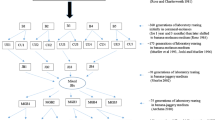
Abstract
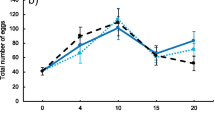
Natural selection favours animals that forage more efficiently, such as those finding food faster, choosing better patches or increasing their rate of energy gain. Indeed, foraging behaviour has an effect on fitness, with better foragers producing more offspring. However, the evolution of foraging behaviour and its consequences on reproductive success has usually been tested in one generation only and the quantitative effects on fitness are not always measured. Here we artificially selected Drosophila melanogaster adults for their ability to find food quickly in a simple maze and measured the effect of this selection on their fitness. We tested two treatments, with four selected and two control independent replicates: i) selected flies, which were the fastest to find a food source inside a maze, and ii) control flies, which were randomly selected regarding their location in the maze. Fitness was calculated measuring fecundity and offspring survivorship. Our results show that after five generations, flies selected for better foraging found food two times faster than control flies. Additionally, selected flies had higher fitness than control flies, mainly because of greater fecundity in females during the first generation. These results show that foraging behaviour has a strong response to selection in few generations and that selection on foraging ability can have a correlated positive effect on fitness, nevertheless, this response is diluted over generations possibly due to the negative effect associated with the selection process.
Significant statement
Natural selection favours animals that forage more efficiently, such as those finding food faster. However, most of the literature measuring the relationship between foraging efficiency and fitness usually only measures the effects in the first generation. In an experiment, we artificially selected Drosophila adult flies that found food faster in a simple maze and we also measured the fecundity and offspring survivorships in selected and non-selected flies. After five generations, the selected flies found food 2.46 times faster than their F1 (founding population) and the control flies that were not selected. Fecundity was higher for selected flies only in the first generation, correlation that was lost as the generations passed. These results show that foraging behaviour has a strong response to selection in a few generations and that the possible correlated positive effects on fitness are diluted over generations.




Similar content being viewed by others
Data availability
Data related with this manuscript is available on https://data.mendeley.com/datasets/z2yp9pjcjm/draft?a=3c4b1330-e899-47dc-b579-9c287ffb236d.
References
Aguila JR, Suszko J, Gibbs AG, Hoshizaki DK (2007) The role of larval fat cells in adult Drosophila melanogaster. J Exp Biol 210:956–963. https://doi.org/10.1242/jeb.001586
Allemand R, Boulétreau-Merle J (1989) Correlated responses in lines of Drosophila melanogaster selected for different oviposition behaviours. Exp 4511(45):1147–1150. https://doi.org/10.1007/BF01950184
Allen AM, Anreiter I, Vesterberg A et al (2018) Pleiotropy of the <1>Drosophila melanogaster</i> foraging gene on larval feeding-related traits. J Neurogenet 32:256–266. https://doi.org/10.1080/01677063.2018.1500572
Anreiter I, Kramer JM, Sokolowski MB (2017) Epigenetic mechanisms modulate differences in Drosophila foraging behavior. Proc Natl Acad Sci USA 114:12518–12523. https://doi.org/10.1073/PNAS.1710770114/SUPPL_FILE/PNAS.201710770SI.PDF
Baker MC, Belcher CS, Deutsch LC et al (1981) Foraging success in junco flocks and the effects of social hierarchy. Anim Behav 29:137–142. https://doi.org/10.1016/S0003-3472(81)80160-1
