Abstract
Purpose
Implant-associated osteomyelitis still represents a demanding challenge due to unfavourable biological conditions, bacterial properties and incremental resistance to antibiotic treatment. Therefore different bactericide or bacteriostatic implant coatings have been developed recently to control local intramedullary infections. Controlled local release of gentamicin base from a highly lipophilic gentamicin palmitate compound achieves extended intramedullary retention times and thus may improve its bactericide effect.
Methods
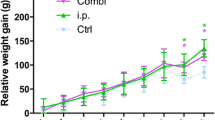
Forty male Sprague-Dawley rats were divided into two groups receiving an intramedullary femoral injection of 102 colony-forming units (CFU) of a common methicillin susceptible Staphylococcus aureus strain (MSSA Rosenbach) and either an uncoated femur nail (Group I) or a nail coated with gentamicin palmitate (Group II). Animals were observed for 28 and 42 days. Serum haptoglobin and relative weight gain were assessed as well as rollover cultures of explanted femur nails and histological scores of periprosthetic infection in dissected femurs.
Results
Implants coated with gentamicin palmitate significantly reduced periprosthetic bacterial growth as well as signs of systemic inflammation compared with uncoated implants.
Conclusions
Gentamicin palmitate appears to be a viable coating for the prevention of implant-associated infections. These findings will have to be confirmed in larger animal models as well as in clinical trials.






Similar content being viewed by others
References
Uçkay I, Jugun K, Gamulin A et al (2012) Chronic osteomyelitis. Curr Infect Dis Rep 14:566–575
Li G-Q, Guo F-F, Ou Y et al (2013) Epidemiology and outcomes of surgical site infections following orthopedic surgery. Am J Infect Control 41:1268–1271
Chang Y, Tai C-L, Hsieh P-H, Ueng SWN (2013) Gentamicin in bone cement: a potentially more effective prophylactic measure of infectionin joint arthroplasty. Bone Joint Res 2:220–226
Seghrouchni K, van Delden C, Dominguez D et al (2012) Remission after treatment of osteoarticular infections due to Pseudomonas aeruginosa versus Staphylococcus aureus: a case-controlled study. Int Orthop 36:1065–1071
Schmidmaier G, Lucke M, Wildemann B et al (2006) Prophylaxis and treatment of implant-related infections by antibiotic-coated implants: a review. Injury 37(Suppl 2):S105–S112
Lew DP, Waldvogel FA (2004) Osteomyelitis. Lancet 364:369–379
Rod-Fleury T, Dunkel N, Assal M et al (2011) Duration of post-surgical antibiotic therapy for adult chronic osteomyelitis: a single-centre experience. Int Orthop 35:1725–1731
Betz M, Abrassart S, Vaudaux P et al (2014) Increased risk of joint failure in hip prostheses infected with Staphylococcus aureus treated with debridement, antibiotics and implant retention compared to Streptococcus. Int Orthop. doi:10.1007/s00264-014-2510-z
Antoci V, Adams CS, Parvizi J et al (2008) The inhibition of Staphylococcus epidermidis biofilm formation by vancomycin-modified titanium alloy and implications for the treatment of periprosthetic infection. Biomaterials 29:4684–4690
Joosten U, Joist A, Gosheger G et al (2005) Effectiveness of hydroxyapatite-vancomycin bone cement in the treatment of Staphylococcus aureus induced chronic osteomyelitis. Biomaterials 26:5251–5258
Dion A, Langman M, Hall G, Filiaggi M (2005) Vancomycin release behaviour from amorphous calcium polyphosphate matrices intended for osteomyelitis treatment. Biomaterials 26:7276–7285
Kittinger C, Marth E, Windhager R et al (2011) Antimicrobial activity of gentamicin palmitate against high concentrations of Staphylococcus aureus. J Mater Sci Mater Med 22:1447–1453
Gracia E, Laclériga A, Monzón M et al (1998) Application of a rat osteomyelitis model to compare in vivo and in vitro the antibiotic efficacy against bacteria with high capacity to form biofilms. J Surg Res 79:146–153
