Abstract
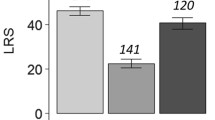
Females usually encounter males sequentially and should discriminate between potential partners among the males that they encounter. Thus, appropriate sequential mating decisions allow females to gain mate choice benefits in the circumstance of multiple encounters. For example, if mated females can assess the genetic quality of a male prior to mating, they can remate with genetically superior males and benefit indirectly by production of offspring of superior quality (trading-up hypothesis). Additionally, because female remating generates opportunities for postcopulatory sexual selection, females can improve son quality in postcopulation processes. In the beetle Gnatocerus cornutus, females prefer mates that court vigorously before copulation and prefer attractive males, which provide indirect benefits to females because they sire attractive sons. However, there is no information about male attractiveness and the resulting fitness benefits in female remating decisions, and here we focused on this. We found that females readily remated with attractive males and that females who had mated with unattractive males readily accepted remating. Subsequently, we established four experimental treatments: a single mating with an attractive or unattractive male, mating with an attractive and then an unattractive male, and mating with an unattractive and then an attractive male. All females that mated with attractive males produced attractive sons, regardless of the order of mating with the attractive male. These results suggest that females improve son quality by trading up and biasing fertilizations toward sperm of attractive males in G. cornutus. On the other hand, no direct effect of remating on female fitness was observed.
Significance statement
When females encounter males sequentially, they must choose appropriate partners among the mates that they encounter. In the beetle Gnatocerus cornutus, proper sequential mating decisions increase the likelihood of securing sperm of attractive males when females encounter multiple mates. That is, females readily remated with attractive males, and females that mated with unattractive males readily accepted remating. Additionally, they produced attractive sons regardless of the order of the mating with attractive males. This suggests that females bias fertilizations toward the sperm of attractive males to produce attractive sons. Therefore, females may produce attractive sons by remating with attractive males and biasing fertilization toward the sperm of attractive males, indirectly increasing female fitness in G. cornutus. By contrast, remating did not affect female fitness (longevity and offspring number), and so there was no direct effect of remating on female fitness.





Similar content being viewed by others
Data availability
The data is provided as supplementary material.
References
Abraham S, Goane L, Cladera J, Vera MT (2011) Effects of male nutrition on sperm storage and remating behavior in wild and laboratory Anastrepha fraterculus (Diptera: Tephritidae) females. J Insect Physiol 57:1501–1509. https://doi.org/10.1016/j.jinsphys.2011.08.004
Andersson M (1994) Sexual selection. Princeton University Press, Princeton
Ashby B, Gupta S (2013) Sexually transmitted infections in polygamous mating systems. Philos Trans R Soc Lond B Biol Sci 368:20120048. https://doi.org/10.1098/rstb.2012.0048
Bateman PW, Gilson LN, Ferguson JWH (2001) Male size and sequential mate preference in the cricket Gryllus bimaculatus. Anim Behav 61:631–637. https://doi.org/10.1006/anbe.2000.1617