Barrett SCH, Charlesworth D (1991) Effects of a change in the level of inbreeding on the genetic load. Nature 352:522–524. https://doi.org/10.1038/352522a0
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48
Blanckenhorn WV (1991) Fitness consequences of foraging success in water striders (Gerris remigis; Heteroptera: Gerridae). Behav Ecol 2:46–55. https://doi.org/10.1093/beheco/2.1.46
Boggs CL, Freeman KD (2005) Larval food limitation in butterflies: Effects on adult resource allocation and fitness. Oecologia 144:353–361. https://doi.org/10.1007/s00442-005-0076-6
Bonnet T, Morrissey MB, de Villemereuil P et al (2022) Genetic variance in fitness indicates rapid contemporary adaptive evolution in wild animals. Science 376:1012–1016. https://doi.org/10.1126/SCIENCE.ABK0853/SUPPL_FILE/SCIENCE.ABK0853_CODE_S1.ZIP
Brown EB, Patterson C, Pancoast R, Rollmann SM (2017) Artificial selection for odor-guided behavior in Drosophila reveals changes in food consumption. BMC Genomics 18:1–13. https://doi.org/10.1186/S12864-017-4233-1/FIGURES/4
Burnet B, Sewell D, Bos M (1977) Genetic analysis of larval feeding behaviour in Drosophila melanogaster: II. Growth relations and competition between selected lines. Genet Res (Camb) 30:149–161. https://doi.org/10.1017/S0016672300017559
Chess KF, Ringo JM (1985) Oviposition site selection by Drosophila melanogaster and Drosophila simulans. Evolution (N Y) 39:869–877. https://doi.org/10.1111/j.1558-5646.1985.tb00428.x
Chippindale AK, Chu TJF, Rose MR (1996) Complex trade-offs and the evolution of starvation resistance in Drosophila melanogaster. Evolution (N Y) 50:753–766. https://doi.org/10.1111/j.1558-5646.1996.tb03885.x
Cole GL, Endler JA (2015) Artificial selection for food colour preferences. Proc R Soc B Biol Sci 282:20143108–20143108. https://doi.org/10.1098/rspb.2014.3108
Cowie RJ (1977) Optimal foraging in great tits (Parus major). Nature 268:137–139. https://doi.org/10.1038/268137a0
de Belle JS, Hilliker AJ, Sokolowski MB (1989) Genetic localization of foraging (for): a major gene for larval behavior in Drosophila melanogaster. Genetics 123:157–163
Drosophila Stock Center Mexico (2023) Drosophila Stock Center Mexico. https://bancodemoscas.fciencias.unam.mx/. Accessed 20 May 2023
Edelsparre AH, Vesterberg A, Lim JH et al (2014) Alleles underlying larval foraging behaviour influence adult dispersal in nature. Ecol Lett 17:333–339. https://doi.org/10.1111/ELE.12234
Endler JA (1986) Natural selection in the wild. Princeton University Press
Fox GA (2001) Failure-time analysis. In: Scheiner S, Gurevitch J (eds) Design and analysis of ecological experiments. Oxford University Press, New York, pp 235–266
Frey-roos F, Brodmann PA, Reyer HU (1995) Relationships between food resources, foraging patterns, and reproductive success in the water pipit. Anthus sp. spinoletta. Behav Ecol 6:287–295. https://doi.org/10.1093/BEHECO/6.3.287
Gibbons ME, Ferguson AM, Lee DR (2005) Both learning and heritability affect foraging behaviour of red-backed salamanders, Plethodon cinereus. Anim Behav 69:721–732. https://doi.org/10.1016/J.ANBEHAV.2004.06.021
Godin J-GJ, Smith SA (1988) A fitness cost of foraging in the guppy. Nature 333:69–71. https://doi.org/10.1038/333069a0
González-Candelas F, Ménsua JL, Mova A (1990) Larval competition in Drosophila melanogaster: effects on development time. Genetica 82:33–44. https://doi.org/10.1007/BF00057671
Grima B, Chélot E, Xia R, Rouyer F (2004) Morning and evening peaks of activity rely on different clock neurons of the Drosophila brain. Nature 431:869–873. https://doi.org/10.1038/nature02935
Grosjean Y, Rytz R, Farine J-P et al (2011) An olfactory receptor for food-derived odours promotes male courtship in Drosophila. Nature 478:236–240. https://doi.org/10.1038/nature10428
Hassell MP, Southwood TRE (1978) Foraging strategies of insects. Annu Rev Ecol Syst 9:75–98
