Abstract
Objectives
This study aimed to evaluate the level of evidence of expert recommendations and guidelines for clinical indications and procedurals in hybrid nuclear cardiovascular imaging.
Methods
From inception to August 2023, a PubMed literature analysis of the latest version of guidelines for clinical hybrid cardiovascular imaging techniques including SPECT(/CT), PET(/CT), and PET(/MRI) was performed in two categories: (1) for clinical indications for all-in primary diagnosis; subgroup in prognosis and therapy evaluation; and for (2) imaging procedurals. We surveyed to what degree these followed a standard methodology to collect the data and provide levels of evidence, and for which topic systematic review evidence was executed.
Results
A total of 76 guidelines, published between 2013 and 2023, were included. The evidence of guidelines was based on systematic reviews in 7.9% of cases, non-systematic reviews in 47.4% of cases, a mix of systematic and non-systematic reviews in 19.7%, and 25% of guidelines did not report any evidence. Search strategy was reported in 36.8% of cases. Strengths of recommendation were clearly reported in 25% of guidelines. The notion of external review was explicitly reported in 23.7% of cases. Finally, the support of a methodologist was reported in 11.8% of the included guidelines.
Conclusion
The use of evidence procedures for developing for evidence-based cardiovascular hybrid imaging recommendations and guidelines is currently suboptimal, highlighting the need for more standardized methodological procedures.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The number of guidelines released for positron emission tomography (PET) and PET/computed tomography (CT) has significantly increased in the past decade, and this trend is expected to continue growing with the increasing clinical applications of novel tracers and the adoption of new imaging modalities. In addition to the guidelines developed by highly respected professional organizations on major topics, numerous guidelines have been formulated by national and regional organizations or even expert panels to address specific questions relevant to local practice. While this diversity of guidelines aims to cater to the needs of different populations, it also raises concerns about the reliability of recommendations.
Guidelines from various specialties have consistently exhibited low methodological quality and inconsistent recommendations [1, 2], with some even failing to meet basic methodological standards [3]. Surprisingly, no methodological studies have been conducted for guidelines pertaining to PET, PET/CT, or PET/magnetic resonance imaging (MRI). The collection and utilization of evidence are fundamental processes that significantly impact the quality of guideline development and, consequently, the formulation of essential recommendations [4]. Among all forms of evidence, systematic reviews are considered the gold standard for guideline development worldwide [5]. This emphasis on developing recommendations based on systematic review evidence is also underscored in the definition of guidelines by the Institute of Medicine (IOM), which defines clinical practice guidelines as statements that include recommendations intended to optimize patient care, informed by a systematic review of evidence and an assessment of the benefits and harms of alternative care options [6]. Nevertheless, the extent to which systematic review evidence is used in developing recommendations for nuclear imaging guidelines remains unknown, or may not be properly executed.
The purpose of our study was to comprehensively review all clinical and technical practice guidelines, recommendations, and expert opinions related to hybrid nuclear cardiovascular imaging and available on PubMed. Our aim was to evaluate the extent to which these adhere to standardized methodologies for evidence collection and utilization. We also sought to identify areas within recommendation topics where systematic review evidence may be inadequately referenced.
Methods
This was a cross-sectional systematic survey of published literature that did not involve human subjects, and hence was exempt from institutional review board approval.
Selection of guidelines
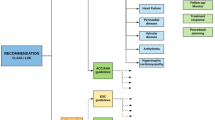
We included international clinical practice guidelines published in peer-reviewed journals indexed in PubMed for the indications or procedures of SPECT(/CT), PET(/CT), or PET(/MRI) imaging applied to the following fields of cardiovascular imaging: heart failure, coronary artery disease, extra-cardiac atherosclerosis, infection (endocarditis, cardiovascular implantable electronic devices) and inflammation (large vessel vasculitis), amyloidosis, sarcoidosis, cardiotoxicity, radiation dose and safety, and artificial intelligence. For all topics, only guidelines published in English language were retained. Were considered as “guidelines”: (i) the publications which were self-identified as “guideline” and were developed or endorsed by official international consortiums or (ii) recommendations, high level position, and expert papers by experts in the fields; (iii) a single document may exist of more than one cardiovascular topic and counted additionally.
Search of guidelines
For any of the fields of cardiovascular imaging, we searched PubMed from inception to August 2023 to identify guidelines pertaining to cardiovascular hybrid SPECT(/CT), PET(/CT), or PET(/MRI) for the diagnosis, treatment monitoring, or procedurals using the combination of the following terms: “((PET*) OR (positron) OR (SPECT*) OR (hybrid)) AND ((guideline*) OR (recommendation*) OR (position) OR (expert) OR (consensus)) AND ((imaging procedure) AND ((disease) AND (diagnosis/therapy evaluation OR monitoring OR treatment/prognosis OR event* OR survival OR follow up))).” In all cases, two predefined investigators dedicated to a field of interest and blinded to each other screened the documents retrieved by this search. Final results were centralized and checked by two investigators (R. H. J. A. Slart and F. L. Besson). Any disagreements were resolved by consensus with the investigators dedicated to the field of interest.
