glioma. Plast Reconstr Surg 135:475487
Michaels J IV, Dobryansky M, Galiano RD et al (2004) Ex vivo transduction of microvascular free flaps for localized peptide delivery. Ann Plast Surg 52:581584
Lampert FM, Momeni A, Filev F et al (2015) Utilization of a genetically modified muscle flap for local BMP-2 production and its effects on bone healing: a histomorphometric and radiological study in a rat model. J Orthop Surg Res 10:55
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest to disclose.
Human or Animal Rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
For this type of study, informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Khetpal, S., Ghosh, D. & Roostaeian, J. Innovations in Skin and Soft Tissue Aging—A Systematic Literature Review and Market Analysis of Therapeutics and Associated Outcomes. Aesth Plast Surg 47, 1609–1622 (2023). https://doi.org/10.1007/s00266-023-03322-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00266-023-03322-1