Hayden BY (2018) Economic choice: the foraging perspective. Curr Opin Behav Sci 24:1–6. https://doi.org/10.1016/J.COBEHA.2017.12.002
Helfrich-Förster C (2000) Differential control of morning and evening components in the activity rhythm of Drosophila melanogaster - Sex-specific differences suggest a different quality of activity. J Biol Rhythms 15:135–154. https://doi.org/10.1177/074873040001500208
Horváth B, Kalinka AT (2016) Effects of larval crowding on quantitative variation for development time and viability in Drosophila melanogaster. Ecol Evol 6:8460–8473. https://doi.org/10.1002/ece3.2552
Isaac RE, Li C, Leedale AE, Shirras AD (2010) Drosophila male sex peptide inhibits siesta sleep and promotes locomotor activity in the post-mated female. Proc R Soc B Biol Sci 277:65–70. https://doi.org/10.1098/RSPB.2009.1236
Jaenike J (1982) Environmental modification of oviposition behavior in Drosophila. Am Nat 119:784–802. https://doi.org/10.2307/2460963
Jordan KW, Carbone MA, Yamamoto A et al (2007) Quantitative genomics of locomotor behavior in Drosophila melanogaster. Genome Biol 8:1–17. https://doi.org/10.1186/GB-2007-8-8-R172/FIGURES/7
Kassambara A, Kosinski M, Biecek P, Fabian S (2019) Package “survminer”: Drawing Survival Curves using “ggplot2”. https://cran.r-project.org/web/packages/survminer/index.html. Accessed 29 Jan 2020
Krebs RA, Thompson KA (2006) Direct and correlated effects of selection on flight after exposure to thermal stress in Drosophila melanogaster. Genet 1281(128):217–225. https://doi.org/10.1007/S10709-005-5704-X
Latter BDH, Robertson A (1962) The effects of inbreeding and artificial selection on reproductive fitness. Genet Res 3:110–138. https://doi.org/10.1017/S001667230000313X
Lee YCG, Yang Q, Chi W et al (2017) Genetic architecture of natural variation underlying adult foraging behavior that is essential for survival of Drosophila melanogaster. Genome Biol Evol 9:1357–1369. https://doi.org/10.1093/GBE/EVX089
Lemon WC (1991) Fitness consequences of foraging behaviour in the zebra finch. Nature 352:153–155. https://doi.org/10.1038/352153a0
Lin CC, Prokop-Prigge KA, Preti G, Potter CJ (2015) Food odors trigger Drosophila males to deposit a pheromone that guides aggregation and female oviposition decisions. Elife 4. https://doi.org/10.7554/ELIFE.08688.001
Mackay TFC, Heinsohn SL, Lyman RF, Moehring AJ, Morgan TJ, Rollmann SM (2005) Genetics and genomics of Drosophila mating behavior. Proc Natl Acad Sci USA 102:6622–6629
Manning A (1961) The effects of artificial selection for mating speed in Drosophila melanogaster. Anim Behav 9:82–92. Academic Press
Mittelbach GG (1983) Optimal foraging and growth in bluegills. Oecologia 59:157–162. https://doi.org/10.1007/BF00378832
Morris DW, Davidson DL (2000) Optimally foraging mice match patch use with habitat differences in fitness. Ecology 81:2061. https://doi.org/10.2307/177095
Morrison ML, Ralph CJ, Verner J, Jehl JR Jr (1990) Avian foraging: theory, methodology, and applications | Michael L. Cooper Ornithological Society, Los Angeles, Morrison
Moth JJ, Barker JSF (1977) Interspecific competition between Drosophila melanogaster and Drosophila simulans: Effects of adult density on adult viability. Genetica 47:203–218. https://doi.org/10.1007/BF00123242
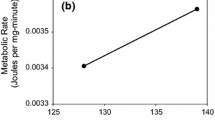
Mueller LD, Folk DG, Nguyen N et al (2005) Evolution of larval foraging behaviour in Drosophila and its effects on growth and metabolic rates. Physiol Entomol 30:262–269. https://doi.org/10.1111/J.1365-3032.2005.00458.X
Nakagawa S, Johnson PCD, Schielzeth H (2017) The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J R Soc Interface 14. https://doi.org/10.1098/rsif.2017.0213
