Abstract
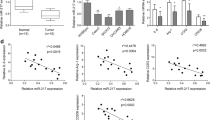
The microRNA let-7d has been reported to be a tumor suppressor in renal cell carcinoma (RCC). Tumor-associated macrophages (TAM) are M2-polarized macrophages that can enhance tumor growth and angiogenesis in many human cancers. However, the role of let-7d in TAM-associated RCC progression remains elusive. First, we observed a strongly inverse correlation between let-7d expression and microvessel density in RCC tissues. Furthermore, the proliferation, migration, and tube formation of HUVECs were significantly inhibited by conditioned medium from a coculture system of the phorbol myristate acetate pretreated human THP-1 macrophages and let-7d-overexpressing RCC cells. Moreover, the proportion of M2 macrophages was significantly lower in the group that was cocultured with let-7d-overexpressing RCC cells. Subcutaneous xenografts formed by the injection of let-7d-overexpressing RCC cells together with THP-1 cells resulted in a significant decrease in the M2 macrophage ratio and microvessel density compared with those formed by the injection of control RCC cells with THP-1 cells. In silico and experimental analysis revealed interleukin-10 (IL-10) and IL-13 as let-7d target genes. Importantly, the addition of IL-10 and IL-13 counteracted the inhibitory effects of the conditioned medium from the coculture system with let-7d-overexpressing RCC cells in vitro. Additionally, overexpression of IL-10 and IL-13 reversed the effects of let-7d on macrophage M2 polarization and tumor angiogenesis in vivo. Finally, the expression of IL-10 and IL-13 were inversely correlated with the expression of let-7d in RCC clinical specimens. These results suggest that let-7d may inhibit intratumoral macrophage M2 polarization and subsequent tumor angiogenesis by targeting IL-10 and IL-13.







Similar content being viewed by others
Data availability
All data supporting the conclusions of this article are included in the article.
Change history
14 February 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00262-023-03389-4
Abbreviations
- ccRCC:
-
Clear cell renal cell carcinoma
- EGF:
-
Epidermal growth factor
- FGF:
-
Fibroblast growth factor
- HUVEC:
-
Human umbilical vein endothelial cell
- KICH:
-
Kidney chromophobe
- KIRC:
-
Kidney renal clear cell carcinoma
- KIRP:
-
Kidney renal papillary cell carcinoma
- MVD:
-
Microvessel density
- PDGF:
-
Platelet-derived growth factor
- PMA:
-
Phorbol myristate acetate
- PIGF:
-
Placental growth factor
- qRT–PCR:
-
Quantitative reverse transcription–polymerase chain reaction
- RCC:
-
Renal cell carcinoma
- TAM:
-
Tumor-associated macrophage
- TCGA:
-
The cancer genome atlas
- TGF-β:
-
Transforming growth factor-β
- TNF-α:
-
Tumor necrosis factor α
- 3′-UTR:
-
3′-Untranslated regions
- VEGF:
-
Vascular endothelial growth factor
References
McDermott DF, Huseni MA, Atkins MB et al (2018) Clinical activity and molecular correlates of response to atezolizumab alone or in combination with bevacizumab versus sunitinib in renal cell carcinoma. Nat Med 24(6):749–757
Viallard C, Larrivee B (2017) Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis 20(4):409–426
Mantovani A, Sozzani S, Locati M et al (2002) Macrophage polarization: tumor-associated macrophages as a paradigm for polarized M2 mononuclear phagocytes. Trends Immunol 23(11):549–555
Lu G, Zhang R, Geng S et al (2015) Myeloid cell-derived inducible nitric oxide synthase suppresses M1 macrophage polarization. Nat Commun 6:6676
Murray PJ, Allen JE, Biswas SK et al (2014) Macrophage activation and polarization: nomenclature and experimental guidelines. Immunity 41(1):14–20