Two categories of guidelines
All included guidelines were classified into two categories as follows: (1) guidelines for clinical purpose (i.e., diagnosis and/or therapy evaluation) and (2) guidelines for procedures, if providing step-by-step instructions for cardiovascular hybrid imaging.
Survey of the collection and use of evidence
To survey the use of scientific evidence in the guidelines, we developed a questionnaire (Fig. 1) adapted from the article of Li et al. [7]. For each field of interest, the two pre-defined investigators surveyed the guidelines and cross-validated their findings. Disagreements were resolved by consensus. In the case of procedural guidelines, the following recommendation topics were searched and had to be fulfilled: patient preparation, radiopharmaceutical dosage, acquisition and reconstruction of images, analysis and interpretation of images, and radiation safety.
Analysis
We conducted a qualitative analysis of the data providing absolute frequencies and proportions in % or median (IQR) when appropriate. Data analysis and visualization were conducted using Microsoft Excel (version 16.77.1).
Results
The overall flow diagram for literature screening is provided in Fig. 2. The overall search identified 76 records as guidelines for cardiovascular hybrid imaging.
Characteristics of the included guidelines
The main characteristics of the guidelines are provided in the Table 1 and Fig. 3. The included guidelines were published between 2013 and 2023, with a rate per year ranging from 1 (2013) [8] to 13 (2021) [9,10,11,12,13,14,15,16,17,18,19,20,21]. A total of 47 (62%) guidelines concerned clinical purposes [9, 11, 13,14,15,16, 20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60], whereas 22 (29%) guidelines concerned procedures [8, 10, 12, 17, 18, 61,62,63,64,65,66,67,68,69,70,71,72,73,74,75] and seven (9%) concerned both [10, 19, 76,77,78,79,80]. Three out of these papers existed of two different guidelines and counted double [10, 18, 61].
A total of 56 guidelines were identified as “original” [9,10,11, 13, 15,16,17,18,19, 21,22,23,24,25,26,27,28,29,30, 32, 34,35,36,37,38,39, 41,42,43,44,45,46,47,48, 50,51,52,53, 55, 56, 59,60,61,62,63,64, 67,68,69,70, 73, 75,76,77, 79, 80], 14 as “updates” [8, 14, 20, 31, 33, 40, 49, 57, 58, 65, 66, 71, 74, 78], and the remaining four where summaries [18, 54, 72] or reprint [12]. We included 28 guidelines for PET(/CT-MRI) [10, 14, 15, 18, 22, 23, 25, 26, 30,31,32, 34, 41, 47,48,49, 51, 52, 55, 57, 61,62,63, 66, 67, 69, 71, 78], 28 guidelines for both PET(/CT) and SPECT(/CT) [8, 11,12,13, 19, 20, 24, 27,28,29, 33, 35, 37, 39, 43, 45, 50, 53, 54, 56, 58,59,60, 64, 73, 75,76,77], and 17 guidelines for SPECT(/CT) [9, 16, 17, 21, 36, 38, 40, 42, 44, 46, 65, 68, 70, 72, 74, 79, 80]. Amyloidosis was the most represented condition (n = 18) [9, 11,12,13, 16,17,18, 21, 36, 38, 40, 42, 44, 46, 68, 72, 79, 80], followed by CAD (n = 13) [8, 20, 25, 34, 48, 53,54,55, 60, 61, 70, 74, 78], sarcoidosis (n = 11) [14, 22, 26, 30, 41, 57, 58, 62, 66, 71, 75], heart failure (n = 7) [27, 28, 35, 37, 45, 50, 56], CIED (n = 7) [10, 18, 33, 39, 49, 63, 64], LVV (n = 5) [10, 15, 31, 32, 69], extra-cardiac atherosclerosis (n = 3) [47, 52, 61], radiation dose and safety (n = 2) [76, 77], non-obstructive CAD (n = 2) [23, 43], cardiotoxicity (n = 2) [51, 59], reporting [67, 73], AI (n = 1) [19], gated blood pool assessment for LV dysfunction (n = 1) [65], cardiomyopathy (n = 1) [24], and Takotsubo (n = 1) [29]. The radiotracers explicitly concerned were mostly 99mTc-based for SPECT (n = 29 guidelines) [8, 9, 11,12,13,14, 16, 17, 21, 24, 35, 36, 38, 40, 42, 44,45,46, 50, 58, 59, 65, 68, 70, 72,73,74, 79, 80] and 18F-FDG for PET (n = 37) [10, 14, 15, 18, 22, 24, 26,27,28,29,30,31,32,33,34,35, 39, 41, 45, 47,48,49,50,51,52, 57,58,59, 61,62,63,64, 66, 69, 71, 73, 75]. Also mentioned were 123I-mIBG for SPECT (n = 8) [27,28,29, 42, 45, 50, 59, 73] and 82Rb for PET (n = 8) [8, 14, 28, 35, 45, 59, 73, 78]. Other radiotracers including radiolabeled