Abstract
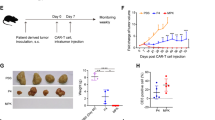
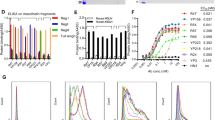
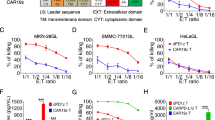
Chimeric antigen receptor (CAR)-T cell therapy has impressive efficacy in hematological malignancies, but its application in solid tumors remains a challenge. Multiple hurdles associated with the biological and immunological features of solid tumors currently limit the application of CAR-T cells in the treatment of solid tumors. Using syngeneic mouse models, we recently reported that CAR-T cells engineered to concomitantly produce interleukin (IL)-7 and chemokine (C–C motif) ligand 19 (CCL19)-induced potent anti-tumor efficacy against solid tumors through an improved ability of migration and proliferation even in an immunosuppressive tumor microenvironment. In this study, for a preclinical evaluation preceding clinical application, we further explored the potential of IL-7/CCL19-producing human CAR-T cells using models that mimic the clinical features of solid tumors. Human anti-mesothelin CAR-T cells producing human IL-7/CCL19 achieved complete eradication of orthotopic pre-established malignant mesothelioma and prevented a relapse of tumors with downregulated antigen expression. Moreover, mice with patient-derived xenograft of mesothelin-positive pancreatic cancers exhibited significant inhibition of tumor growth and prolonged survival following treatment with IL-7/CCL19-producing CAR-T cells, compared to treatment with conventional CAR-T cells. Transfer of IL-7/CCL19-producing CAR-T cells resulted in an increase in not only CAR-T cells but also non-CAR-T cells within the tumor tissues and downregulated the expression of exhaustion markers, including PD-1 and TIGIT, on the T cells. Taken together, our current study elucidated the exceptional anti-tumor efficacy of IL-7/CCL19-producing human CAR-T cells and their potential for clinical application in the treatment of patients with solid tumors.






Similar content being viewed by others
Availability of data and material
The datasets generated and analyzed during the current study are available from the corresponding author upon a reasonable request.
Abbreviations
- CAR:
-
Chimeric antigen receptor
- DCs:
-
Dendritic cells
- E:T:
-
Effector to target
- IL:
-
Interleukin
- IFN:
-
Interferon
- I.v.:
-
Intravenously
- NSG:
-
NOD.Cg-PrkdcscidIl2rgtm1Wjl/Szj
- PD-1:
-
Programed cell death 1
- PDX:
-
Patient-derived xenograft
- S.c.:
-
Subcutaneously
- ScFv:
-
Single-chain variable fragment
- TIGIT:
-
T cell immunoreceptor with Ig and ITIM domains
- TILs:
-
Tumor-infiltrating lymphocytes
References
SL Maude TW Laetsch J Buechner S Rives M Boyer H Bittencourt P Bader MR Verneris HE Stefanski GD Myers M Qayed B Moerloose De H Hiramatsu K Schlis KL Davis PL Martin ER Nemecek GA Yanik C Peters A Baruchel N Boissel F Mechinaud A Balduzzi J Krueger CH June BL Levine P Wood T Taran M Leung KT Mueller Y Zhang K Sen D Lebwohl MA Pulsipher SA Grupp 2018 Tisagenlecleucel in children and young adults with b-cell lymphoblastic leukemia N Engl J Med 378 5 439 448 https://doi.org/10.1056/NEJMoa1709866
SS Neelapu FL Locke NL Bartlett LJ Lekakis DB Miklos CA Jacobson I Braunschweig OO Oluwole T Siddiqi Y Lin JM Timmerman PJ Stiff JW Friedberg IW Flinn A Goy BT Hill MR Smith A Deol U Farooq P McSweeney J Munoz I Avivi JE Castro JR Westin JC Chavez A Ghobadi KV Komanduri R Levy ED Jacobsen TE Witzig P Reagan A Bot J Rossi L Navale Y Jiang J Aycock M Elias D Chang J Wiezorek WY Go 2017 Axicabtagene ciloleucel car t-cell therapy in refractory large b-cell lymphoma N Engl J Med 377 26 2531 2544 https://doi.org/10.1056/NEJMoa1707447
