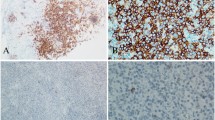
Abstract
The engagement of CD27 on lymphocytes with its ligand, CD70, on tumors is believed to mediate tumor immune evasion and the elevation of serum soluble CD27 (sCD27) levels in patients with CD70-positive malignancies. We previously showed that CD70 is expressed in extranodal natural killer/T-cell lymphoma, nasal type (ENKL), an Epstein–Barr virus (EBV)-related malignancy. However, little is known about serum sCD27 expression and its association with the clinical characteristics of, and the CD27/CD70 interaction in, ENKL. In the present study, we show that serum sCD27 is significantly elevated in the sera of patients with ENKL. The levels of serum sCD27 provided excellent diagnostic accuracy for discriminating patients with ENKL from healthy subjects, correlated positively with the levels of other diagnostic markers (lactate dehydrogenase, soluble interleukin-2 receptor, and EBV-DNA), and decreased significantly following treatment. Elevated serum sCD27 levels also correlated significantly with advanced clinical stage and tended to correspond with shorter survival, in patients with ENKL. Immunohistochemistry indicated that CD27-positive tumor-infiltrating immune cells exist adjacent to CD70-positive lymphoma cells. In addition, serum sCD27 levels in patients with CD70-positive ENKL were significantly higher than those in patients with CD70-negative ENKL, suggesting that the intra-tumoral CD27/CD70 interaction boosts the release of sCD27 in serum. Furthermore, the EBV-encoded oncoprotein latent membrane protein 1 upregulated CD70 expression in ENKL cells. Our results suggest that sCD27 may serve as a novel diagnostic biomarker and also may serve as a tool for evaluating the applicability of CD27/CD70-targeted therapies by predicting intra-tumoral CD70 expression and CD27/CD70 interaction in ENKL.




Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- Ab:
-
Antibody
- AUC:
-
Area under the curve
- CCL:
-
Chemokine (C–C motif) ligand
- CD40L:
-
CD40 ligand
- EBER:
-
Epstein–Barr virus-encoded small RNA
- EBV:
-
Epstein–Barr virus
- ECOG PS:
-
Eastern Cooperative Oncology Group Performance Status
- ENKL:
-
Extranodal natural killer/T-cell lymphoma, nasal type
- FFPE:
-
Formalin-fixed, paraffin-embedded
- IL:
-
Interleukin
- IPI:
-
International Prognostic Index
- LDH:
-
Lactate dehydrogenase
- LMP1:
-
Latent membrane protein 1
- NK:
-
Natural killer
- PINK:
-
Prognostic index of natural killer lymphoma
- RCC:
-
Renal cell carcinoma
- ROC:
-
Receiver operating characteristic
- sCD27:
-
Soluble CD27
- sIL-2R:
-
Soluble IL-2 receptor
- sPD-L1:
-
Soluble programmed death-ligand 1
- TIL:
-
Tumor-infiltrating lymphocyte
- TNF:
-
Tumor necrosis factor
References
Takahara M, Kumai T, Kishibe K, Nagato T, Harabuchi Y (2021) Extranodal NK/T-cell lymphoma, nasal type: genetic, biologic, and clinical aspects with a central focus on Epstein–Barr virus relation. Microorganisms. https://doi.org/10.3390/microorganisms9071381
Nagato T, Takahara M, Harabuchi Y (2020) Ulcerated lesions of the midline gingiva and soft palate. Am J Med Sci 360:77. https://doi.org/10.1016/j.amjms.2020.03.012