white blood cell, 201Thallium, 11C-based, 13N, 18F-Na, 68 Ga-somatostatin analogues, and 67 Ga were less frequently mentioned. The majority of the overall guidelines were developed by organizations located in western geographical areas (n = 64), of whom 31 from Europe [9,10,11, 16, 18, 19, 21, 23, 24, 27, 28, 31,32,33,34, 37, 39, 41, 43, 45, 47, 50, 51, 53, 54, 56, 59, 61, 64, 73, 74], 19 from North America [8, 14, 15, 20, 22, 25, 26, 30, 35, 36, 40, 49, 62, 67, 68, 71, 77, 78, 80], and 14 from mixed collaborations [12, 13, 29, 38, 46, 48, 52, 55, 63, 65, 69, 72, 75, 76]. The remaining of the guidelines were produced by Asian expert societies (n = 9) [17, 42, 44, 57, 58, 60, 66, 79], and six guidelines concerned broad international consortiums [38, 48, 52, 72, 76]. Near 75% of the guidelines concerned adult population (n = 57), one guideline dealing on radiation dose and safety focused exclusively on a pediatric population [76], and nine guidelines did not mention any targeted population [10, 27, 28, 37, 45, 50, 56, 63]. Only one paper assigned an appropriateness score for using nuclear imaging in the therapy management of the disease [13]; 12 other publications just mentioned nuclear imaging as an possible option [11, 24, 33, 41, 42, 57, 62, 65, 66, 68, 69, 75]. To note, the number of citations per guideline was highly heterogeneous, ranging from 0 ([14, 35, 44, 48, 60, 79] to 1337 of a previous clinical guideline [39], with a median number of citations per guideline of 18.5 (IQR = 4–59). Over the past 10 years (2013–2023), the cumulative citation rate per year ranged from 9 (2023) to 1465 (2015), with a median number of cumulative citations per year of 375 (IQR = 171.5–425).
Quality assessment of the guidelines: evidence, search strategy, strength of recommendation, external review, and methodology
The quality assessment of the included guidelines is provided in Table 2 and Fig. 4. The evidence was based on non-systematic reviews mainly (47.4%), followed by mix-based systematic and non-systematic reviews (19.7%) and systematic reviews (7.9%). In 25% of the cases, no evidence was mentioned. A search strategy was clearly reported in 28 guidelines (36.8%) [8, 12,13,14,15, 20, 22, 24, 26, 30,31,32,33,34,35, 37, 39, 40, 49, 58, 60,61,62, 66, 69, 71, 74, 75]. The strengths of recommendations were clearly reported in 19 guidelines (25%) [11, 14, 15, 20, 22, 26, 31, 32, 35,36,37, 40, 42, 49, 58, 60], partially reported in one guideline [69], and not reported in the remaining 56 guidelines (73.7%) [9, 10, 12, 16,17,18,19, 21, 27, 28, 30, 38, 44,45,46,47,48, 52, 56, 57, 63, 66, 72, 79, 80]. An external review was reported in 18 guidelines (23.7%) [8, 14, 15, 20, 22, 26, 30,31,32, 35, 37, 40, 42, 56, 77]. The involvement of a methodologist was clearly reported in nine guidelines (11.8% of the cases) [15, 26, 30,31,32, 40, 42]. In the specific subgroup of procedural guidelines (n = 22), key technical or interpretation procedures were clearly and exhaustively specified in ten guidelines (45.5%) [8, 12, 18, 61, 65, 68, 69, 73, 74] and partially reported in 11 guidelines (58.8%) [10, 17, 18, 62,63,64, 66, 67, 71, 72, 75]. In the specific subgroup of guidelines dealing both with clinical and procedural topics (n = 7), key technical or interpretation procedures were partially reported in 57.1% of cases [10, 78,79,80] and not reported in the remaining 42.9% [19, 76, 77]. The differences in methodology between the two continents with the highest number of official documents (Europe and North America) is provided in Table 3.
Discussion
The aim of our study was to evaluate the level of evidence of expert recommendations and guidelines for clinical indications and procedurals in hybrid cardiovascular nuclear imaging published for the last ten years. Clinical and procedural guidelines are relevant documents both in hybrid cardiovascular imaging and in other fields, aiming to improve the quality of care while reducing variability in clinical practice and containing healthcare costs [81].