Birkhead TR (2000) Defining and demonstrating postcopulatory female choice—again. Evolution 54:1057–1060. https://doi.org/10.1111/j.0014-3820.2000.tb00108.x
Birkhead TR, Møller AP (1998) Sperm competition. Academic Press, New York
Birkhead TR, Briskie JV, Moller AP (1993) Male sperm reserves and copulation frequency in birds. Behav Ecol Sociobiol 32:85–93. https://doi.org/10.1007/BF00164040
Bleu J, Bessa-Gomes C, Laloi D (2012) Evolution of female choosiness and mating frequency: effects of mating cost, density and sex ratio. Anim Behav 83:131–136. https://doi.org/10.1016/j.anbehav.2011.10.017
Burnham KP, Anderson DR (2002) Model selection and multimodel inference, 2nd edn. Springer, New York
Bussiégre LF, Hunt J, Jennions MD, Brooks R (2006) Sexual conflict and cryptic female choice in the black field cricket, Teleogryllus commodus. Evolution 60:792–800. https://doi.org/10.1111/j.0014-3820.2006.tb01157.x
Byrne PG, Rice WR (2005) Remating in Drosophila melanogaster: an examination of the trading-up and intrinsic male-quality hypotheses. J Evol Biol 18:1324–1331. https://doi.org/10.1111/j.1420-9101.2005.00918.x
Chapman T, Liddle LF, Kalb JM, Wolfner MF, Partridge L (1995) Cost of mating in Drosophila melanogaster females is mediated by male accessory gland products. Nature 373:241–244. https://doi.org/10.1038/373241a0
Crudgington HS, Siva-Jothy MT (2000) Genital damage, kicking and early death. Nature 407:855–856. https://doi.org/10.1038/35038154
Cunningham RT, Farias GH, Nakagawa S, Chambers DL (1971) Reproduction in the Mediterranean fruit fly: depletion of stored sperm in females. Ann Entomol Soc Am 64:312–313. https://doi.org/10.1093/aesa/64.1.312
De Simone GA, Manrique G, Pompilio L (2018) Females’ sequential mating decisions depend on both the quality of the courting male and the quality of the potential mates in a blood-sucking bug. Behav Ecol Sociobiol 72:1–9. https://doi.org/10.1007/s00265-018-2560-0
Demuth JP, Naidu A, Mydlarz LD (2012) Sex, war, and disease: the role of parasite infection on weapon development and mating success in a horned beetle (Gnatocerus cornutus). PLoS ONE 7:e28690. https://doi.org/10.1371/journal.pone.0028690
Dougherty LR, Shuker DM (2015) The effect of experimental design on the measurement of mate choice: a meta-analysis. Behav Ecol 26:311–319. https://doi.org/10.1093/beheco/aru125
Eberhard GW (1996) Female control: sexual selection by cryptic female choice. Princeton University Press, Princeton
Evans JP, Pilastro A, Ramnarine IW (2003) Sperm transfer through forced matings and its evolutionary implications in natural guppy (Poecilia reticulata) populations. Biol J Linn Soc 78:605–612. https://doi.org/10.1046/j.0024-4066.2002.00193.x
Fedina TY, Lewis SM (2007) Female mate choice across mating stages and between sequential mates in flour beetles. J Evol Biol 20:2138–2143. https://doi.org/10.1111/j.1420-9101.2007.01432.x
Firman RC, Gasparini C, Manier MK, Pizzari T (2017) Postmating female control: 20 years of cryptic female choice. Trends Ecol Evol 32:368–382. https://doi.org/10.1016/j.tree.2017.02.010
Fisher RA (1930) The genetical theory of natural selection. Clarendon Press, Oxford
Gabor CR, Halliday TR (1997) Sequential mate choice by multiple mating smooth newts: females become more choosy. Behav Ecol 8:162–166. https://doi.org/10.1093/beheco/8.2.162
Gibson RM, Langen TA (1996) How do animals choose their mates? Trends Ecol Evol 11:468–470. https://doi.org/10.1016/0169-5347(96)10050-1