Harabuchi Y, Yamanaka N, Kataura A, Imai S, Kinoshita T, Mizuno F, Osato T (1990) Epstein–Barr virus in nasal T-cell lymphomas in patients with lethal midline granuloma. Lancet 335:128–130. https://doi.org/10.1016/0140-6736(90)90002-m
Harabuchi Y, Imai S, Wakashima J, Hirao M, Kataura A, Osato T, Kon S (1996) Nasal T-cell lymphoma causally associated with Epstein–Barr virus: clinicopathologic, phenotypic, and genotypic studies. Cancer 77:2137–2149
Ishii H, Takahara M, Nagato T, Kis LL, Nagy N, Kishibe K, Harabuchi Y, Klein E (2012) Monocytes enhance cell proliferation and LMP1 expression of nasal natural killer/T-cell lymphoma cells by cell contact-dependent interaction through membrane-bound IL-15. Int J Cancer 130:48–58. https://doi.org/10.1002/ijc.25969
Takahara M, Nagato T, Komabayashi Y, Yoshino K, Ueda S, Kishibe K, Harabuchi Y (2013) Soluble ICAM-1 secretion and its functional role as an autocrine growth factor in nasal NK/T cell lymphoma cells. Exp Hematol 41:711–718. https://doi.org/10.1016/j.exphem.2013.03.009
Komabayashi Y, Kishibe K, Nagato T, Ueda S, Takahara M, Harabuchi Y (2014) Downregulation of miR-15a due to LMP1 promotes cell proliferation and predicts poor prognosis in nasal NK/T-cell lymphoma. Am J Hematol 89:25–33. https://doi.org/10.1002/ajh.23570
Nagato T, Ueda S, Takahara M, Kishibe K, Komabayashi Y, Kumai T, Ohara K, Hirata-Nozaki Y, Harabuchi S, Hayashi R, Ohkuri T, Bernasconi M, Nadal D, Kobayashi H, Harabuchi Y (2019) Cyclin-dependent kinase 1 and survivin as potential therapeutic targets against nasal natural killer/T-cell lymphoma. Lab Investig 99:612–624. https://doi.org/10.1038/s41374-018-0182-9
Nagato T, Ohkuri T, Ohara K, Hirata Y, Kishibe K, Komabayashi Y, Ueda S, Takahara M, Kumai T, Ishibashi K, Kosaka A, Aoki N, Oikawa K, Uno Y, Akiyama N, Sado M, Takei H, Celis E, Harabuchi Y, Kobayashi H (2017) Programmed death-ligand 1 and its soluble form are highly expressed in nasal natural killer/T-cell lymphoma: a potential rationale for immunotherapy. Cancer Immunol Immunother 66:877–890. https://doi.org/10.1007/s00262-017-1987-x
Jacobs J, Deschoolmeester V, Zwaenepoel K, Rolfo C, Silence K, Rottey S, Lardon F, Smits E, Pauwels P (2015) CD70: an emerging target in cancer immunotherapy. Pharmacol Ther 155:1–10. https://doi.org/10.1016/j.pharmthera.2015.07.007
Flieswasser T, Van den Eynde A, Van Audenaerde J, De Waele J, Lardon F, Riether C, de Haard H, Smits E, Pauwels P, Jacobs J (2022) The CD70–CD27 axis in oncology: the new kids on the block. J Exp Clin Cancer Res 41:12. https://doi.org/10.1186/s13046-021-02215-y
Croft M (2009) The role of TNF superfamily members in T-cell function and diseases. Nat Rev Immunol 9:271–285. https://doi.org/10.1038/nri2526
Nolte MA, van Olffen RW, van Gisbergen KP, van Lier RA (2009) Timing and tuning of CD27–CD70 interactions: the impact of signal strength in setting the balance between adaptive responses and immunopathology. Immunol Rev 229:216–231. https://doi.org/10.1111/j.1600-065X.2009.00774.x
Ruf M, Mittmann C, Nowicka AM, Hartmann A, Hermanns T, Poyet C, van den Broek M, Sulser T, Moch H, Schraml P (2015) pVHL/HIF-regulated CD70 expression is associated with infiltration of CD27+ lymphocytes and increased serum levels of soluble CD27 in clear cell renal cell carcinoma. Clin Cancer Res 21:889–898. https://doi.org/10.1158/1078-0432.CCR-14-1425