First, we observed an increasing number of guidelines on hybrid cardiovascular nuclear imaging in recent years. This is expected and in line with the demonstration that the number of clinical practice guidelines produced for healthcare has risen exponentially in the last 20 years [81]. Two recent clinical guidelines of the European Society of Cardiology assigned a high class of recommendation and high level of evidence for our nuclear medicine techniques: IB for [18F]FDG in infective endocarditis and IB for [99mTc]-bone seeking agents in patients suspected of cardiac amyloidosis [24, 33]. Of note, amyloidosis concerned the majority of the guidelines retrieved during the period 2020–2023. This could be explained by the FDA clearance of the first anti-amyloid drugs in 2019 and related treatment strategy issues, which potentially stimulated the interest for patient screening in this field.
Second, we observed several heterogeneous characteristics in the selected guidelines about type of guidelines, disease and hybrid imaging evaluated, radiotracers, targeted population, guideline developing organizations, and guideline citations. This heterogeneity was expected since the only common denominator of the selected guidelines was hybrid cardiovascular imaging.
The most important aim of our study was the quality assessment of the included guidelines on cardiovascular hybrid imaging focusing on use of evidence, search strategy, strength of recommendations, external review, and methodology.
About the use of the evidence, notably, only few selected guidelines are evidence-based documents based on systematic reviews. Evidence-based guidelines are an important tool for healthcare professionals to make informed decisions about patient care. Compared to non-evidence-based guidelines, they can help to minimize bias and enhance the quality and consistency of clinical practice or public health policy. Evidence-based guidelines are developed using a rigorous process that involves identifying the best available evidence, evaluating its quality, and synthesizing it into recommendations for clinical practice or public health policy. Translating evidence into practice can not only improve outcomes and quality of life for patients but also improve productivity and reduce healthcare costs [82]. Beyond cardiovascular hybrid imaging, not evidence-based guidelines are frequent encountered for other pathologies. Using non-systematic methods in clinical practice and procedural guidelines compromises the validity and reliability of the evidence used to inform guideline recommendations, leading potentially to misleading and untrustworthy results [83].
Search strategies are important in evidence-based guidelines and systematic reviews because they help researchers identify all relevant studies that meet the inclusion criteria, while minimizing the risk of missing important studies [84]. A search strategy was reported only in about half of the selected guidelines on cardiovascular hybrid imaging. As a consequence, the transparency and reproducibility of guidelines are compromised.
In most of the included guidelines, strength of recommendations is not reported. The strength of recommendations in guidelines is a measure of its confidence in the effectiveness of an intervention or a diagnostic method. The strength of recommendation is a grading scale that is used to rate the quality, quantity, and consistency of evidence. Unfortunately, many guidelines are inconsistent in rating the quality of evidence and the strength of recommendations. The GRADE system is a consensus on rating quality of evidence and strength of recommendations, which is increasingly being adopted by organizations worldwide [85, 86].
External reviews of guidelines are a way to ensure that the guidelines are of high quality and based on the best available evidence. External reviews can be conducted by independent experts or organizations, and they can provide valuable feedback on the content, format, and implementation of guidelines. External reviewers should comprise a full spectrum of relevant stakeholders, including scientific and clinical experts, organizations, agencies, patients, and representatives of the public [6].
Unfortunately, only a limited number of guidelines in our study included an external review.
Finally, a methodologist in the context of clinical practice guidelines is a professional who specializes in the development and implementation of clinical practice guidelines. Methodologists are responsible for ensuring that guidelines are based on the best available evidence and that they are developed using a rigorous and transparent process, working closely with the guideline development groups. Overall, a thorough methodological approach is needed for developing, reporting, and assessing evidence-based clinical practice guidelines [87]. Only few of the included guidelines on hybrid cardiovascular imaging included a methodologist.
Currently, there remains ongoing inconsistency in quality of clinical practice guidelines and procedural guidelines on hybrid cardiovascular imaging. Of note, our results are in line with two recent similar methodological works applied in the general field of PET imaging [7, 88]. The excessive number of low-quality guidelines also wastes resources and the efforts of care providers who rely on guidelines to inform their decision-making and clinical practice [81]. To address this issue, significant efforts are mandatory to improve the methodological quality of guidelines and establish a standardized approach to develop evidence-based guidelines in hybrid cardiovascular imaging as in other medical fields. Incorporation of our (hybrid) nuclear medicine imaging techniques in (clinical) guidelines is pivotal, for visibility and clinical use. Because the role and support from the international consortiums is essential, the EANM has recently launched a dedicated Guidelines and Publications Council to further improve the development of guidelines and ensure high-quality standards.
Conclusions
The use of evidence procedures for evidence-based developing cardiovascular hybrid imaging recommendation guidelines is currently suboptimal, highlighting the need for more standardized methodological procedures.
References
Carneiro AV. Methodological appraisal of guidelines. The AGREE instrument. Rev Port Cardiol. 2004;23:447–56.