Gillot C (2003) Male accessory gland secretions: modulators of female reproductive physiology and behavior. Annu Rev Entomol 48:163–184. https://doi.org/10.1146/annurev.ento.48.091801.112657
Halliday TR (1983) The study of mate choice. In: Bateson P (ed) Mate choice. Cambridge University Press, Cambridge, pp 3–32
Head ML, Hunt J, Jennions MD, Brooks R (2005) The indirect benefits of mating with attractive males outweigh the direct costs. PLoS Biol 3:e33. https://doi.org/10.1371/journal.pbio.0030033
Head ML, Hunt J, Brooks R (2006) Genetic association between male attractiveness and female differential allocation. Biol Lett 2:341–344. https://doi.org/10.1098/rsbl.2006.0474
Hosken DJ, Blanckenhorn WU (1999) Female multiple mating, inbreeding avoidance, and fitness: it is not only the magnitude of costs and benefits that counts. Behav Ecol 10:462–464. https://doi.org/10.1093/beheco/10.4.462
Hosken DJ, House CM (2011) Sexual selection. Curr Biol 21:R62–R65. https://doi.org/10.1016/j.cub.2010.11.053
Hosken DJ, Garner TWJ, Tregenza T, Wedell N, Ward PI (2003) Superior sperm competitors sire higher quality offspring. Proc R Soc Lond B Biol Sci 270:1933–1938. https://doi.org/10.1098/rspb.2003.2443
Institute SAS (2013) JMP release 11. SAS Institute Inc., Cary, NC
Janetos AC (1980) Strategies of female mate choice: a theoretical analysis. Behav Ecol Sociobiol 7:107–112. https://doi.org/10.1007/BF00299515
Jennions MD, Petrie M (2000) Why do females mate multiply? A review of the genetic benefits. Biol Rev 75:21–64. https://doi.org/10.1111/j.1469-185X.1999.tb00040.x
Katsuki M, Okada Y, Okada K (2012a) Impacts of diet quality on life-history and reproductive traits in male and female armed beetle, Gnatocerus cornutus. Ecol Entomol 37:463–470. https://doi.org/10.1111/j.1365-2311.2012.01390.x
Katsuki M, Harano T, Miyatake T, Okada K, Hosken DJ (2012b) Intralocus sexual conflict and offspring sex ratio. Ecol Lett 15:193–197. https://doi.org/10.1111/j.1461-0248.2011.01725.x
Kelly CD, Adam-Granger É (2020) Mating with sexually attractive males provides female Gryllus firmus field crickets with direct but not indirect fitness benefits. Behav Ecol Sociobiol 74:1–12. https://doi.org/10.1007/s00265-020-02859-4
Kirkpatrick M (1996) Good genes and direct selection in the evolution of mating preferences. Evolution 50:2125–2140. https://doi.org/10.2307/2410684
Kirkpatrick M, Barton NH (1997) The strength of indirect selection on female mating preferences. Proc Natl Acad Sci USA 94:1282–1286. https://doi.org/10.1073/pnas.94.4.1282
Kiyose K, Katsuki M, Suzaki Y, Okada K (2015) Competitive males but not attractive males reduce female fitness in Gnatocerus cornutus. Anim Behav 109:265–272. https://doi.org/10.1016/j.anbehav.2015.09.002
Klemme I, Eccard JA, Ylönen H (2006) Do female bank voles (Clethrionomys glareolus) mate multiply to improve on previous mates? Behav Ecol Sociobiol 60:415–421. https://doi.org/10.1007/s00265-006-0181-5
Klowden MJ (2001) Sexual receptivity in Anopheles gambiae mosquitoes: absence of control by male accessory gland substances. J Insect Physiol 47:661–666. https://doi.org/10.1016/S0022-1910(00)00127-X
Lande R (1981) Models of speciation by sexual selection on polygenic traits. Proc Nat Acad Sci USA 78:3721–3725. https://doi.org/10.1073/pnas.78.6.3721
Malo AF, Roldan ER, Garde J, Soler AJ, Gomendio M (2005) Antlers honestly advertise sperm production and quality. Proc R Soc Lond B Biol Sci 272:149–157. https://doi.org/10.1098/rspb.2004.2933