Tjiu JW, Chen JS, Shun CT et al (2009) Tumor-associated macrophage-induced invasion and angiogenesis of human basal cell carcinoma cells by cyclooxygenase-2 induction. J Invest Dermatol 129(4):1016–1025
Erreni M, Mantovani A, Allavena P (2011) Tumor-associated Macrophages (TAM) and Inflammation in Colorectal Cancer. Cancer Microenviron 4(2):141–154
Pollard JW (2004) Tumour-educated macrophages promote tumour progression and metastasis. Nat Rev Cancer 4(1):71–78
Qian BZ, Pollard JW (2010) Macrophage diversity enhances tumor progression and metastasis. Cell 141(1):39–51
Xu L, Zhu Y, Chen L et al (2014) Prognostic value of diametrically polarized tumor-associated macrophages in renal cell carcinoma. Ann Surg Oncol 21(9):3142–3150
Su B, Zhao W, Shi B et al (2014) Let-7d suppresses growth, metastasis, and tumor macrophage infiltration in renal cell carcinoma by targeting COL3A1 and CCL7. Mol Cancer 13:206
Miyake M, Hori S, Morizawa Y et al (2016) CXCL1-mediated interaction of cancer cells with tumor-associated macrophages and cancer-associated fibroblasts promotes tumor progression in human bladder cancer. Neoplasia 18(10):636–646
Desai N, Ludgin J, Goldberg J et al (2013) Development of a xeno-free non-contact co-culture system for derivation and maintenance of embryonic stem cells using a novel human endometrial cell line. J Assist Reprod Genet 30(5):609–615
Wang D, Stockard CR, Harkins L et al (2008) Immunohistochemistry in the evaluation of neovascularization in tumor xenografts. Biotech Histochem 83(3–4):179–189
Denton AE, Roberts EW, Fearon DT (2018) Stromal cells in the tumor microenvironment. Adv Exp Med Biol 1060:99–114
Tuna B, Yorukoglu K, Unlu M et al (2006) Association of mast cells with microvessel density in renal cell carcinomas. Eur Urol 50(3):530–534
Jones J, Otu H, Spentzos D et al (2005) Gene signatures of progression and metastasis in renal cell cancer. Clin Cancer Res 11(16):5730–5739
Li W, Zhu W, Che J et al (2013) Microarray profiling of human renal cell carcinoma: identification for potential biomarkers and critical pathways. Kidney Blood Press Res 37(4–5):506–513
Chevrier S, Levine JH, Zanotelli V et al (2017) An immune atlas of clear cell renal cell carcinoma. Cell 169(4):736–749
Yang Z, Xie H, He D et al (2016) Infiltrating macrophages increase RCC epithelial mesenchymal transition (EMT) and stem cell-like populations via AKT and mTOR signaling. Oncotarget 7(28):44478–44491
Toge H, Inagaki T, Kojimoto Y et al (2009) Angiogenesis in renal cell carcinoma: the role of tumor-associated macrophages. Int J Urol 16(10):801–807
Fischer C, Jonckx B, Mazzone M et al (2007) Anti-PlGF inhibits growth of VEGF(R)-inhibitor-resistant tumors without affecting healthy vessels. Cell 131(3):463–475
Tan HY, Wang N, Man K et al (2015) Autophagy-induced RelB/p52 activation mediates tumour-associated macrophage repolarisation and suppression of hepatocellular carcinoma by natural compound baicalin. Cell Death Dis 6:e1942
Jin H, He Y, Zhao P et al (2019) Targeting lipid metabolism to overcome EMT-associated drug resistance via integrin beta3/FAK pathway and tumor-associated macrophage repolarization using legumain-activatable delivery. Theranostics 9(1):265–278
Ohba K, Miyata Y, Matsuo T et al (2014) High expression of Twist is associated with tumor aggressiveness and poor prognosis in patients with renal cell carcinoma. Int J Clin Exp Pathol 7(6):3158–3165
Lee JH, Lee GT, Woo SH et al (2013) BMP-6 in renal cell carcinoma promotes tumor proliferation through IL-10-dependent M2 polarization of tumor-associated macrophages. Cancer Res 73(12):3604–3614
Barros MH, Hauck F, Dreyer JH et al (2013) Macrophage polarisation: an immunohistochemical approach for identifying M1 and M2 macrophages. PLoS ONE 8(11):e80908