Ferket BS, Genders TS, Colkesen EB, Visser JJ, Spronk S, Steyerberg EW, et al. Systematic review of guidelines on imaging of asymptomatic coronary artery disease. J Am Coll Cardiol. 2011;57:1591–600. https://doi.org/10.1016/j.jacc.2010.10.055.
Shaneyfelt TM, Mayo-Smith MF, Rothwangl J. Are guidelines following guidelines? The methodological quality of clinical practice guidelines in the peer-reviewed medical literature. JAMA. 1999;281:1900–5. https://doi.org/10.1001/jama.281.20.1900.
Qaseem A, Kansagara D, Lin JS, Mustafa RA, Wilt TJ, Clinical Guidelines Committee of the American College of P, et al. The Development of Clinical Guidelines and Guidance Statements by the Clinical Guidelines Committee of the American College of Physicians: Update of Methods. Ann Intern Med. 2019;170:863–70. https://doi.org/10.7326/M18-3290.
WHO Handbook for Guideline development. Extra info: WHO handbook for guideline development – 2nd ed. 1. Guidelines as Topic – standards. 2. Review. 3. Meta-Analysis. 4. Peer Review. 5. Evidence-Based Medicine. 6. World Health Organization. I. World Health Organization. 2014
Graham R, Mancher M, Miller Wolman D, Greenfield S, Steinberg E. Clinical practice guidelines we can trust. Extra info: The National Academies Press 500 Fifth Street, N.W. Washington, DC. 2011
Li Q, Hou W, Li L, Su M, Ren Y, Wang W, et al. The use of systematic review evidence to support the development of guidelines for positron emission tomography: a cross-sectional survey. Eur Radiol. 2021;31:6992–7002. https://doi.org/10.1007/s00330-021-07756-6.
Dorbala S, Di Carli MF, Delbeke D, Abbara S, DePuey EG, Dilsizian V, et al. SNMMI/ASNC/SCCT guideline for cardiac SPECT/CT and PET/CT 1.0. J Nucl Med. 2013;54:1485–507. https://doi.org/10.2967/jnumed.112.105155.
Bistola V, Parissis J, Foukarakis E, Valsamaki PN, Anastasakis A, Koutsis G, et al. Practical recommendations for the diagnosis and management of transthyretin cardiac amyloidosis. Heart Fail Rev. 2021;26:861–79. https://doi.org/10.1007/s10741-020-10062-w.
Casali M, Lauri C, Altini C, Bertagna F, Cassarino G, Cistaro A, et al. State of the art of (18)F-FDG PET/CT application in inflammation and infection: a guide for image acquisition and interpretation. Clin Transl Imaging. 2021;9:299–339. https://doi.org/10.1007/s40336-021-00445-w.
Condoluci A, Theaudin M, Schwotzer R, Pazhenkottil AP, Arosio P, Averaimo M, et al. Management of transthyretin amyloidosis. Swiss Med Wkly. 2021;151:w30053. https://doi.org/10.4414/smw.2021.w30053.
Dorbala S, Ando Y, Bokhari S, Dispenzieri A, Falk RH, Ferrari VA, et al. ASNC/AHA/ASE/EANM/HFSA/ISA/SCMR/SNMMI Expert Consensus Recommendations for Multimodality Imaging in Cardiac Amyloidosis: Part 1 of 2-Evidence Base and Standardized Methods of Imaging. Circ Cardiovasc Imaging. 2021;14:e000029. https://doi.org/10.1161/HCI.0000000000000029.
Dorbala S, Ando Y, Bokhari S, Dispenzieri A, Falk RH, Ferrari VA, et al. ASNC/AHA/ASE/EANM/HFSA/ISA/SCMR/SNMMI Expert Consensus Recommendations for Multimodality Imaging in Cardiac Amyloidosis: Part 2 of 2-Diagnostic Criteria and Appropriate Utilization. Circ Cardiovasc Imaging. 2021;14:e000030. https://doi.org/10.1161/HCI.0000000000000030.
Expert Panel on Cardiac Imaging, Rajiah P, Kirsch J, Bolen MA, Batlle JC, Brown RKJ, et al. ACR Appropriateness Criteria® Nonischemic myocardial disease with clinical manifestations (ischemic cardiomyopathy already excluded). J Am Coll Radiol. 2021;18:S83–105. https://doi.org/10.1016/j.jacr.2021.01.019.
Expert Panel on Vascular Imaging, Aghayev A, Steigner ML, Azene EM, Burns J, Chareonthaitawee P, et al. ACR Appropriateness Criteria(R) noncerebral vasculitis. J Am Coll Radiol. 2021;18:S380–93. https://doi.org/10.1016/j.jacr.2021.08.005.
Garcia-Pavia P, Rapezzi C, Adler Y, Arad M, Basso C, Brucato A, et al. Diagnosis and treatment of cardiac amyloidosis: a position statement of the ESC Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2021;42:1554–68. https://doi.org/10.1093/eurheartj/ehab072.
