Abstract
Background
LncRNAs play an important role in the regulation of the killing effect of cytotoxic CD8 + T cells in various cancers. However, the role and underlying mechanisms of UCA1 in the killing effect of cytotoxic CD8 + T cells in anaplastic thyroid carcinoma (ATC) are not clear.
Methods
UCA1, miR-148a, and PD-L1 expression were detected by quantitative real-time PCR and/or Western blot. The ratio of PD-L1+ATC cells/ATC cells was determined using flow cytometry. The ability of CD8 + T cells to kill target ATC cells was detected by Chromium-51 (51Cr) release assay. The targeted relationship between UCA1 and miR-148a was confirmed by dual-luciferase reporter gene assay.
Results
UCA1 and PD-L1 expression levels were elevated in ATC tissues and cells. Silencing UCA1 and PD-L1 enhanced the killing effect of cytotoxic CD8 + T cells on ATC cells. UCA1 negatively regulated the expression of miR-148a, and miR-148a targeted PD-L1 to down-regulate its expression. Besides, we found that UCA1 attenuated the killing effect of cytotoxic CD8 + T cells and reduced cytokine secretion through PD-L1 and miR-148a. Finally, silencing UCA1 or PD-L1 in ATC cells restored the suppression of the killing effect of CD8 + T cells in vivo.
Conclusion
UCA1 attenuated the killing effect of cytotoxic CD8 + T cells on ATC cells through the miR-148a/PD-L1 pathway.






Similar content being viewed by others
References
Molinaro E et al (2017) Anaplastic thyroid carcinoma: from clinicopathology to genetics and advanced therapies. Nat Rev Endocrinol 13:644
Cao X et al (2019) Targeting super-enhancer-driven oncogenic transcription by CDK7 inhibition in anaplastic thyroid carcinoma. Thyroid 29(6):809–823
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144(5):646–674
Andrada E, Liébana R, Merida I (2017) Diacylglycerol kinase ζ limits cytokine-dependent expansion of CD8(+) T cells with broad antitumor capacity. EBioMedicine 19:39–48
Hodge G et al (2014) Lung cancer is associated with decreased expression of perforin, granzyme B and interferon (IFN)-γ by infiltrating lung tissue T cells, natural killer (NK) T-like and NK cells. Clin Exp Immunol 178(1):79–85
Goodman A, Patel SP, Kurzrock R (2017) PD-1-PD-L1 immune-checkpoint blockade in B-cell lymphomas. Nat Rev Clin Oncol 14(4):203–220
Frydenlund N, Mahalingam M (2017) PD-L1 and immune escape: insights from melanoma and other lineage-unrelated malignancies. Hum Pathol 66:13–33
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12(4):252–264
Han J, Hong Y, Lee YS (2017) PD-L1 expression and combined status of PD-L1/PD-1-positive tumor infiltrating mononuclear cell density predict prognosis in glioblastoma patients. J Pathol Transl Med 51(1):40–48
Bastman JJ et al (2016) Tumor-infiltrating T Cells and the PD-1 checkpoint pathway in advanced differentiated and anaplastic thyroid cancer. J Clin Endocrinol Metab 101(7):2863–2873
Lau J et al (2017) Tumour and host cell PD-L1 is required to mediate suppression of anti-tumour immunity in mice. Nat Commun 8:14572–14572
Brauner E et al (2016) Combining BRAF inhibitor and anti PD-L1 antibody dramatically improves tumor regression and anti tumor immunity in an immunocompetent murine model of anaplastic thyroid cancer. Oncotarget 7(13):17194–17211
Dong P et al (2019) Long noncoding RNA NEAT1 drives aggressive endometrial cancer progression via miR-361-regulated networks involving STAT3 and tumor microenvironment-related genes. J Exp Clin Cancer Res 38(1):295
Qian Y et al (2017) Upregulation of the long noncoding RNA UCA1 affects the proliferation, invasion, and survival of hypopharyngeal carcinoma. Mol Cancer 16(1):68
Li GY et al (2018) Long non-coding RNAs AC026904.1 and UCA1: a “one-two punch” for TGF-beta-induced SNAI2 activation and epithelial-mesenchymal transition in breast cancer. Theranostics 8(10):2846–2861
Yang Z et al (2018) Long non-coding RNA UCA1 upregulation promotes the migration of hypoxia-resistant gastric cancer cells through the miR-7-5p/EGFR axis. Exp Cell Res 368(2):194–201
He Z et al (2017) The lncRNA UCA1 interacts with miR-182 to modulate glioma proliferation and migration by targeting iASPP. Arch Biochem Biophys 623–624:1–8
Zuo ZK et al (2017) TGFbeta1-induced LncRNA UCA1 upregulation promotes gastric cancer invasion and migration. DNA Cell Biol 36(2):159–167
