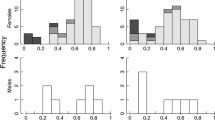
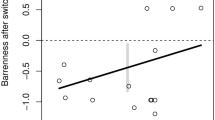
Abstract
Controversy has surrounded the question of why lek-breeding has evolved in certain ungulate species. Can the behavior be explained simply by males mapping onto a female distribution that is determined by factors unrelated to mating? Or are leks created because estrous females distinguish between males and favor males who cluster? Here I address these questions by looking at spatial distribution in lekking topi antelopes (Damaliscus lunatus). Contrary to the predictions of a model assuming male clustering in the zone of maximum female range overlap, territories were highly clustered also within this zone, and lek size correlated positively with population density. In support of models derived from the ideal free distribution of males onto female dispersion, leks were in areas with high female density during the rut. However, models not taking into account both individual variation in male quality and female mate preferences failed to explain the extreme male clumping also within high density areas, which was revealed by a strongly male-biased sex-ratio on leks. Additional support for the female preference-based model came from the finding that estrous females concentrated onto leks. Female preference for clustered males may develop if males initially follow an ideal free distribution of unequal competitors with high quality males slightly clustered at density hotspots; positive feedback between female benefits of preference for clustered males and male benefits of clustering could lead to contraction of the territorial network and lek behavior. Thus only the female preference-based model correctly predicted a negative correlation between male mating rate and resource density.




Similar content being viewed by others
References
Alexander RD (1975) Natural selection and specialized chorusing behaviour in acoustical insects. In Pimental D (ed) Insects, science and society. Academic Press, New York, pp 35–77
Bakker TCM, and Pomiankowski A (1995) The genetic basis of female mate preferences. J Evol Biol 8:129–171
Balmford A (1990) Lekking in Uganda kob. PhD thesis, University of Cambridge
Balmford A, Turyaho M (1992) Predation risk and lek-breeding in Uganda kob. Anim Behav 44:117–127
Balmford A, Rosser AM, Albon SD (1992) Correlates of female choice in a resource-defending antelope. Behav Ecol Sociobiol 31:107–114
Balmford A, Deutsch JC, Nefdt RJC, Clutton-Brock T (1993) Testing hotspot models of lek evolution: data from three species of ungulates. Behav Ecol Sociobiol 33:57–65
Bradbury JW, Gibson RM, Tsai IM (1986) Hotspots and the dispersion of leks. Anim Behav 34:1694–1709
Bradbury JW, Gibson RM, McCarthy CE, Vehrencamp SL (1989) Dispersion of displaying male sage grouse. 2. The role of female dispersion. Behav Ecol Sociobiol 24:15–24
Brashares JS, Garland Jr T, Arcese P (2000) Phylogenetic analysis of coadaptation in behavior, diet, and body size in the African antelope. Behav Ecol 11:452–463
Bro-Jørgensen J (2001) Lek-breeding in topi antelopes (Damaliscus lunatus). PhD thesis, University of London
Bro-Jørgensen J (2002) Overt female mate competition and preference for central males in a lekking antelope. Proc Natl Acad Sci USA 99:9290–9293
Bro-Jørgensen J (2003) No peace for estrous topi cows on lek. Behav Ecol (in press)
Bro-Jørgensen J, Durant SM (2003) Mating strategies of topi bulls: getting in the centre of attention. Anim Behav (in press)
Carbone C, Taborsky M (1996) Mate choice or harassment avoidance? A question of female control at the lek. Behav Ecol 7:370–373
Clutton-Brock TH (1989) Mammalian mating systems. Proc R Soc Lond B 236:339–372
Clutton-Brock TH, Hiraiwa-Hasegawa M, Robertson A (1989) Mate choice on fallow deer leks. Nature 340:463–465
Clutton-Brock TH, Price OF, MacColl ADC (1992) Mate retention, harassment, and the evolution of ungulate leks. Behav Ecol 3:234–242
Clutton-Brock TH, Deutsch JC, Nefdt RJC (1993) The evolution of ungulate leks. Anim Behav 46:1121–1138
Clutton-Brock TH, McComb KE, Deutsch JC (1996) Multiple factors affect the distribution of females in lek-breeding ungulates: a rejoinder. Behav Ecol 7:373–378
Daly M (1978) The cost of mating. Am Nat 112:771–773
Deutsch JC (1994) Lekking by default: female habitat preferences and male strategies in Uganda kob. J Anim Ecol 63:101–115
Duncan P (1975) Topi and their food supply. PhD thesis, University of Nairobi
Estes RD (1991) The behavior guide to African mammals. University of California Press, Berkeley, Calif.
Fretwell SD (1969) Dominance behavior and winter habitat distribution in juncos (Juncos hyemalis). Bird-Banding 34:293–306
Fretwell SD (1972) Populations in a seasonal environment. Princeton University Press, Princeton, N.J.
Fretwell SD, Lucas HL (1970) On territorial behaviour and other factors influencing habitat distribution in birds. Acta Biotheor 19:16–36
Gibson RM, Bachman GC (1992) The costs of female choice in a lekking bird. Behav Ecol 3:300–309
Gosling LM (1986) The evolution of mating strategies in male antelopes. In Rubenstein DI, Wrangham RW (eds) Ecological aspects of social evolution. Princeton University Press, Princeton, N.J., pp 244–281
Gosling LM, Petrie M (1990) Lekking in topi: a consequence of satellite behavior by small males at hotspots. Anim Behav 40:272–287
Hooge PN, Eichenlaub W, Solomon E (1999) The animal movement program: USGS, Alaska Biological Science Center, Anchorage
Höglund J, Alatalo RV (1995) Leks. Princeton University Press, Princeton, N.J.