Huang YH, Lin YH, Yen RF, Hou CJ, Wang SY, Tsai SC, et al. 2021 Advocacy statements for the role of (99m)Tc-pyrophosphate scintigraphy in the diagnosis of transthyretin cardiac amyloidosis: a report of the Taiwan Society of Cardiology and the Society of Nuclear Medicine of the Republic of China. Acta Cardiol Sin. 2021;37:221–31. https://doi.org/10.6515/ACS.202105_37(3).20210420A.
Slart R, Glaudemans A, Gheysens O, Lubberink M, Kero T, Dweck MR, et al. Procedural recommendations of cardiac PET/CT imaging: standardization in inflammatory-, infective-, infiltrative-, and innervation- (4Is) related cardiovascular diseases: a joint collaboration of the EACVI and the EANM: summary. Eur Heart J Cardiovasc Imaging. 2020;21:1320–30. https://doi.org/10.1093/ehjci/jeaa299.
Slart R, Williams MC, Juarez-Orozco LE, Rischpler C, Dweck MR, Glaudemans A, et al. Position paper of the EACVI and EANM on artificial intelligence applications in multimodality cardiovascular imaging using SPECT/CT, PET/CT, and cardiac CT. Eur J Nucl Med Mol Imaging. 2021;48:1399–413. https://doi.org/10.1007/s00259-021-05341-z.
Gulati M, Levy PD, Mukherjee D, Amsterdam E, Bhatt DL, et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation and Diagnosis of Chest Pain: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. 2021;78:e187–285. https://doi.org/10.1016/j.jacc.2021.07.053.
Yilmaz A, Bauersachs J, Bengel F, Buchel R, Kindermann I, Klingel K, et al. Diagnosis and treatment of cardiac amyloidosis: position statement of the German Cardiac Society (DGK). Clin Res Cardiol. 2021;110:479–506. https://doi.org/10.1007/s00392-020-01799-3.
Al-Khatib SM, Stevenson WG, Ackerman MJ, Bryant WJ, Callans DJ, Curtis AB, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2018;15:e190–252. https://doi.org/10.1016/j.hrthm.2017.10.035.
Alexander Y, Osto E, Schmidt-Trucksass A, Shechter M, Trifunovic D, Duncker DJ, et al. Endothelial function in cardiovascular medicine: a consensus paper of the European Society of Cardiology Working Groups on Atherosclerosis and Vascular Biology, Aorta and Peripheral Vascular Diseases, Coronary Pathophysiology and Microcirculation, and Thrombosis. Cardiovasc Res. 2021;117:29–42. https://doi.org/10.1093/cvr/cvaa085.
Arbelo E, Protonotarios A, Gimeno JR, Arbustini E, Barriales-Villa R, Basso C, et al. 2023 ESC Guidelines for the management of cardiomyopathies. Eur Heart J. 2023;44:3503–626. https://doi.org/10.1093/eurheartj/ehad194.
Bateman TM, Dilsizian V, Beanlands RS, DePuey EG, Heller GV, Wolinsky DA. American Society of Nuclear Cardiology and Society of Nuclear Medicine and Molecular Imaging Joint Position Statement on the Clinical Indications for Myocardial Perfusion PET. J Nucl Med. 2016;57:1654–6. https://doi.org/10.2967/jnumed.116.180448.
Birnie DH, Sauer WH, Bogun F, Cooper JM, Culver DA, Duvernoy CS, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014;11:1305–23. https://doi.org/10.1016/j.hrthm.2014.03.043.
Celutkiene J, Lainscak M, Anderson L, Gayat E, Grapsa J, Harjola VP, et al. Imaging in patients with suspected acute heart failure: timeline approach position statement on behalf of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2020;22:181–95. https://doi.org/10.1002/ejhf.1678.
Celutkiene J, Plymen CM, Flachskampf FA, de Boer RA, Grapsa J, Manka R, et al. Innovative imaging methods in heart failure: a shifting paradigm in cardiac assessment. Position statement on behalf of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail. 2018;20:1615–33. https://doi.org/10.1002/ejhf.1330.
Citro R, Okura H, Ghadri JR, Izumi C, Meimoun P, Izumo M, et al. Multimodality imaging in takotsubo syndrome: a joint consensus document of the European Association of Cardiovascular Imaging (EACVI) and the Japanese Society of Echocardiography (JSE). Eur Heart J Cardiovasc Imaging. 2020;21:1184–207. https://doi.org/10.1093/ehjci/jeaa149.
Crouser ED, Maier LA, Wilson KC, Bonham CA, Morgenthau AS, Patterson KC, et al. Diagnosis and detection of sarcoidosis. An Official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;201:e26–51. https://doi.org/10.1164/rccm.202002-0251ST.
Dejaco C, Ramiro S, Bond M, Bosch P, Ponte C, Mackie SL, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice: 2023 update. Ann Rheum Dis. 2023. https://doi.org/10.1136/ard-2023-224543.
