Abstract
Purpose
To develop a radiomics-based hepatocellular carcinoma (HCC) grade classifier model based on data from gadoxetic acid-enhanced MRI.
Methods
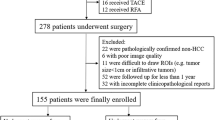
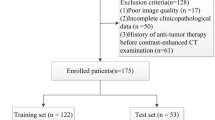
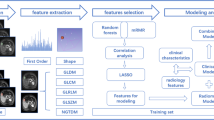
This retrospective study included 137 patients who underwent hepatectomy for a single HCC and gadoxetic acid-enhanced MRI within 60 days before surgery. HCC grade was categorized as low or high (modified Edmondson–Steiner grade I–II vs. III–IV). We used the hepatobiliary phase (HBP), portal venous phase, T2-weighted image(T2WI), and T1-weighted image(T1WI). From the volume of interest in HCC, 833 radiomic features were extracted. Radiomic and clinical features were selected using a random forest regressor, and the classification model was trained and validated using a random forest classifier and tenfold stratified cross-validation. Eight models were developed using the radiomic features alone or by combining the radiomic and clinical features. Models were validated with internal enrolled data (internal validation) and a dataset (28 patients) at a separate institution (external validation). The area under the curve (AUC) of the validation results was compared using the DeLong test.
Results
In internal and external validation, the HBP radiomics-only model showed the highest AUC (internal 0.80 ± 0.09, external 0.70 ± 0.09). In external validation, all models showed lower AUC than those for internal validation, while the T2WI and T1WI models failed to predict the HCC grade (AUC 0.30–0.58) in contrast to the internal validation results (AUC 0.67–0.78).
Conclusion
The radiomics-based machine learning model from gadoxetic acid-enhanced liver MRI could distinguish between low- and high-grade HCCs. The radiomics-only HBP model showed the best AUC among the eight models, good performance in internal validation, and fair performance in external validation.
Graphical abstract






Similar content being viewed by others
Data availability
The datasets generated or analyzed during the study are not publicly available because they contain information that could compromise research participant privacy but are available from the corresponding author upon reasonable request.
References
Amado V, Rodríguez-Perálvarez M, Ferrín G, De la Mata M (2019) Selecting patients with hepatocellular carcinoma for liver transplantation: incorporating tumor biology criteria. J Hepatocell Carcinoma 6:1-10. https://doi.org/10.2147/jhc.S174549
European Association for the Study of the Liver (2018) EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol 69:182-236. https://doi.org/10.1016/j.jhep.2018.03.019
Korean Liver Cancer Association (KLCA); National Cancer Center (NCC) G, Korea (2019) 2018 Korean Liver Cancer Association-National Cancer Center Korea Practice Guidelines for the Management of Hepatocellular Carcinoma. Korean J Radiol 20:1042-1113. https://doi.org/10.3348/kjr.2019.0140
Zhou YM, Yang JM, Li B, Yin ZF, Xu F, Wang B, Xu W, Kan T (2010) Risk factors for early recurrence of small hepatocellular carcinoma after curative resection. Hepatobiliary Pancreat Dis Int 9:33-37
Jung KS, Kim SU, Choi GH, Park JY, Park YN, Kim DY, Ahn SH, Chon CY, Kim KS, Choi EH, Choi JS, Han KH (2012) Prediction of recurrence after curative resection of hepatocellular carcinoma using liver stiffness measurement (FibroScan®). Ann Surg Oncol 19:4278-4286. https://doi.org/10.1245/s10434-012-2422-3
Park MS, Lee KW, Kim H, Choi YR, Hong G, Yi NJ, Suh KS (2017) Primary Living-donor Liver Transplantation Is Not the Optimal Treatment Choice in Patients With Early Hepatocellular Carcinoma With Poor Tumor Biology. Transplant Proc 49:1103-1108. https://doi.org/10.1016/j.transproceed.2017.03.016