Jarman P (1974) The social organisation of antelope in relation to their ecology. Behaviour 48:215–267
Jennrich RI, Turner FB (1969) Measurement of non-circular home range. J Theor Biol 22:227–237
Kokko H, Lindström J (1996) Kin selection and the evolution of leks: whose success do young males maximize? Proc R Soc Lond B 263:919–923
Krebs CJ (1989) Ecological methodology. Harper Collins, New York
Kruijt JP, de Vos JG, Bossema I (1972) The arena system of black grouse, Lyrurus tetrix tetrix (L.). Proc Int Ornithol Congr 15:399–423
McElligott AG, Gammell MP, Harty HC, Paini DR, Murphy DT, Walsh JT, Hayden TJ (2001) Sexual size dimorphism in fallow deer (Dama dama): do larger, heavier males gain greater mating success? Behav Ecol Sociobiol 49:266–272
McElligott AG, Altwegg R, Hayden TJ (2002) Age-specific survival and reproductive probabilities: evidence of senescence in male fallow deer (Dama dama). Proc R Soc Lond B 269:1129–1137
McNaughton SJ (1988) Mineral nutrition and spatial concentrations of African ungulates. Nature 334:343–345
Mertens H (1984) Age-determination in the topi (Damaliscus korrigum Ogilby) in the Virunga National-Park (Zaire). Mammalia 48:425–435
Montfort-Braham N (1974) Contribution à l'étude des structures sociales et du comportement des ongulés du Parc National de l'Akagera. 1ere partie: le topi (Damaliscus korrigum -Ogilby). FULREAC, Université de Liège, Liège
Monfort-Braham N (1975) Variations dans la structure sociale du topi, Damaliscus korrigum Ogilby, au Parc National de l'Akagera, Rwanda. Z Tierpsychol 39:332–364
Nefdt RJC (1992) Lek-breeding in Kafue lechwe. PhD thesis, University of Cambridge
Ottichilo WK, De Leeuw J, Skidmore AK, Prins HHT, Said MY (2000) Population trends of large non-migratory wild herbivores and livestock in the Masai Mara ecosystem, Kenya, between 1977 and 1997. Afr J Ecol 38:202–216
Parker GA, Sutherland WJ (1986) Ideal free distributions when individuals differ in competitive ability: phenotype-limited ideal free models. Anim Behav 34:1222–1242
Pelabon C, Komers PE, and Höglund J (1999) Do leks limit the frequency of aggressive encounters in fallow deer? Linking local male density and lek occurrence. Can J Zool 77:667–670
Pomiankowski A (1987) The costs of choice in sexual selection. J Theor Biol 128:195–218
Ranjitsinh MK (1989) The Indian blackbuck. Natraj, Dehra Dun, India
Saether SA (2002) Kin selection, female preferences and the evolution of leks: direct benefits may explain kin structuring. Anim Behav 63:1017–1020
Sutherland WJ (1996) From individual behaviour to population ecology. Oxford University Press, Oxford
Sutherland WJ, Parker GA (1985) Distribution of unequal competitors. In: Sibly RM, Smith RH (eds) Behavioural ecology: ecological consequences of adaptive behaviour. Blackwell, Oxford, pp 255–273
Sutherland WJ, Parker GA (1992) The relationship between continuous input and interference models of ideal free distributions of unequal competitors. Anim Behav 44:345–356
Sutherland WJ, Townsend CR, Patmore JM (1988) A test of the ideal free distribution with unequal competitors. Behav Ecol Sociobiol 23:51–53
Thirgood S, Langbein J, Putman RJ (1999) Intraspecific variation in ungulate mating strategies: the case of the flexible fallow deer: Adv Study Behav 28:333–361
Acknowledgements
I am grateful to S.M. Durant, L.M. Gosling, A. Pomiankowski, G. Cowlishaw, W.J. Sutherland, I.F. Owens, C. Roberts, M. Petrie, M. Rowcliffe, T.M. Jones and the anonymous referees for their advice and comments, to M. Borner and S. Cleaveland for their help in locating leks in Serengeti NP, to P. Duncan for providing data from an aerial topi census there, to the Department of Resource Surveys and Remote Sensing in Nairobi for access to topi surveys from Masai Mara National Reserve, and to the Masai Mara Ecological Monitoring Program and the Masai Mara Hyena Project (K. Holekamp) for providing data on rainfall. For permissions to do field work, I thank the Office of the President, Narok County Council and Kenya Wildlife Service in Kenya, COSTECH and TAWIRI in Tanzania, and the chief park wardens of Masai Mara National Reserve and Serengeti National Park. Funding was provided by the Danish Research Agency with additional support from the Institute of Zoology, Zoological Society of London, Ottilie Brorson's Travel Fund, and British Airways.
Author information
Authors and Affiliations
Additional information
Communicated by C. Nunn
An erratum to this article is available at http://dx.doi.org/10.1007/s00265-016-2249-1.
Rights and permissions
About this article
Cite this article
Bro-Jørgensen, J. The significance of hotspots to lekking topi antelopes (Damaliscus lunatus). Behav Ecol Sociobiol 53, 324–331 (2003). https://doi.org/10.1007/s00265-002-0573-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00265-002-0573-0