Abstract
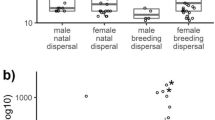
The costs and benefits of dispersal that select for sex-biased dispersal are still poorly understood. Many studies examine fitness consequences of dispersal after first breeding, while dispersal costs are most likely paid before first breeding during the movement, settlement, and post-settlement stages. We studied survival correlates of dispersal between flock settlement and first breeding during the first winter of juvenile willow tits (Poecile montanus), a small passerine that has female-biased natal dispersal, but shows no dispersal-associated survival differences after first breeding. This resident food-hoarding species winters in small stable non-kin territorial flocks. We collected capture-recapture data by following flocks from autumn to the following spring. We compared monthly survival and return rates of juveniles that were born and recruited within the study area (philopatric juveniles) and juveniles that originated from outside the study area (immigrant juveniles). Among males, survival was highest for philopatric juveniles whereas survival of females was higher among immigrant juveniles, providing one explanation for the female-biased natal dispersal observed in the species. Philopatric males may benefit from prior residency either through increased site familiarity and knowledge of winter food resources and/or by gaining higher social ranks during flock establishment. However, rank data provided little support for the latter hypothesis. Other mechanisms such as increased ability to find high-quality flocks and mates may be important for female survival. Our results provide further evidence that dispersal costs are paid mainly before first breeding and that sex-specific costs of dispersal play a role in the evolution of sex-biased dispersal.
Significance statement
This paper shows that female-biased dispersal can be a consequence of sex-specific costs and benefits of dispersal during the post-settlement stage of the dispersal process which is a very poorly understood stage in dispersal theory. By examining correlates of dispersal before rather than after first breeding as it is usually done, our study aids in understanding the selection pressures modifying dispersal strategies. Our results have wide applicability because there are many resident taxa similar to the willow tit (our study species) that have a settlement stage and a prolonged non-reproductive phase before their first reproduction.


Similar content being viewed by others
References
Arnold TW (2010) Uninformative parameters and model selection using Akaike’s information criterion. J Wildlife Manage 74:1175–1178
Baker PJ, Robertson CPJ, Funk SM, Harris S (1998) Potential fitness benefits of group living in the red fox, Vulpes vulpes. Anim Behav 56:1411–1424
Bonte D, Van Dyck H, Bullock JM et al (2012) Costs of dispersal. Biol Rev 87:290–312
Bonte D, De Roissart A, Wybouw N, Van Leeuwen T (2014) Fitness maximization by dispersal: evidence from an invasion experiment. Ecology 95:3104–3111
Bowler DE, Benton TG (2005) Causes and consequences of animal dispersal strategies: relating individual behaviour to spatial dynamics. Biol Rev 80:205–225
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach. Springer, New York
Clinchy M, Sheriff MJ, Zanette LY (2013) Predator-induced stress and the ecology of fear. Funct Ecol 27:56–65
Clobert J, Baguette M, Benton TG (2012) Dispersal ecology and evolution. Oxford University Press, Oxford
Devillard S, Bray Y (2009) Assessing the effect on survival of natal dispersal using multistate capture–recapture models. Ecology 90:2902–2912
Doligez B, Pärt T (2008) Estimating fitness consequences of dispersal: a road to ‘know-where’? Non-random dispersal and the underestimation of dispersers’ fitness. J Anim Ecol 77:1199–1211