Oishi K, Itamoto T, Amano H, Fukuda S, Ohdan H, Tashiro H, Shimamoto F, Asahara T (2007) Clinicopathologic features of poorly differentiated hepatocellular carcinoma. J Surg Oncol 95:311-316. https://doi.org/10.1002/jso.20661
Okusaka T, Okada S, Ueno H, Ikeda M, Shimada K, Yamamoto J, Kosuge T, Yamasaki S, Fukushima N, Sakamoto M (2002) Satellite lesions in patients with small hepatocellular carcinoma with reference to clinicopathologic features. Cancer 95:1931-1937. https://doi.org/10.1002/cncr.10892
Weng S, Xu X, Li Y, Yan C, Chen J, Ye R, Zhu Y, Wen L, Hong J (2020) Quantitative analysis of multiphase magnetic resonance images may assist prediction of histopathological grade of small hepatocellular carcinoma. Ann Transl Med 8:1023. https://doi.org/10.21037/atm-20-2874
An C, Park MS, Jeon HM, Kim YE, Chung WS, Chung YE, Kim MJ, Kim KW (2012) Prediction of the histopathological grade of hepatocellular carcinoma using qualitative diffusion-weighted, dynamic, and hepatobiliary phase MRI. Eur Radiol 22:1701-1708. https://doi.org/10.1007/s00330-012-2421-6
Yoneda N, Matsui O, Kobayashi S, Kitao A, Kozaka K, Inoue D, Yoshida K, Minami T, Koda W, Gabata T (2019) Current status of imaging biomarkers predicting the biological nature of hepatocellular carcinoma. Jpn J Radiol 37:191-208. https://doi.org/10.1007/s11604-019-00817-3
Surov A, Pech M, Omari J, Fischbach F, Damm R, Fischbach K, Powerski M, Relja B, Wienke A (2021) Diffusion-Weighted Imaging Reflects Tumor Grading and Microvascular Invasion in Hepatocellular Carcinoma. Liver Cancer 10:10-24. https://doi.org/10.1159/000511384
Mao B, Zhang L, Ning P, Ding F, Wu F, Lu G, Geng Y, Ma J (2020) Preoperative prediction for pathological grade of hepatocellular carcinoma via machine learning-based radiomics. Eur Radiol 30:6924-6932. https://doi.org/10.1007/s00330-020-07056-5
Liu QP, Xu X, Zhu FP, Zhang YD, Liu XS (2020) Prediction of prognostic risk factors in hepatocellular carcinoma with transarterial chemoembolization using multi-modal multi-task deep learning. EClinicalMedicine 23:100379. https://doi.org/10.1016/j.eclinm.2020.100379
Oh J, Lee JM, Park J, Joo I, Yoon JH, Lee DH, Ganeshan B, Han JK (2019) Hepatocellular Carcinoma: Texture Analysis of Preoperative Computed Tomography Images Can Provide Markers of Tumor Grade and Disease-Free Survival. Korean J Radiol 20:569-579. https://doi.org/10.3348/kjr.2018.0501
Kim YY, Park MS, Aljoqiman KS, Choi JY, Kim MJ (2019) Gadoxetic acid-enhanced magnetic resonance imaging: Hepatocellular carcinoma and mimickers. Clin Mol Hepatol 25:223-233. https://doi.org/10.3350/cmh.2018.0107
Wei J, Jiang H, Zeng M et al (2021) Prediction of Microvascular Invasion in Hepatocellular Carcinoma via Deep Learning: A Multi-Center and Prospective Validation Study. Cancers (Basel) 13:2368. https://doi.org/10.3390/cancers13102368
Yu Y, Fan Y, Wang X, Zhu M, Hu M, Shi C, Hu C (2022) Gd-EOB-DTPA-enhanced MRI radiomics to predict vessels encapsulating tumor clusters (VETC) and patient prognosis in hepatocellular carcinoma. Eur Radiol 32:959-970. https://doi.org/10.1007/s00330-021-08250-9
Tian Y, Hua H, Peng Q, Zhang Z, Wang X, Han J, Ma W, Chen J (2022) Preoperative Evaluation of Gd-EOB-DTPA-Enhanced MRI Radiomics-Based Nomogram in Small Solitary Hepatocellular Carcinoma (≤3 cm) With Microvascular Invasion: A Two-Center Study. J Magn Reson Imaging. https://doi.org/10.1002/jmri.28157
Zhou W, Zhang L, Wang K, Chen S, Wang G, Liu Z, Liang C (2017) Malignancy characterization of hepatocellular carcinomas based on texture analysis of contrast-enhanced MR images. J Magn Reson Imaging 45:1476-1484. https://doi.org/10.1002/jmri.25454
Choi JM, Yu JS, Cho ES, Kim JH, Chung JJ (2020) Texture Analysis of Hepatocellular Carcinoma on Magnetic Resonance Imaging: Assessment for Performance in Predicting Histopathologic Grade. J Comput Assist Tomogr 44:901-910. https://doi.org/10.1097/rct.0000000000001087