Nunney L (1996) The response to selection for fast larval development in Drosophila melanogaster and its effect on adult weight: an example of a fitness trade-off. Evolution 50:1193–1204. Evolution
Partridge L, Fowler K (1992) Direct and correlated responses to selection on age at reproduction in Drosophila melanogaster. Evolution (N Y) 46:76–91. https://doi.org/10.1111/J.1558-5646.1992.TB01986.X
Partridge L, Prowse N, Pignatelli P (1999) Another set of responses and correlated responses to selection on age at reproduction in Drosophila melanogaster. Proc R Soc London Ser B Biol Sci 266:255–261. https://doi.org/10.1098/RSPB.1999.0630
Patrick SC, Weimerskirch H (2014) Personality, Foraging and Fitness Consequences in a Long Lived Seabird. PLoS One 9:e87269. https://doi.org/10.1371/journal.pone.0087269
Pereira HS, Sokolowski MB (1993) Mutations in the larval foraging gene affect adult locomotory behavior after feeding in Drosophila melanogaster. Proc Natl Acad Sci USA 90:5044–5046. https://doi.org/10.1073/pnas.90.11.5044
Perry G, Pianka ER (1997) Animal foraging: past, present and future. Trends Ecol Evol 12:360–364. https://doi.org/10.1016/S0169-5347(97)01097-5
Promislow DEL, Bugbee M (2000) Direct and correlated responses to selection on age at physiological maturity in Drosophila simulans. J Evol Biol 13:955–966. https://doi.org/10.1046/J.1420-9101.2000.00240.X
Pyke GH (1984) Optimal Foraging Theory: A Critical Review. Annu Rev Ecol Syst 15:523–575. https://doi.org/10.1146/annurev.es.15.110184.002515
Pyle DW (1978) (1978) Correlated responses to selection for a behavioral trait in Drosophila melanogaster. Behav Genet 84(8):333–340. https://doi.org/10.1007/BF01067396
Rathie KA, Nicholas FW (1980) Artificial selection with differing population structures. Genet Res (Camb) 36:117–131. Cambridge University Press
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Reader SM, Laland KN (2000) Diffusion of foraging innovations in the guppy. Anim Behav 60:175–180. https://doi.org/10.1006/ANBE.2000.1450
Réale D, Reader SM, Sol D et al (2007) Integrating animal temperament within ecology and evolution. Biol Rev 82:291–318. https://doi.org/10.1111/j.1469-185X.2007.00010.x
Reddiex AJ, Gosden TP, Bonduriansky R, Chenoweth SF (2013) Sex-specific fitness consequences of nutrient intake and the evolvability of diet preferences. Am Nat 182:91–102. https://doi.org/10.1086/670649
Reed DH, Frankham R (2003) Correlation between fitness and genetic diversity. Conserv Biol 17:230–237. https://doi.org/10.1046/j.1523-1739.2003.01236.x
Richmond RC, Gerking JL (1979) Oviposition site preference in Drosophila. Behav Genet 9:233–241
Ritchie ME (1990) Optimal foraging and fitness in Columbian ground squirrels. Oecologia 82:56–67. https://doi.org/10.1007/BF00318534
Rose MR (1984) Artificial selection on a fitness-component in Drosophila melanogaster. Evolution (N Y) 38:516. https://doi.org/10.2307/2408701
Sambucetti P, Scannapieco AC, Norry FM (2010) Direct and correlated responses to artificial selection for high and low knockdown resistance to high temperature in Drosophila buzzatii. J Therm Biol 35:232–238. https://doi.org/10.1016/J.JTHERBIO.2010.05.006
Schwasinger-Schmidt TE, Kachman SD, Harshman LG (2012) Evolution of starvation resistance in Drosophila melanogaster: measurement of direct and correlated responses to artificial selection. J Evol Biol 25:378–387. https://doi.org/10.1111/J.1420-9101.2011.02428.X
Scott AM, Dworkin I, Dukas R (2021) Evolution of sociability by artificial selection. Evolution (N Y). https://doi.org/10.1111/EVO.14370
Sewell D, Burnet B, Connolly K (1974) Genetic analysis of larval feeding behaviour in Drosophila melanogaster. Genet Res 24:163–173. https://doi.org/10.1017/S0016672300015196
Sokolowski MB (1980) Foraging strategies of Drosophila melanogaster: A chromosomal analysis. Behav Genet 10:291–302. https://doi.org/10.1007/BF01067774