Eden SF (1987a) Dispersal and competitive ability in the magpie: an experimental study. Anim Behav 35:764–772
Eden SF (1987b) Natal philopatry of the magpie Pica pica. Ibis 129:477–490
Ekman J (1979) Coherence, composition and territories of winter social groups of the willow tit Parus montanus and the Crested Tit P. cristatus. Ornis Scand 10:56–68
Ekman J (1990) Alliances in winter flocks of willow tits—effects of rank on survival and reproductive success in male–female associations. Behav Ecol Sociobiol 26:239–245
Ekman J, Griesser M (2016) Siberian jays: delayed dispersal in absence of cooperative breeding. In: Koenig WD, Dickinson J (eds) Cooperative breeding in vertebrates: studies of ecology, evolution, and behavior. Cambridge University Press, Cambridge, pp 6–18
Ekman J, Cederholm G, Askenmo C (1981) Spacing and survival in winter groups of willow tit Parus montanus and crested tit P. cristatus—a removal study. J Anim Ecol 50:1–9
Ekman J, Bylin A, Tegelström H (2000) Parental nepotism enhances survival of retained offspring in the Siberian jay. Behav Ecol 11:416–420
Ekman J, Eggers S, Griesser M, Tegelström H (2001) Queuing for preferred territories: delayed dispersal of Siberian jays. J Anim Ecol 70:317–324
Ekman J, Eggers S, Griesser M (2002) Fighting to stay; the role of sibling rivalry for delayed dispersal. Anim Behav 64:453–459
Gienapp P, Merilä J (2011) Sex-specific fitness consequences of dispersal in Siberian Jays. Behav Ecol Sociobiol 65:131–140
Greenwood PJ (1980) Mating systems, philopatry and dispersal in birds and mammals. Anim Behav 28:1140–1162
Griesser M, Nystrand M, Ekman J (2006) Reduced mortality selects for family cohesion in a social species. Proc R Soc Lond B 273:1881–1886
Gros A, Hovestadt T, Poethke HJ (2008) Evolution of sex-biased dispersal: the role of sex-specific dispersal costs, demographic stochasticity and inbreeding. Ecol Model 219:226–233
Hogstad O (1987) Social rank in winter flocks of willow tits Parus montanus. Ibis 129:1–9
Hogstad O (1990) Dispersal date and settlement of juvenile willow tits Parus montanus in winter flocks. Fauna Norv Ser C Cinclus 3:49–55
Hogstad O (2014) Ecology and behaviour of winter floaters in a subalpine population of Willow Tits, Poecile montanus. Ornis Fennica 91:29–38
Koivula K, Lahti K, Orell M, Rytkönen S (1993) Prior residency as a key determinant of social dominance in the willow tit (Parus montanus). Behav Ecol Sociobiol 33:283–287
Koivula K, Orell M, Rytkönen S (1996) Winter survival and breeding success of dominant and subordinate Willow Tits Parus montanus. Ibis 138:624–629
Kullberg C (1998) Spatial niche dynamics under predation risk in the willow tit Parus montanus. J Avian Biol 29:235–240
Kullberg C, Ekman J (2000) Does predation maintain tit community diversity? Oikos 89:41–45
Lahti K, Koivula K, Orell M (1994) Is the social hierarchy always linear in tits? J Avian Biol 25:347–348
Lahti K, Koivula K, Orell M, Rytkonen S (1996) Social dominance in free-living willow tits Parus montanus: determinants and some implications of hierarchy. Ibis 138:539–544
Lahti K, Orell M, Rytkönen S, Koivula K (1998) Time and food dependence in willow tit winter survival. Ecology 79:2904–2916
Lambrechts M, Dhondt AA (1986) Male quality, reproduction and survival in the great tit (Parus major). Behav Ecol Sociobiol 19:57–63
Lawson Handley LJ, Perrin N (2007) Advances in our understanding of mammalian sex-biased dispersal. Mol Ecol 16:1559–1578
Lebreton J-D, Burnham KP, Clobert J, Anderson DR (1992) Modelling survival and testing biological hypothesis using marked animals: a unified approach with case studies. Ecol Monogr 62:67–118
Massot M, Clobert J, Lecomte J, Barbault R (1994) Incumbent advantage in common lizards and their colonizing ability. J Anim Ecol 63:431–440
Nevoux M, Arlt D, Nicoll M, Jones C, Norris K (2013) The short- and long-term fitness consequences of natal dispersal in a wild bird population. Ecol Lett 16:438–445