JH Park I Riviere M Gonen X Wang B Senechal KJ Curran C Sauter Y Wang B Santomasso E Mead M Roshal P Maslak M Davila RJ Brentjens M Sadelain 2018 Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia N Engl J Med 378 5 449 459 https://doi.org/10.1056/NEJMoa1709919
SJ Schuster MR Bishop CS Tam EK Waller P Borchmann JP McGuirk U Jager S Jaglowski C Andreadis JR Westin I Fleury V Bachanova SR Foley PJ Ho S Mielke JM Magenau H Holte S Pantano LB Pacaud R Awasthi J Chu O Anak G Salles RT Maziarz 2019 Tisagenlecleucel in adult relapsed or refractory diffuse large b-cell lymphoma N Engl J Med 380 1 45 56 https://doi.org/10.1056/NEJMoa1804980
CH June RS O'Connor OU Kawalekar S Ghassemi MC Milone 2018 CAR T cell immunotherapy for human cancer Science 359 6382 1361 1365 https://doi.org/10.1126/science.aar6711
S Guedan M Ruella CH June 2019 Emerging cellular therapies for cancer Annu Rev Immunol 37 145 171 https://doi.org/10.1146/annurev-immunol-042718-041407
RG Majzner CL Mackall 2018 Tumor antigen escape from Car t-cell therapy Cancer Discov 8 10 1219 1226 https://doi.org/10.1158/2159-8290.CD-18-0442
AJ Petty B Heyman Y Yang 2020 Chimeric antigen receptor cell therapy: overcoming obstacles to battle cancer Cancers (Basel) https://doi.org/10.3390/cancers12040842
TF Gajewski SR Woo Y Zha R Spaapen Y Zheng L Corrales S Spranger 2013 Cancer immunotherapy strategies based on overcoming barriers within the tumor microenvironment Curr Opin Immunol 25 2 268 276 https://doi.org/10.1016/j.coi.2013.02.009
M Martinez EK Moon 2019 CAR T cells for solid tumors: new strategies for finding, infiltrating, and surviving in the tumor microenvironment Front Immunol 10 128 https://doi.org/10.3389/fimmu.2019.00128
M Koneru TJ Purdon D Spriggs S Koneru RJ Brentjens 2015 IL-12 secreting tumor-targeted chimeric antigen receptor T cells eradicate ovarian tumors in vivo Oncoimmunology 4 3 e994446 https://doi.org/10.4161/2162402x.2014.994446
T Shum B Omer H Tashiro RL Kruse DL Wagner K Parikh Z Yi T Sauer D Liu R Parihar P Castillo H Liu MK Brenner LS Metelitsa S Gottschalk CM Rooney 2017 Constitutive signaling from an engineered IL7 receptor promotes durable tumor elimination by tumor-redirected T cells Cancer Discov 7 11 1238 1247 https://doi.org/10.1158/2159-8290.CD-17-0538
M Nakajima Y Sakoda K Adachi H Nagano K Tamada 2019 Improved survival of chimeric antigen receptor-engineered T (CAR-T) and tumor-specific T cells caused by anti-programmed cell death protein 1 single-chain variable fragment-producing CAR-T cells Cancer Sci 110 10 3079 3088 https://doi.org/10.1111/cas.14169
W Hu Z Zi Y Jin G Li K Shao Q Cai X Ma F Wei 2019 CRISPR/Cas9-mediated PD-1 disruption enhances human mesothelin-targeted CAR T cell effector functions Cancer Immunol Immunother 68 3 365 377 https://doi.org/10.1007/s00262-018-2281-2
CC Kloss J Lee A Zhang F Chen JJ Melenhorst SF Lacey MV Maus JA Fraietta Y Zhao CH June 2018 Dominant-negative TGF-beta receptor enhances PSMA-targeted human CAR T cell proliferation and augments prostate cancer eradication Mol Ther 26 7 1855 1866 https://doi.org/10.1016/j.ymthe.2018.05.003
Y Kagoya S Tanaka T Guo M Anczurowski CH Wang K Saso MO Butler MD Minden N Hirano 2018 A novel chimeric antigen receptor containing a JAK-STAT signaling domain mediates superior antitumor effects Nat Med https://doi.org/10.1038/nm.4478
K Adachi Y Kano T Nagai N Okuyama Y Sakoda K Tamada 2018 IL-7 and CCL19 expression in CAR-T cells improves immune cell infiltration and CAR-T cell survival in the tumor Nat Biotechnol https://doi.org/10.1038/nbt.4086