Wischhusen J, Jung G, Radovanovic I, Beier C, Steinbach JP, Rimner A, Huang H, Schulz JB, Ohgaki H, Aguzzi A, Rammensee HG, Weller M (2002) Identification of CD70-mediated apoptosis of immune effector cells as a novel immune escape pathway of human glioblastoma. Cancer Res 62:2592–2599
Huang J, Jochems C, Anderson AM, Talaie T, Jales A, Madan RA, Hodge JW, Tsang KY, Liewehr DJ, Steinberg SM, Gulley JL, Schlom J (2013) Soluble CD27-pool in humans may contribute to T cell activation and tumor immunity. J Immunol 190:6250–6258. https://doi.org/10.4049/jimmunol.1300022
Yoshino K, Kishibe K, Nagato T, Ueda S, Komabayashi Y, Takahara M, Harabuchi Y (2013) Expression of CD70 in nasal natural killer/T cell lymphoma cell lines and patients; its role for cell proliferation through binding to soluble CD27. Br J Haematol 160:331–342. https://doi.org/10.1111/bjh.12136
Harris NL, Jaffe ES, Diebold J, Flandrin G, Muller-Hermelink HK, Vardiman J, Lister TA, Bloomfield CD (2000) The World Health Organization classification of neoplasms of the hematopoietic and lymphoid tissues: report of the Clinical Advisory Committee meeting—Airlie House, Virginia, November 1997. Hematol J 1:53–66. https://doi.org/10.1038/sj.thj.6200013
Jacobs J, Zwaenepoel K, Rolfo C, Van den Bossche J, Deben C, Silence K, Hermans C, Smits E, Van Schil P, Lardon F, Deschoolmeester V, Pauwels P (2015) Unlocking the potential of CD70 as a novel immunotherapeutic target for non-small cell lung cancer. Oncotarget 6:13462–13475. https://doi.org/10.18632/oncotarget.3880
Nagato T, Kobayashi H, Kishibe K, Takahara M, Ogino T, Ishii H, Oikawa K, Aoki N, Sato K, Kimura S, Shimizu N, Tateno M, Harabuchi Y (2005) Expression of interleukin-9 in nasal natural killer/T-cell lymphoma cell lines and patients. Clin Cancer Res 11:8250–8257. https://doi.org/10.1158/1078-0432.CCR-05-1426
Hayashi R, Nagato T, Kumai T, Ohara K, Ohara M, Ohkuri T, Hirata-Nozaki Y, Harabuchi S, Kosaka A, Nagata M, Yajima Y, Yasuda S, Oikawa K, Kono M, Kishibe K, Takahara M, Katada A, Hayashi T, Celis E, Harabuchi Y, Kobayashi H (2020) Expression of placenta-specific 1 and its potential for eliciting anti-tumor helper T-cell responses in head and neck squamous cell carcinoma. Oncoimmunology 10:1856545. https://doi.org/10.1080/2162402X.2020.1856545
Kumai T, Nagato T, Kobayashi H, Komabayashi Y, Ueda S, Kishibe K, Ohkuri T, Takahara M, Celis E, Harabuchi Y (2015) CCL17 and CCL22/CCR4 signaling is a strong candidate for novel targeted therapy against nasal natural killer/T-cell lymphoma. Cancer Immunol Immunother 64:697–705. https://doi.org/10.1007/s00262-015-1675-7
Niedobitek G, Fahraeus R, Herbst H, Latza U, Ferszt A, Klein G, Stein H (1992) The Epstein–Barr virus encoded membrane protein (LMP) induces phenotypic changes in epithelial cells. Virchows Arch B Cell Pathol Incl Mol Pathol 62:55–59. https://doi.org/10.1007/BF02899665
Choi IK, Wang Z, Ke Q, Hong M, Qian Y, Zhao X, Liu Y, Kim HJ, Ritz J, Cantor H, Rajewsky K, Wucherpfennig KW, Zhang B (2018) Signaling by the Epstein–Barr virus LMP1 protein induces potent cytotoxic CD4(+) and CD8(+) T cell responses. Proc Natl Acad Sci U S A 115:E686–E695. https://doi.org/10.1073/pnas.1713607115