Nilsson J-Å (1989) Causes and consequences of natal dispersal in the marsh tit, Parus palustris. J Anim Ecol 58:619–63
Nilsson J-Å (1990) Establishment success of experimentally delayed juvenile Marsh tits Parus palustris. Ethology 85:73–79
Nilsson J-Å, Smith HG (1988) Effects of dispersal date on winter flock establishment and social dominance in marsh tits, Parus palustris. J Anim Ecol 57:917–928
Orell M, Rytkönen S, Koivula K (1994) Causes of divorce in the monogamous willow tit, Parus montanus, and consequences for reproductive success. Anim Behav 48:1143–1154
Orell M, Lahti K, Koivula K, Rytkönen S, Welling P (1999) Immigration and gene flow in a northern willow tit (Parus montanus) population. J Evol Biol 12:283–295
Pakanen V-M, Rönkä A, Belda E-J, Luukkonen A, Kvist L, Koivula K (2010) Impact of dispersal status on estimates of local population growth rates in a Temminck’s stint (Calidris temminckii) population. Oikos 119:1493–1503
Pakanen V-M, Hildén O, Rönkä A, Belda E-J, Luukkonen A, Kvist L, Koivula K (2011) Breeding dispersal strategies following reproductive failure explain low apparent survival of immigrant Temminck’s stints. Oikos 120:615–622
Pärn H, Jensen H, Ringsby TH, Sæther B-E (2009) Sex-specific fitness correlates of dispersal in a house sparrow metapopulation. J Anim Ecol 78:1216–1225
Pärt T (1995) The importance of local familiarity and search costs for age-biased and sex-biased philopatry in the collared flycatcher. Anim Behav 49:1029–1038
R Development Core Team (2015) R: A language and environment for statistical computing. Version 3.1.3. R Foundation for statistical computing, Vienna, Austria. Available at: http://www.r-project.org/
Sandell M, Smith HG (1991) Dominance, prior occupancy, and winter residency in the great tit (Parus major). Behav Ecol Sociobiol 29:147–152
Silverin B (1997) The stress response and autumn dispersal behaviour in willow tits. Anim Behav 53:451–459
Tarwater CE, Beissinger SR (2012) Dispersal polymorphisms from natal phenotype–environment interactions have carry-over effects on lifetime reproductive success of a tropical parrot. Ecol Lett 15:1218–1229
Terraube J, Vasko V, Korpimäki E (2014) Mechanisms and reproductive consequences of breeding dispersal in a specialist predator under temporally varying food conditions. Oikos 124:762–771
Verhulst S, Nilsson J-Å (2008) The timing of birds’ breeding seasons: a review of experiments that manipulated timing of breeding. Philos T Roy Soc B 363:399–410
Waser PM, Nichols KM, Hadfield JD (2013) Fitness consequences of dispersal: is leaving home the best of a bad lot? Ecology 94:1287–1295
White GC, Burnham KP (1999) Program MARK: survival estimation from populations of marked animals. Bird Study 46:120–139
Young AJ, Monfort SL (2009) Stress and the costs of extra-territorial movement in a social carnivore. Biol Lett 5:439–441
Acknowledgments
We thank all field workers, especially Ari-Pekka Auvinen. We thank Robert L. Thomson for checking the text for English and three anonymous referees for valuable comments on the manuscript. This study was funded by the Academy of Finland for VMP (grant no. 278759) and MO (grant nos. 3548, 1051114, 1051369).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Funding
This study was funded by the Academy of Finland (grant numbers 278759, 3548, 1051114, and 1051369).
Additional information
Communicated by D. Rubenstein
Rights and permissions
About this article
Cite this article
Pakanen, VM., Koivula, K., Orell, M. et al. Sex-specific mortality costs of dispersal during the post-settlement stage promote male philopatry in a resident passerine. Behav Ecol Sociobiol 70, 1727–1733 (2016). https://doi.org/10.1007/s00265-016-2178-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-016-2178-z