Rakaee M, Busund LR, Jamaly S et al (2019) Prognostic value of macrophage phenotypes in resectable non-small cell lung cancer assessed by multiplex immunohistochemistry. Neoplasia 21(3):282–293
Ichimura T, Morikawa T, Kawai T et al (2014) Prognostic significance of CD204-positive macrophages in upper urinary tract cancer. Ann Surg Oncol 21(6):2105–2112
Kawachi A, Yoshida H, Kitano S et al (2018) Tumor-associated CD204(+) M2 macrophages are unfavorable prognostic indicators in uterine cervical adenocarcinoma. Cancer Sci 109(3):863–870
Wittke F, Hoffmann R, Buer J et al (1999) Interleukin 10 (IL-10): an immunosuppressive factor and independent predictor in patients with metastatic renal cell carcinoma. Br J Cancer 79(7–8):1182–1184
Daurkin I, Eruslanov E, Stoffs T et al (2011) Tumor-associated macrophages mediate immunosuppression in the renal cancer microenvironment by activating the 15-lipoxygenase-2 pathway. Cancer Res 71(20):6400–6409
Roszer T (2015) Understanding the mysterious M2 macrophage through activation markers and effector mechanisms. Mediators Inflamm 2015:816460
Hallett MA, Venmar KT, Fingleton B et al (2012) Cytokine stimulation of epithelial cancer cells: the similar and divergent functions of IL-4 and IL-13. Cancer Res 72(24):6338–6343
Terabe M, Matsui S, Noben-Trauth N et al (2000) NKT cell-mediated repression of tumor immunosurveillance by IL-13 and the IL-4R-STAT6 pathway. Nat Immunol 1(6):515–520
Gabitass RF, Annels NE, Stocken DD et al (2011) Elevated myeloid-derived suppressor cells in pancreatic, esophageal and gastric cancer are an independent prognostic factor and are associated with significant elevation of the Th2 cytokine interleukin-13. Cancer Immunol Immunother 60(10):1419–1430
Chang Y, Xu L, An H et al (2015) Expression of IL-4 and IL-13 predicts recurrence and survival in localized clear-cell renal cell carcinoma. Int J Clin Exp Pathol 8(2):1594–1603
Satoh T, Takeuchi O, Vandenbon A et al (2010) The Jmjd3-Irf4 axis regulates M2 macrophage polarization and host responses against helminth infection. Nat Immunol 11(10):936–944
Gao S, Zhou J, Liu N et al (2015) Curcumin induces M2 macrophage polarization by secretion IL-4 and/or IL-13. J Mol Cell Cardiol 85:131–139
Acknowledgements
The authors thank Yunfei Fan for the support and help for technical assistance.
Funding
This study was supported by Grants from the National Natural Science Foundation (Grant No.8170101445), the Beijing Natural Science Foundation (No.7182191) and the Capital Health Research and Development of Special (code: 2016-1-2241).
Author information
Authors and Affiliations
Contributions
Design: BS, HH, YG, WZ, GZ and LZ Experimental operation: BSu, HH, WH, XL, JY and JY. Acquisition of data and Analysis (acquired and managed patients’ information and statistical analysis): BS, HH, YG, XL and GZ. Writing, review, and/or revision of the manuscript: BS, HH, GZ and LZ. All authors read and approved the submitted manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval and consent to participate
All Procedures performed in studies involving human participants or animals were reviewed and granted by the Ethics Committee of Peking University First Hospital and the Ethics Committee of Tsinghua Changgung Hospital, Beijing, China. Informed consent was obtained from all the patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Su, B., Han, H., Gong, Y. et al. Let-7d inhibits intratumoral macrophage M2 polarization and subsequent tumor angiogenesis by targeting IL-13 and IL-10. Cancer Immunol Immunother 70, 1619–1634 (2021). https://doi.org/10.1007/s00262-020-02791-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02791-6