van Oers MH, Pals ST, Evers LM, van der Schoot CE, Koopman G, Bonfrer JM, Hintzen RQ, von dem Borne AE, van Lier RA (1993) Expression and release of CD27 in human B-cell malignancies. Blood 82:3430–3436
Nilsson A, de Milito A, Mowafi F, Winberg G, Bjork O, Wolpert EZ, Chiodi F (2005) Expression of CD27–CD70 on early B cell progenitors in the bone marrow: implication for diagnosis and therapy of childhood ALL. Exp Hematol 33:1500–1507. https://doi.org/10.1016/j.exphem.2005.10.005
Ho AW, Hatjiharissi E, Ciccarelli BT, Branagan AR, Hunter ZR, Leleu X, Tournilhac O, Xu L, O’Connor K, Manning RJ, Santos DD, Chemaly M, Patterson CJ, Soumerai JD, Munshi NC, McEarchern JA, Law CL, Grewal IS, Treon SP (2008) CD27–CD70 interactions in the pathogenesis of Waldenstrom macroglobulinemia. Blood 112:4683–4689. https://doi.org/10.1182/blood-2007-04-084525
Ishii H, Ogino T, Berger C, Kochli-Schmitz N, Nagato T, Takahara M, Nadal D, Harabuchi Y (2007) Clinical usefulness of serum EBV DNA levels of BamHI W and LMP1 for Nasal NK/T-cell lymphoma. J Med Virol 79:562–572. https://doi.org/10.1002/jmv.20853
Suzuki R, Yamaguchi M, Izutsu K, Yamamoto G, Takada K, Harabuchi Y, Isobe Y, Gomyo H, Koike T, Okamoto M, Hyo R, Suzumiya J, Nakamura S, Kawa K, Oshimi K, Group NK-cTS (2011) Prospective measurement of Epstein–Barr virus-DNA in plasma and peripheral blood mononuclear cells of extranodal NK/T-cell lymphoma, nasal type. Blood 118:6018–6022. https://doi.org/10.1182/blood-2011-05-354142
Kok M, Bonfrer JM, Korse CM, de Jong D, Kersten MJ (2003) Serum soluble CD27, but not thymidine kinase, is an independent prognostic factor for outcome in indolent non-Hodgkin’s lymphoma. Tumour Biol 24:53–60. https://doi.org/10.1159/000070661
Goto N, Tsurumi H, Takemura M, Kanemura N, Kasahara S, Hara T, Yasuda I, Shimizu M, Yamada T, Sawada M, Takahashi T, Yamada T, Seishima M, Moriwaki H, Takami T (2012) Serum soluble CD27 level is associated with outcome in patients with diffuse large B-cell lymphoma treated with rituximab, cyclophosphamide, doxorubicin, vincristine and prednisolone. Leuk Lymphoma 53:1494–1500. https://doi.org/10.3109/10428194.2012.660627
Kashima J, Okuma Y, Hosomi Y, Hishima T (2019) High serum soluble CD27 level correlates with poor performance status and reduced survival in patients with advanced lung cancer. Oncology 97:365–372. https://doi.org/10.1159/000502441
Wang QJ, Hanada K, Robbins PF, Li YF, Yang JC (2012) Distinctive features of the differentiated phenotype and infiltration of tumor-reactive lymphocytes in clear cell renal cell carcinoma. Cancer Res 72:6119–6129. https://doi.org/10.1158/0008-5472.CAN-12-0588
Chahlavi A, Rayman P, Richmond AL, Biswas K, Zhang R, Vogelbaum M, Tannenbaum C, Barnett G, Finke JH (2005) Glioblastomas induce T-lymphocyte death by two distinct pathways involving gangliosides and CD70. Cancer Res 65:5428–5438. https://doi.org/10.1158/0008-5472.CAN-04-4395
Diegmann J, Junker K, Loncarevic IF, Michel S, Schimmel B, von Eggeling F (2006) Immune escape for renal cell carcinoma: CD70 mediates apoptosis in lymphocytes. Neoplasia 8:933–938. https://doi.org/10.1593/neo.06451
Yang ZZ, Grote DM, Xiu B, Ziesmer SC, Price-Troska TL, Hodge LS, Yates DM, Novak AJ, Ansell SM (2014) TGF-beta upregulates CD70 expression and induces exhaustion of effector memory T cells in B-cell non-Hodgkin’s lymphoma. Leukemia 28:1872–1884. https://doi.org/10.1038/leu.2014.84