Mao Y, Wang J, Zhu Y, Chen J, Mao L, Kong W, Qiu Y, Wu X, Guan Y, He J (2022) Gd-EOB-DTPA-enhanced MRI radiomic features for predicting histological grade of hepatocellular carcinoma. Hepatobiliary Surg Nutr 11:13–24. https://doi.org/10.21037/hbsn-19-870
Hectors SJ, Lewis S, Besa C et al (2020) MRI radiomics features predict immuno-oncological characteristics of hepatocellular carcinoma. Eur Radiol 30:3759-3769. https://doi.org/10.1007/s00330-020-06675-2
Chen S, Feng S, Wei J, Liu F, Li B, Li X, Hou Y, Gu D, Tang M, Xiao H, Jia Y, Peng S, Tian J, Kuang M (2019) Pretreatment prediction of immunoscore in hepatocellular cancer: a radiomics-based clinical model based on Gd-EOB-DTPA-enhanced MRI imaging. Eur Radiol 29:4177-4187. https://doi.org/10.1007/s00330-018-5986-x
Taylor JM, Ankerst DP, Andridge RR (2008) Validation of biomarker-based risk prediction models. Clin Cancer Res 14:5977-5983. https://doi.org/10.1158/1078-0432.Ccr-07-4534
Park SH, Choi J, Byeon JS (2021) Key Principles of Clinical Validation, Device Approval, and Insurance Coverage Decisions of Artificial Intelligence. Korean J Radiol 22:442-453. https://doi.org/10.3348/kjr.2021.0048
Cho Y, Han YE, Kim MJ, Park BJ, Sim KC, Sung DJ, Han NY, Park YS (2022) Computer-aided hepatocellular carcinoma detection on the hepatobiliary phase of gadoxetic acid-enhanced magnetic resonance imaging using a convolutional neural network: feasibility evaluation with multi-sequence data. Comput Methods Programs Biomed 225:107032. https://doi.org/10.1016/j.cmpb.2022.107032
Edmondson HA, Steiner PE (1954) Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer 7:462-503. https://doi.org/10.1002/1097-0142(195405)7:3<462::aid-cncr2820070308>3.0.co;2-e
van Griethuysen JJM, Fedorov A, Parmar C, Hosny A, Aucoin N, Narayan V, Beets-Tan RGH, Fillion-Robin JC, Pieper S, Aerts HJWL (2017) Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res 77:e104-e107. https://doi.org/10.1158/0008-5472.Can-17-0339
Pyradiomics community (2021) Welcome to pyradiomics documentation! https://pyradiomics.readthedocs.io/en/latest/index.html. Accessed 20 July 2021
Breiman L (2001) Random forests. Mach Learn 45:5-32. https://doi.org/10.1023/A:1010933404324
Thompson B (2019) A limitation of Random Forest Regression. https://towardsdatascience.com/a-limitation-of-random-forest-regression-db8ed7419e9f. Accessed 06 January 2022
Li F, He H (2018) Assessing the Accuracy of Diagnostic Tests. Shanghai Arch Psychiatry 30:207–212. https://doi.org/10.11919/j.issn.1002-0829.218052
Asayama Y, Yoshimitsu K, Nishihara Y, Irie H, Aishima S, Taketomi A, Honda H (2008) Arterial Blood Supply of Hepatocellular Carcinoma and Histologic Grading: Radiologic-Pathologic Correlation. AJR Am J Roentgenol 190:W28-W34. https://doi.org/10.2214/AJR.07.2117
Kitao A, Matsui O, Yoneda N, Kozaka K, Shinmura R, Koda W, Kobayashi S, Gabata T, Zen Y, Yamashita T, Kaneko S, Nakanuma Y (2011) The uptake transporter OATP8 expression decreases during multistep hepatocarcinogenesis: correlation with gadoxetic acid enhanced MR imaging. Eur Radiol 21:2056-2066. https://doi.org/10.1007/s00330-011-2165-8
Kadoya M, Matsui O, Takashima T, Nonomura A (1992) Hepatocellular carcinoma: correlation of MR imaging and histopathologic findings. Radiology 183:819-825. https://doi.org/10.1148/radiology.183.3.1316622
Enomoto S, Tamai H, Shingaki N, Mori Y, Moribata K, Shiraki T, Deguchi H, Ueda K, Inoue I, Maekita T, Iguchi M, Yanaoka K, Oka M, Ichinose M (2011) Assessment of hepatocellular carcinomas using conventional magnetic resonance imaging correlated with histological differentiation and a serum marker of poor prognosis. Hepatol Int 5:730-737. https://doi.org/10.1007/s12072-010-9245-8