Michalczyk Ł, Millard AL, Martin OY, Lumley AJ, Emerson BC, Gage MJG (2011) Experimental evolution exposes female and male responses to sexual selection and conflict in Tribolium castaneum. Evolution 65:713–724. https://doi.org/10.1111/j.1558-5646.2010.01174.x
Møller AP, Jennions MD (2001) How important are direct fitness benefits of sexual selection? Naturwissenschaften 88:401–415. https://doi.org/10.1007/s001140100255
Moya-Laraño J, Fox CW (2006) Ejaculate size, second male size, and moderate polyandry increase female fecundity in a seed beetle. Behav Ecol 17:940–946. https://doi.org/10.1093/beheco/arl029
Okada K, Miyatake T (2009) Genetic correlations between weapons, body shape and fighting behaviour in the horned beetle Gnatocerus cornutus. Anim Behav 77:1057–1065. https://doi.org/10.1016/j.anbehav.2009.01.008
Okada K, Miyatake T (2010) Plasticity of size and allometry in multiple sexually selected traits in an armed beetle Gnatocerus cornutus. Evol Ecol 24:1339–1351. https://doi.org/10.1007/s10682-010-9370-9
Okada K, Katsuki M, Okada Y, Miyatake T (2011) Immature performance linked with exaggeration of a sexually selected trait in an armed beetle. J Evol Biol 24:1737–1743. https://doi.org/10.1111/j.1420-9101.2011.02303.x
Okada K, Katsuki M, Sharma MD, House CM, Hosken DJ (2014) Sexual conflict over mating in Gnatocerus cornutus: females prefer lovers not fighters. Proc R Soc Lond B Biol Sci 281:20140388. https://doi.org/10.1098/rspb.2014.0281
Okada K, Archer CR, Katsuki M, Suzaki Y, Sharma MD, House CM, Hosken DJ (2015) Polyandry and fitness in female horned flour beetles (Gnatocerus cornutus). Anim Behav 106:11–16. https://doi.org/10.1016/j.anbehav.2015.05.008
Okada K, Okada Y, Dall SRX, Hosken DJ (2019) Loser-effect duration evolves independently of fighting ability. Proc R Soc Lond B Biol Sci 286:20190582. https://doi.org/10.1098/rspb.2019.0582
Okada K, Katsuki M, Kiyose K, Okada Y (2020) Older males are more competitive in male fights and more aggressive toward females in the broad-horned flour beetle Gnatocerus cornutus. Behav Ecol Sociobiol 74:36. https://doi.org/10.1007/s00265-020-2815-4
Okada K, Katsuki M, Sharma MD, Kiyose K, Seko T, Okada Y, Wilson AJ, Hosken DJ (2021) Natural selection increases female fitness by reversing the exaggeration of a male sexually selected trait. Nat Commun 12:3420. https://doi.org/10.1038/s41467-021-23804-7
Pai A, Yan G (2002) Polyandry produces sexy sons at the cost of daughters in red flour beetles. Proc R Soc Lond B Biol Sci 269:361–368. https://doi.org/10.1098/rspb.2001.1893
Perry JC, Sirot LK, Wigby S (2013) The seminal symphony: how to compose an ejaculate. Trends Ecol Evol 28:414–422. https://doi.org/10.1016/j.tree.2013.03.005
Pilastro A, Simonato M, Bisazza A, Evans JP (2004) Cryptic female preference for colorful males in guppies. Evolution 58:665–669. https://doi.org/10.1111/j.0014-3820.2004.tb01690.x
Pitcher TE, Neff BD, Roff FH, Rowe L (2003) Multiple mating and sequential mate choice in guppies: females trade up. Proc R Soc Lond B Biol Sci 270:1623–1629. https://doi.org/10.1098/rspb.2002.2280
Pizzari T, Froman DP, Birkhead TR (2002) Pre- and post-insemination episodes of sexual selection in the fowl Gallus G Domesticus. Heredity 88:112–116. https://doi.org/10.1038/sj.hdy.6800014
R Core Team (2020). R: a language and environment for statistical computing. R Found Stat Computing, https://www.R-project.org/
Real LA (1991) Search theory and mate choice. II. Mutual interaction, assortative mating, and equilibrium variation in male and female fitness. Am Nat 138:901–917. https://doi.org/10.1086/285259