Dejaco C, Ramiro S, Duftner C, Besson FL, Bley TA, Blockmans D, et al. EULAR recommendations for the use of imaging in large vessel vasculitis in clinical practice. Ann Rheum Dis. 2018;77:636–43. https://doi.org/10.1136/annrheumdis-2017-212649.
Delgado V, Ajmone Marsan N, de Waha S, Bonaros N, Brida M, Burri H, et al. 2023 ESC Guidelines for the management of endocarditis. Eur Heart J. 2023;44:3948–4042. https://doi.org/10.1093/eurheartj/ehad193.
Dweck MR, Maurovich-Horvat P, Leiner T, Cosyns B, Fayad ZA, Gijsen FJH, et al. Contemporary rationale for non-invasive imaging of adverse coronary plaque features to identify the vulnerable patient: a position paper from the European Society of Cardiology Working Group on Atherosclerosis and Vascular Biology and the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2020;21:1177–83. https://doi.org/10.1093/ehjci/jeaa201.
Expert Panel on Cardiac Imaging, White RD, Kirsch J, Bolen MA, Batlle JC, Brown RKJ, et al. ACR Appropriateness Criteria((R)) suspected new-onset and known nonacute heart failure. J Am Coll Radiol. 2018;15:S418–31. https://doi.org/10.1016/j.jacr.2018.09.031.
Fine NM, Davis MK, Anderson K, Delgado DH, Giraldeau G, Kitchlu A, et al. Canadian Cardiovascular Society/Canadian Heart Failure Society Joint Position Statement on the Evaluation and Management of Patients With Cardiac Amyloidosis. Can J Cardiol. 2020;36:322–34. https://doi.org/10.1016/j.cjca.2019.12.034.
Garbi M, Edvardsen T, Bax J, Petersen SE, McDonagh T, Filippatos G, et al. EACVI appropriateness criteria for the use of cardiovascular imaging in heart failure derived from European National Imaging Societies voting. Eur Heart J Cardiovasc Imaging. 2016;17:711–21. https://doi.org/10.1093/ehjci/jew081.
Gertz M, Adams D, Ando Y, Beirao JM, Bokhari S, Coelho T, et al. Avoiding misdiagnosis: expert consensus recommendations for the suspicion and diagnosis of transthyretin amyloidosis for the general practitioner. BMC Fam Pract. 2020;21:198. https://doi.org/10.1186/s12875-020-01252-4.
Habib G, Lancellotti P, Antunes MJ, Bongiorni MG, Casalta JP, Del Zotti F, et al. ESC Guidelines for the management of infective endocarditis: The Task Force for the Management of Infective Endocarditis of the European Society of Cardiology (ESC). Endorsed by: European Association for Cardio-Thoracic Surgery (EACTS), the European Association of Nuclear Medicine (EANM). Eur Heart J. 2015;36:3075–128. https://doi.org/10.1093/eurheartj/ehv319.
Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e876–94. https://doi.org/10.1161/CIR.0000000000001062.
Ho JSY, Chilvers ER, Thillai M. Cardiac sarcoidosis - an expert review for the chest physician. Expert Rev Respir Med. 2019;13:507–20. https://doi.org/10.1080/17476348.2018.1511431.
Kitaoka H, Izumi C, Izumiya Y, Inomata T, Ueda M, Kubo T, et al. JCS 2020 Guideline on Diagnosis and Treatment of Cardiac Amyloidosis. Circ J. 2020;84:1610–71. https://doi.org/10.1253/circj.CJ-20-0110.
Kunadian V, Chieffo A, Camici PG, Berry C, Escaned J, Maas A, et al. An EAPCI expert consensus document on ischaemia with non-obstructive coronary arteries in collaboration with European Society of Cardiology Working Group on Coronary Pathophysiology & Microcirculation Endorsed by Coronary Vasomotor Disorders International Study Group. Eur Heart J. 2020;41:3504–20. https://doi.org/10.1093/eurheartj/ehaa503.
Lin W, Chattranukulchai P, Lee AP, Lin YH, Yu WC, Liew HB, et al. Clinical recommendations to diagnose and monitor patients with transthyretin amyloid cardiomyopathy in Asia. Clin Cardiol. 2022;45:898–907. https://doi.org/10.1002/clc.23882.
Marwick TH, Gimelli A, Plein S, Bax JJ, Charron P, Delgado V, et al. Multimodality imaging approach to left ventricular dysfunction in diabetes: an expert consensus document from the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2022;23:e62–84. https://doi.org/10.1093/ehjci/jeab220.
Maurer MS, Bokhari S, Damy T, Dorbala S, Drachman BM, Fontana M, et al. Expert consensus recommendations for the suspicion and diagnosis of transthyretin cardiac amyloidosis. Circ Heart Fail. 2019;12: e006075. https://doi.org/10.1161/CIRCHEARTFAILURE.119.006075.