Ozturk AM, Tabak AY, Aktekin CN et al (2008) Alendronate enhances antibiotic-impregnated bone grafts in the treatment of osteomyelitis. Int Orthop 32:821–827
Fassbender M, Minkwitz S, Kronbach Z et al (2013) Local gentamicin application does not interfere with bone healing in a rat model. Bone 55:298–304
Matl FD, Obermeier A, Repmann S et al (2008) New anti-infective coatings of medical implants. Antimicrob Agents Chemother 52:1957–1963
Lucke M, Schmidmaier G, Sadoni S et al (2003) A new model of implant-related osteomyelitis in rats. J Biomed Mater Res B Appl Biomater 67:593–602
Vogt S, Kühn K-D, Gopp U, Schnabelrauch M (2005) Resorbable antibiotic coatings for bone substitutes and implantable devices. Materwiss Werksttech 36:814–819
Heller DN, Peggins JO, Nochetto CB et al (2005) LC/MS/MS measurement of gentamicin in bovine plasma, urine, milk, and biopsy samples taken from kidneys of standing animals. J Chromatogr B Anal Technol Biomed Life Sci 821:22–30
Cowan ST, Shaw C, Williams RE (1954) Type strain for Staphylococcus aureus Rosenbach. J Gen Microbiol 10:174–176
Giffen PS, Turton J, Andrews CM et al (2003) Markers of experimental acute inflammation in the wistar han rat with particular reference to haptoglobin and C-reactive protein. Arch Toxicol 77:392–402
Welch JM, Weaver CM, Turner CH (2004) Adaptations to free-fall impact are different in the shafts and bone ends of rat forelimbs. J Appl Physiol 97:1859–1865
Gristina AG (1987) Biomaterial-centered infection: microbial adhesion versus tissue integration. Science 237:1588–1595
Lewis CS, Supronowicz PR, Zhukauskas RM et al (2012) Local antibiotic delivery with demineralized bone matrix. Cell Tissue Bank 13:119–127
Hanssen AD (2005) Local antibiotic delivery vehicles in the treatment of musculoskeletal infection. Clin Orthop Relat Res 437:91–96
Lucke M, Schmidmaier G, Sadoni S et al (2003) Gentamicin coating of metallic implants reduces implant-related osteomyelitis in rats. Bone 32:521–531
Lewis CS, Katz J, Baker MI et al (2011) Local antibiotic delivery with bovine cancellous chips. J Biomater Appl 26:491–506
Joosten U, Joist A, Frebel T et al (2004) Evaluation of an in situ setting injectable calcium phosphate as a new carrier material for gentamicin in the treatment of chronic osteomyelitis: studies in vitro and in vivo. Biomaterials 25:4287–4295
Cornell CN, Tyndall D, Waller S et al (1993) Treatment of experimental osteomyelitis with antibiotic-impregnated bone graft substitute. J Orthop Res 11:619–626
Hamanishi C, Kitamoto K, Tanaka S et al (1996) A self-setting TTCP-DCPD apatite cement for release of vancomycin. J Biomed Mater Res 33:139–143
Lucke M, Wildemann B, Sadoni S et al (2005) Systemic versus local application of gentamicin in prophylaxis of implant-related osteomyelitis in a rat model. Bone 36:770–778
Helbig L, Simank HG, Lorenz H et al (2014) Establishment of a new methicillin resistant Staphylococcus aureus animal model of osteomyelitis. Int Orthop 38:891–897
Acknowledgments
The present study was supported by Synthes GmbH, Umkirch, Germany. The authors thank Mr. Guido Schemken and his staff at the Central Animal Housing Facility in Marburg, as well as Prof. Dr. Markus Schofer and Dr. Stefan Lakemeier for their kind support in performing this study.
Conflict of interest
All authors declare that there is no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fölsch, C., Federmann, M., Kuehn, K.D. et al. Coating with a novel gentamicinpalmitate formulation prevents implant-associated osteomyelitis induced by methicillin-susceptible Staphylococcus aureus in a rat model. International Orthopaedics (SICOT) 39, 981–988 (2015). https://doi.org/10.1007/s00264-014-2582-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-014-2582-9