SA Luther A Bidgol DC Hargreaves A Schmidt Y Xu J Paniyadi M Matloubian JG Cyster 2002 Differing activities of homeostatic chemokines CCL19, CCL21, and CXCL12 in lymphocyte and dendritic cell recruitment and lymphoid neogenesis J Immunol 169 1 424 433
A Link TK Vogt S Favre MR Britschgi H Acha-Orbea B Hinz JG Cyster SA Luther 2007 Fibroblastic reticular cells in lymph nodes regulate the homeostasis of naive T cells Nat Immunol 8 11 1255 1265 https://doi.org/10.1038/ni1513
L Bergan JA Gross B Nevin N Urban N Scholler 2007 Development and in vitro validation of anti-mesothelin biobodies that prevent CA125/Mesothelin-dependent cell attachment Cancer Lett 255 2 263 274 https://doi.org/10.1016/j.canlet.2007.04.012
E Lanitis M Poussin IS Hagemann G Coukos R Sandaltzopoulos N Scholler DJ Powell Jr 2012 Redirected antitumor activity of primary human lymphocytes transduced with a fully human anti-mesothelin chimeric receptor Mol Ther 20 3 633 643 https://doi.org/10.1038/mt.2011.256
RA Morgan JC Yang M Kitano ME Dudley CM Laurencot SA Rosenberg 2010 Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2 Mol Ther 18 4 843 851 https://doi.org/10.1038/mt.2010.24
K Tamada D Geng Y Sakoda N Bansal R Srivastava Z Li E Davila 2012 Redirecting gene-modified T cells toward various cancer types using tagged antibodies Clin Cancer Res: Off J Am Assoc Cancer Res 18 23 6436 6445 https://doi.org/10.1158/1078-0432.CCR-12-1449
M Ruella MV Maus 2016 Catch me if you can: leukemia escape after CD19-directed T Cell immunotherapies Comput Struct Biotechnol J 14 357 362 https://doi.org/10.1016/j.csbj.2016.09.003
A Rodriguez-Garcia A Palazon E Noguera-Ortega DJ Powell Jr S Guedan 2020 CAR-T cells hit the tumor microenvironment: strategies to overcome tumor escape Front Immunol 11 1109 https://doi.org/10.3389/fimmu.2020.01109
J Cheng L Zhao Y Zhang Y Qin Y Guan T Zhang C Liu J Zhou 2019 Understanding the mechanisms of resistance to CAR T-Cell therapy in malignancies Front Oncol 9 1237 https://doi.org/10.3389/fonc.2019.01237
CE Brown B Badie ME Barish L Weng JR Ostberg WC Chang A Naranjo R Starr J Wagner C Wright Y Zhai JR Bading JA Ressler J Portnow M D'Apuzzo SJ Forman MC Jensen 2015 Bioactivity and safety of IL13Ralpha2-redirected chimeric antigen receptor CD8+ T cells in patients with recurrent glioblastoma Clin Cancer Res: Off J Am Assoc Cancer Res 21 18 4062 4072 https://doi.org/10.1158/1078-0432.Ccr-15-0428
DM Orourke MP Nasrallah A Desai JJ Melenhorst K Mansfield JJD Morrissette M Martinezlage S Brem E Maloney A Shen R Isaacs S Mohan G Plesa SF Lacey JM Navenot Z Zheng BL Levine H Okada CH June JL Brogdon MV Maus 2017 A single dose of peripherally infused EGFRvIII-directed CAR T cells mediates antigen loss and induces adaptive resistance in patients with recurrent glioblastoma Sci Transl Med https://doi.org/10.1126/scitranslmed.aaa0984
J Jung HS Seol S Chang 2018 The generation and application of patient-derived xenograft model for cancer research Cancer Res Treat 50 1 1 10 https://doi.org/10.4143/crt.2017.307
TT Byrd K Fousek A Pignata C Szot H Samaha S Seaman L Dobrolecki VS Salsman HZ Oo K Bielamowicz D Landi N Rainusso J Hicks S Powell ML Baker WS Wels J Koch PH Sorensen B Deneen MJ Ellis MT Lewis M Hegde BS Fletcher B St Croix N Ahmed 2018 TEM8/ANTXR1-specific CAR T cells as a targeted therapy for triple-negative breast cancer Cancer Res 78 2 489 500 https://doi.org/10.1158/0008-5472.CAN-16-1911
C Deng J Zhao S Zhou J Dong J Cao J Gao Y Bai H Deng 2020 The vascular disrupting agent CA4P improves the antitumor efficacy of CAR-T cells in preclinical models of solid human tumors Mol Ther 28 1 75 88 https://doi.org/10.1016/j.ymthe.2019.10.010