Reynolds JD, Gross MR (1990) Costs and benefits of female mate choice: is there a lek paradox? Am Nat 136:230–243. https://doi.org/10.1086/285093
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225. https://doi.org/10.2307/2409177
Rönn J, Katvala M, Arnqvist G (2007) Coevolution between harmful male genitalia and female resistance in seed beetles. Proc Natl Acad Sci 104:10921–10925. https://doi.org/10.1073/pnas.0701170104
Schäfer MA, Uhl G (2005) Sequential mate encounters: female but not male body size influences female remating behavior. Behav Ecol 16:461–466. https://doi.org/10.1093/beheco/ari013
Simmons LW (2001) Sperm competition and its evolutionary consequences in the insects. Princeton University Press, Princeton
Simmons LW (2005) The evolution of polyandry: sperm competition, sperm selection, and offspring viability. Ann Rev Ecol Evol Syst 36:125–146. https://doi.org/10.1146/annurev.ecolsys.36.102403.112501
Simmons LW, Tinghitella RM, Zuk M (2010) Quantitative genetic variation in courtship song and its covariation with immune function and sperm quality in the field cricket Teleogryllus oceanicus. Behav Ecol 21:1330–1336. https://doi.org/10.1093/beheco/arq154
Slatyer RA, Mautz BS, Backwell PR, Jennions MD (2012) Estimating genetic benefits of polyandry from experimental studies: a meta-analysis. Biol Rev 87:1–33. https://doi.org/10.1111/j.1469-185X.2011.00182.x
Taylor ML, Wigmore C, Hodgson DJ, Wedell N, Hosken DJ (2008) Multiple mating increases female fitness in Drosophila simulans. Anim Behav 76:963–970. https://doi.org/10.1016/j.anbehav.2008.05.015
Thomas ML, Simmons LW (2009) Male dominance influences pheromone expression, ejaculate quality, and fertilization success in the Australian field cricket, Teleogryllus oceanicus. Behav Ecol 20:1118–1124. https://doi.org/10.1093/beheco/arp105
Tregenza T, Wedell N (2002) Polyandrous females avoid costs of inbreeding. Nature 415:71–73. https://doi.org/10.1038/415071a
Tsuda Y, Yoshida T (1984) Population biology of the broad horned flour beetle Gnathocerus cornutus (F) (Coleoptera Tenebrionidae) I. Life table and population parameters. Appl Entomol Zool 19:129–131. https://doi.org/10.1303/aez.19.129
Yamane T, Okada K, Nakayama S, Miyatake T (2010) Dispersal and ejaculate strategies associated with exaggeration of weapon in an armed beetle. Proc R Soc Lond B Biol Sci 77:1705–1710. https://doi.org/10.1098/rspb.2009.2017
Yasui Y (1997) A “good-sperm” model can explain the evolution of multiple mating by females. Am Nat 149:573–584. https://doi.org/10.1086/286006
Zeh JA, Zeh DW (1997) The evolution of polyandry II: post-copulatory defences against genetic incompatibility. Proc R Soc Lond B Biol Sci 264:69–75. https://doi.org/10.1098/rspb.1997.0010
Funding
KO was supported by grants from the Japan Society for the Promotion of Science (KAKENHI 18K06417, 21H02540).
Author information
Authors and Affiliations
Contributions
K. K., M. K., Y. S., and K. O. conceived the study. K. K. and K.O. carried out experiments and analyses. K. K., M. K., Y. S., and K. O. contributed to writing the manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
All authors agree to publish this MS.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by N. Wedell.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kiyose, K., Katsuki, M., Suzaki, Y. et al. Female remating decisions and mate choice benefits in the beetle Gnatocerus cornutus. Behav Ecol Sociobiol 76, 55 (2022). https://doi.org/10.1007/s00265-022-03161-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-022-03161-1