Wang CJ et al (2019) The lncRNA UCA1 promotes proliferation, migration, immune escape and inhibits apoptosis in gastric cancer by sponging anti-tumor miRNAs. Mol Cancer 18(1):115
Wang Y, Hou Z, Li D (2018) Long noncoding RNA UCA1 promotes anaplastic thyroid cancer cell proliferation via miR135amediated cmyc activation. Mol Med Rep 18(3):3068–3076
Shi D et al (2019) LncRNA AFAP1-AS1 promotes tumorigenesis and epithelial-mesenchymal transition of osteosarcoma through RhoC/ROCK1/p38MAPK/Twist1 signaling pathway. J Exp Clin Cancer Res 38(1):375
Zeng Z et al (2019) LncRNA-MTA2TR functions as a promoter in pancreatic cancer via driving deacetylation-dependent accumulation of HIF-1α. Theranostics 9(18):5298–5314
Li P et al (2019) ZNNT1 long noncoding RNA induces autophagy to inhibit tumorigenesis of uveal melanoma by regulating key autophagy gene expression. Autophagy 16(7):1186–1199
Yu Z et al (2019) Long non-coding RNA FENDRR acts as a miR-423-5p sponge to suppress the Treg-mediated immune escape of hepatocellular carcinoma cells. Mol Ther Nucleic Acids 17:516–529
Hu Q et al (2019) Oncogenic lncRNA downregulates cancer cell antigen presentation and intrinsic tumor suppression. Nat Immunol 20(7):835–851
Yan K et al (2019) Repression of lncRNA NEAT1 enhances the antitumor activity of CD8(+)T cells against hepatocellular carcinoma via regulating miR-155/Tim-3. Int J Biochem Cell Biol 110:1–8
Mari L et al (2018) microRNA 125a regulates MHC-I expression on esophageal adenocarcinoma cells, associated with suppression of antitumor immune response and poor outcomes of patients. Gastroenterology 155(3):784–798
Friedrich M et al (2017) The role of the miR-148/-152 family in physiology and disease. Eur J Immunol 47(12):2026–2038
Han C et al (2017) Downregulation of cyclin-dependent kinase 8 by microRNA-148a suppresses proliferation and invasiveness of papillary thyroid carcinomas. Am J Cancer Res 7(10):2081–2090
Feng M et al (2017) PD-1/PD-L1 and immunotherapy for pancreatic cancer. Cancer Lett 407:57–65
Inman BA et al (2017) Atezolizumab: A PD-L1-blocking antibody for bladder cancer. Clin Cancer Res 23(8):1886–1890
Ahn S et al (2017) Comprehensive screening for PD-L1 expression in thyroid cancer. Endocr Relat Cancer 24(2):97–106
Cantara S et al (2019) Blockade of the programmed death ligand 1 (PD-L1) as potential therapy for anaplastic thyroid cancer. Endocrine 64(1):122–129
Osada T et al (2015) CEA/CD3-bispecific T cell-engaging (BiTE) antibody-mediated T lymphocyte cytotoxicity maximized by inhibition of both PD1 and PD-L1. Cancer Immunol Immunother 64(6):677–688
Martínez-Lostao L, Anel A, Pardo J (2015) How do cytotoxic lymphocytes kill cancer cells? Clin Cancer Res 21(22):5047–5056
Gao L et al (2019) MiR-873/PD-L1 axis regulates the stemness of breast cancer cells. EBioMedicine 41:395–407
Chen Y et al (2019) LIN28/let-7/PD-L1 pathway as a target for cancer immunotherapy. Cancer Immunol Res 7(3):487–497
Funding
None.
Author information
Authors and Affiliations
Contributions
XW designed this experiment and performed the experiments. XW and YZ edited this manuscript. YZ and JZ collected data and performed the statistical analysis. JZ, CY and MT revised this manuscript. XL made substantial contributions to the conception, design, and critical revision of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no actual or potential conflicts of interest to declare.
Ethical approval
The study was approved by the ethics committee of The First Affiliated Hospital of Zhengzhou University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
262_2020_2753_MOESM2_ESM.tif
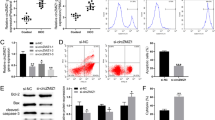
Supplementary file2 Supplementary Fig. 2 The effect of miR-148a on the expression of UCAI in cells and the verification of the interaction between UCA1 and miR-148a. A. Hth74 and 8505C cells were transfected with miR-148a mimic, UCA1 expression was detected by qRT-PCR. *P < 0.05, **P < 0.01 vs. NC. B. The interaction between UCA1 and miR-148a was detected by RNA pull-down assay. **P < 0.01 vs. Bio-NC-probe. NC: negative control (TIF 130 kb)
Rights and permissions
About this article
Cite this article
Wang, X., Zhang, Y., Zheng, J. et al. LncRNA UCA1 attenuated the killing effect of cytotoxic CD8 + T cells on anaplastic thyroid carcinoma via miR-148a/PD-L1 pathway. Cancer Immunol Immunother 70, 2235–2245 (2021). https://doi.org/10.1007/s00262-020-02753-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02753-y