Z Hu X Zheng D Jiao Y Zhou R Sun B Wang Z Tian H Wei 2020 LunX-CAR T cells as a targeted therapy for non-small cell lung cancer Mol Ther Oncolytics 17 361 370 https://doi.org/10.1016/j.omto.2020.04.008
H Jiang B Song P Wang B Shi Q Li M Fan S Di J Yang Z Li 2017 Efficient growth suppression in pancreatic cancer PDX model by fully human anti-mesothelin CAR-T cells Prot Cell 8 12 926 931 https://doi.org/10.1007/s13238-017-0472-9
Z Jiang X Jiang S Chen Y Lai X Wei B Li S Lin S Wang Q Wu Q Liang Q Liu M Peng F Yu J Weng X Du D Pei P Liu Y Yao P Xue P Li 2016 Anti-GPC3-CAR T cells suppress the growth of tumor cells in patient-derived xenografts of hepatocellular carcinoma Front Immunol 7 690 https://doi.org/10.3389/fimmu.2016.00690
N Tang C Cheng X Zhang M Qiao N Li W Mu XF Wei W Han H Wang 2020 TGF-beta inhibition via CRISPR promotes the long-term efficacy of CAR T cells against solid tumors JCI Insight https://doi.org/10.1172/jci.insight.133977
X Wei Y Lai J Li L Qin Y Xu R Zhao B Li S Lin S Wang Q Wu Q Liang M Peng F Yu Y Li X Zhang Y Wu P Liu D Pei Y Yao P Li 2017 PSCA and MUC1 in non-small-cell lung cancer as targets of chimeric antigen receptor T cells Oncoimmunology 6 3 e1284722 https://doi.org/10.1080/2162402X.2017.1284722
R Zhao L Cheng Z Jiang X Wei B Li Q Wu S Wang S Lin Y Long X Zhang Y Wu X Du D Pei P Liu Y Li S Cui Y Yao P Li 2019 DNAX-activating protein 10 co-stimulation enhances the anti-tumor efficacy of chimeric antigen receptor T cells Oncoimmunology 8 1 e1509173 https://doi.org/10.1080/2162402X.2018.1509173
NC Walsh LL Kenney S Jangalwe KE Aryee DL Greiner MA Brehm LD Shultz 2017 Humanized mouse models of clinical disease Annu Rev Pathol 12 187 215 https://doi.org/10.1146/annurev-pathol-052016-100332
Acknowledgment
The authors would thank Drs. Daisuke Umezu, Jun Mori, Takahiro Sasaki, Yasunori Iida for excellent advices on the study. The authors would also thank Hiromi Kurosawa, Mihoko Ida, Nana Okada, Makiko Miyamoto, Satoshi Tatekabe, Nanami Nakamura, and Chisaki Mochida for their excellent technical supports.
Funding
This study was supported by Practical Research for Innovative Cancer Control, and Project for Cancer Research and Therapeutic Evolution (P-CREATE) 16770206 (to K.T.) by Japan Agency for Medical Research and Development (AMED), and Japan Society for the Promotion of Science Grant-in-Aid for Scientific Research (C) Grant Number 19K07625 (to Yu.S.).
Author information
Authors and Affiliations
Contributions
S.G., Yu.S., K.A. and K.T. designed the study and interpreted the data; S.G. performed the experiments and analyzed the data; Yo.S. and S.Y. provided the materials; M.E. and K.T. supervised the study; S.G. and K.T. wrote the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
K.T. and Yu.S. hold stocks of Noile-Immune Biotech Inc., and receive remuneration from Noile-Immune Biotech Inc. Other authors declare no conflict of interest.
Ethical approval
All animal procedures were approved by the Institutional Animal Care and Use Committee of the Yamaguchi University. Human PBMCs were derived from healthy volunteers who gave a written informed consent. Patient-derived xenograft tumor was derived from a pancreatic cancer patient who gave written informed consent. This study was approved by the ethics committee of the Yamaguchi University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Goto, S., Sakoda, Y., Adachi, K. et al. Enhanced anti-tumor efficacy of IL-7/CCL19-producing human CAR-T cells in orthotopic and patient-derived xenograft tumor models. Cancer Immunol Immunother 70, 2503–2515 (2021). https://doi.org/10.1007/s00262-021-02853-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-021-02853-3