Sokolowski MB (1985) Genetics and ecology of Drosophila melanogaster larval foraging and pupation behaviour. J Insect Physiol 31:857–864. https://doi.org/10.1016/0022-1910(85)90103-9
Sokolowski MB, Hansell RIC, Rotin D (1983) Drosophila larval foraging behavior. II. Selection in the sibling species, D. melanogaster and D. simulans. Behav Genet 13:169–177. https://doi.org/10.1007/BF01065665
Sokolowski MB, Pereira HS, Hughes K (1997) Evolution of foraging behavior in Drosophila by density-dependent selection. Proc Natl Acad Sci USA 94:7373–7377. https://doi.org/10.1073/PNAS.94.14.7373/ASSET/B8D66C7D-A47E-41D3-8F9C-679C11FD4CF8/ASSETS/GRAPHIC/PQ1371861002.JPEG
Soto-Yéber L, Soto-Ortiz J, Godoy P, Godoy-Herrera R (2018) The behavior of adult Drosophila in the wild. PLoS One 13:e0209917. https://doi.org/10.1371/journal.pone.0209917
Stephens DW, Krebs JR (1986) Foraging theory. Princeton University Press
Steven De Belle J, Sokoiowski MB (1987) Heredity of rover/sitter: Alternative foraging strategies of Drosophila melanogaster larvae. Heredity (Edinb) 59:73–83
Takahashi KH, Teramura K, Muraoka S et al (2013) (2012) Genetic correlation between the pre-adult developmental period and locomotor activity rhythm in Drosophila melanogaster. Hered 1104(110):312–320. https://doi.org/10.1038/hdy.2012.88
Therneau TM (2015) A package for survival analysis in S. version 2.38. Retrieved from: https://CRAN.Rproject.org/package=survival
Therneau TM (2022) Mixed effects cox models [R package coxme version 2.2-10]. Retrieved from https://cran.rproject
Traniello JFA (1989) Foraging strategies of ants. Annu Rev Entomol 34(34):191–210. https://doi.org/10.1146/ANNUREV.EN.34.010189.001203
Uetz GW (1992) Foraging strategies of spiders. Trends Ecol Evol 7:155–159
Vijendravarma RK, Narasimha S, Kawecki TJ (2012) Evolution of foraging behaviour in response to chronic malnutrition in Drosophila melanogaster. Proc R Soc B Biol Sci 279:3540–3546. https://doi.org/10.1098/RSPB.2012.0966
Wallin A (1988) The genetics of foraging behaviour: artificial selection for food choice in larvae of the fruitfly, Drosophila melanogaster. Anim Behav 36:106–114. https://doi.org/10.1016/S0003-3472(88)80253-7
Warfe DM, Barmuta LA (2004) Habitat structural complexity mediates the foraging success of multiple predator species. Oecologia 141:171–178. https://doi.org/10.1007/s00442-004-1644-x
Weimerskirch H, Gault A, Cherel Y (2005) Prey distribution and patchiness: factors in foraging success and efficiency of wandering albatrosses. Ecology 86:2611–2622. https://doi.org/10.1890/04-1866
Zimmerman M (1982) Optimal foraging: Random movement by pollen collecting bumblebees. Oecologia 53:394–398. https://doi.org/10.1007/BF00389020
Acknowledgments
We especially thank Adriana Ramírez Vargas for her guidance and assistance with flies’ maintenance. We thank members of the Guevara-Fiore Laboratory, for their assistance with selection experiments. John A. Endler and Thomas A. Keaney provided helpful comments on the manuscript. This research was supported by funding from VIEP-BUAP. We also thank the anonymous reviewers for providing valuable comments and advice on the manuscript.
Author information
Authors and Affiliations
Contributions
PGF and MS conceived the study; MS, DP & PL collected the data; MS & PL carried out the carried out the analyses; and MS, PGF & PL contributed to writing up the paper.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by M. Giurfa
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 886 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sevenello, M., Luna, P., De La Rosa-Perea, D. et al. Direct and correlated responses to artificial selection on foraging in Drosophila. Behav Ecol Sociobiol 77, 89 (2023). https://doi.org/10.1007/s00265-023-03363-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-023-03363-1