Kitajima S, Lee KL, Fujioka M, Sun W, You J, Chia GS, Wanibuchi H, Tomita S, Araki M, Kato H, Poellinger L (2018) Hypoxia-inducible factor-2 alpha up-regulates CD70 under hypoxia and enhances anchorage-independent growth and aggressiveness in cancer cells. Oncotarget 9:19123–19135. https://doi.org/10.18632/oncotarget.24919
Israel BF, Gulley M, Elmore S, Ferrini S, Feng WH, Kenney SC (2005) Anti-CD70 antibodies: a potential treatment for EBV+ CD70-expressing lymphomas. Mol Cancer Ther 4:2037–2044. https://doi.org/10.1158/1535-7163.MCT-05-0253
Baba M, Okamoto M, Hamasaki T, Horai S, Wang X, Ito Y, Suda Y, Arima N (2008) Highly enhanced expression of CD70 on human T-lymphotropic virus type 1-carrying T-cell lines and adult T-cell leukemia cells. J Virol 82:3843–3852. https://doi.org/10.1128/JVI.02013-07
Balsas P, Veloza L, Clot G, Sureda-Gomez M, Rodriguez ML, Masaoutis C, Frigola G, Navarro A, Bea S, Nadeu F, Gine E, Lopez-Guillermo A, Martinez A, Ribera-Cortada I, Engel P, Quintanilla-Martinez L, Klapper W, Campo E, Amador V (2021) SOX11, CD70, and Treg cells configure the tumor-immune microenvironment of aggressive mantle cell lymphoma. Blood 138:2202–2215. https://doi.org/10.1182/blood.2020010527
Uchida J, Yasui T, Takaoka-Shichijo Y, Muraoka M, Kulwichit W, Raab-Traub N, Kikutani H (1999) Mimicry of CD40 signals by Epstein–Barr virus LMP1 in B lymphocyte responses. Science 286:300–303. https://doi.org/10.1126/science.286.5438.300
Acknowledgements
The authors thank Dr. Norio Shimizu (Tokyo Medical and Dental University) for generously providing the cell line, and Ms. Rie Matsumoto (Department of Pathology, Asahikawa Medical University) and Ms. Keiko Nishikura (Department of Dermatology, Asahikawa Medical University) for technical assistance.
Funding
This work was supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI [Grant No. 18K09310 (Nagato T), 21K09599 (Nagato T), 21H03082 (Harabuchi Y), and 22H02838 (Kobayashi H)].
Author information
Authors and Affiliations
Contributions
TN and HK conceptualized and designed the study. TN, HK, RH, MT, KK, SH, MK, HY, RW, YH-N, KO, TK, AK, TH, and YH performed patient management and material preparation. TN performed experiments. TN, HK, RH, and HK analyzed and interpreted data. TN, HK, RH, SY, YY, AK, TO, KO, and HK discussed the results. TN wrote the first draft of the manuscript, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Ethics approval
This study was performed in compliance with the principles of the Declaration of Helsinki and subsequent amendments, and the study protocol was approved by the Institutional Review Board of the Asahikawa Medical University (#1332–3).
Informed consent
Informed consent to participate and publish was obtained by the opt-out method on the Asahikawa Medical University website.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nagato, T., Komatsuda, H., Hayashi, R. et al. Expression of soluble CD27 in extranodal natural killer/T-cell lymphoma, nasal type: potential as a biomarker for diagnosis and CD27/CD70-targeted therapy. Cancer Immunol Immunother 72, 2087–2098 (2023). https://doi.org/10.1007/s00262-023-03394-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03394-7