Ebara M, Fukuda H, Kojima Y, Morimoto N, Yoshikawa M, Sugiura N, Satoh T, Kondo F, Yukawa M, Matsumoto T, Saisho H (1999) Small Hepatocellular Carcinoma: Relationship of Signal Intensity to Histopathologic Findings and Metal Content of the Tumor and Surrounding Hepatic Parenchyma. Radiology 210:81-88. https://doi.org/10.1148/radiology.210.1.r99ja4181
Li X, Zhang K, Shi Y, Wang F, Meng X (2016) Correlations between the minimum and mean apparent diffusion coefficient values of hepatocellular carcinoma and tumor grade. J Magn Reson Imaging 44:1442-1447. https://doi.org/10.1002/jmri.25323
Wu M, Tan H, Gao F, Hai J, Ning P, Chen J, Zhu S, Wang M, Dou S, Shi D (2019) Predicting the grade of hepatocellular carcinoma based on non-contrast-enhanced MRI radiomics signature. Eur Radiol 29:2802-2811. https://doi.org/10.1007/s00330-018-5787-2
Davnall F, Yip CS, Ljungqvist G, Selmi M, Ng F, Sanghera B, Ganeshan B, Miles KA, Cook GJ, Goh V (2012) Assessment of tumor heterogeneity: an emerging imaging tool for clinical practice? Insights Imaging 3:573-589. https://doi.org/10.1007/s13244-012-0196-6
Kiryu S, Akai H, Nojima M, Hasegawa K, Shinkawa H, Kokudo N, Yasaka K, Ohtomo K (2017) Impact of hepatocellular carcinoma heterogeneity on computed tomography as a prognostic indicator. Sci Rep 7:12689. https://doi.org/10.1038/s41598-017-12688-7
Ferrell LD (2015) Benign and malignant tumors of the liver. In: Odze RD, Goldblum JR (eds) Odze and goldblum surgical pathology of the GI tract, liver, biliary tract and pancreas, 3rd edn. Saunders/Elsevier, Philadelphia, PA, pp 1539–1572
DuBay D, Sandroussi C, Sandhu L, Cleary S, Guba M, Cattral MS, McGilvray I, Ghanekar A, Selzner M, Greig PD, Grant DR (2011) Liver transplantation for advanced hepatocellular carcinoma using poor tumor differentiation on biopsy as an exclusion criterion. Ann Surg 253:166-172. https://doi.org/10.1097/sla.0b013e31820508f1
Acknowledgements
We would like to thank the Advanced Medical Imaging Institute in the department of radiology, the Korea University Anam Hospital in the Republic of Korea, and researchers for providing software, datasets, and various forms of technical support. The data of this article were based on dissertation work performed by Yeo Eun Han at Korea University College of Medicine.
Funding
This study was supported by DongKook Life Science. Co. Ltd., Republic of Korea (DK-IIT-2019-20). This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (Grant Number: HI22C1302).
Author information
Authors and Affiliations
Contributions
MJK, YEH, and YC: Conceptualization. MJK, YEH, YC, and YSP: Data curation. MJK, YEH, and YC: Formal analysis. MJK, BJP: Funding acquisition. MJK, YEH, YC, NYH, and KCS: Investigation. MJK, YEH, YC, and BJP: Methodology. MJK, YEH, YC, BJP, and DJS: Project administration. BJP, DJS, NYH, KCS, YSP, and BNP: Resources. YC: Software. BJP and DJS: Supervision. BJP, NYH, and KCS: Validation. YEH and YC: Visualization. YEH and YC: Writing—original draft. MJK, YEH, YC, and BJP: Writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Informed consent
The Institutional Review Board waived written informed consent due to the retrospective nature of the data.
Ethical approval
Institutional Review Board of Korea University Anam Hospital and Korea University Guro Hospital approved this study (IRB number 2019AN0412, 2021GR0318).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Han, Y.E., Cho, Y., Kim, M.J. et al. Hepatocellular carcinoma pathologic grade prediction using radiomics and machine learning models of gadoxetic acid-enhanced MRI: a two-center study. Abdom Radiol 48, 244–256 (2023). https://doi.org/10.1007/s00261-022-03679-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-022-03679-y