Messas E, Goudot G, Halliday A, Sitruk J, Mirault T, Khider L, et al. Management of carotid stenosis for primary and secondary prevention of stroke: state-of-the-art 2020: a critical review. Eur Heart J Suppl. 2020;22:M35–42. https://doi.org/10.1093/eurheartj/suaa162.
Mezquita AJV, Biavati F, Falk V, Alkadhi H, Hajhosseiny R, Maurovich-Horvat P, et al. Clinical quantitative coronary artery stenosis and coronary atherosclerosis imaging: a Consensus Statement from the Quantitative Cardiovascular Imaging Study Group. Nat Rev Cardiol. 2023;20:696–714. https://doi.org/10.1038/s41569-023-00880-4.
Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, Gentile F, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2021;143:e35–71. https://doi.org/10.1161/CIR.0000000000000932.
Peix A, Mesquita CT, Paez D, Pereira CC, Felix R, Gutierrez C, et al. Nuclear medicine in the management of patients with heart failure: guidance from an expert panel of the International Atomic Energy Agency (IAEA). Nucl Med Commun. 2014;35:818–23. https://doi.org/10.1097/MNM.0000000000000143.
Raschi E, Diemberger I, Cosmi B, De Ponti F. ESC position paper on cardiovascular toxicity of cancer treatments: challenges and expectations. Intern Emerg Med. 2018;13:1–9. https://doi.org/10.1007/s11739-017-1755-0.
Saba L, Antignani PL, Gupta A, Cau R, Paraskevas KI, Poredos P, et al. International Union of Angiology (IUA) consensus paper on imaging strategies in atherosclerotic carotid artery imaging: from basic strategies to advanced approaches. Atherosclerosis. 2022;354:23–40. https://doi.org/10.1016/j.atherosclerosis.2022.06.1014.
Saraste A, Barbato E, Capodanno D, Edvardsen T, Prescott E, Achenbach S, et al. Imaging in ESC clinical guidelines: chronic coronary syndromes. Eur Heart J Cardiovasc Imaging. 2019;20:1187–97. https://doi.org/10.1093/ehjci/jez219.
Saraste A, Knuuti J. ESC 2019 guidelines for the diagnosis and management of chronic coronary syndromes: recommendations for cardiovascular imaging. Herz. 2020;45:409–20. https://doi.org/10.1007/s00059-020-04935-x.
Schindler TH, Fearon WF, Pelletier-Galarneau M, Ambrosio G, Sechtem U, Ruddy TD, et al. Myocardial perfusion PET for the detection and reporting of coronary microvascular dysfunction: a JACC: Cardiovascular Imaging Expert Panel Statement. JACC Cardiovasc Imaging. 2023;16:536–48. https://doi.org/10.1016/j.jcmg.2022.12.015.
Smiseth OA, Morris DA, Cardim N, Cikes M, Delgado V, Donal E, et al. Multimodality imaging in patients with heart failure and preserved ejection fraction: an expert consensus document of the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging. 2022;23:e34–61. https://doi.org/10.1093/ehjci/jeab154.
Sohn DW, Park JB, Lee SP, Kim HK, Kim YJ. Viewpoints in the diagnosis and treatment of cardiac sarcoidosis: proposed modification of current guidelines. Clin Cardiol. 2018;41:1386–94. https://doi.org/10.1002/clc.23060.
Terasaki F, Azuma A, Anzai T, Ishizaka N, Ishida Y, Isobe M, et al. JCS 2016 Guideline on Diagnosis and Treatment of Cardiac Sarcoidosis-Digest Version. Circ J. 2019;83:2329–88. https://doi.org/10.1253/circj.CJ-19-0508.
Totzeck M, Aide N, Bauersachs J, Bucerius J, Georgoulias P, Herrmann K, et al. Nuclear medicine in the assessment and prevention of cancer therapy-related cardiotoxicity: prospects and proposal of use by the European Association of Nuclear Medicine (EANM). Eur J Nucl Med Mol Imaging. 2023;50:792–812. https://doi.org/10.1007/s00259-022-05991-7.
Ueng KC, Chiang CE, Chao TH, Wu YW, Lee WL, Li YH, et al. 2023 Guidelines of the Taiwan Society of Cardiology on the Diagnosis and Management of Chronic Coronary Syndrome. Acta Cardiol Sin. 2023;39:4–96. https://doi.org/10.6515/ACS.202301_39(1).20221103A.
Bucerius J, Hyafil F, Verberne HJ, Slart RH, Lindner O, Sciagra R, et al. Position paper of the Cardiovascular Committee of the European Association of Nuclear Medicine (EANM) on PET imaging of atherosclerosis. Eur J Nucl Med Mol Imaging. 2016;43:780–92. https://doi.org/10.1007/s00259-015-3259-3.
Chareonthaitawee P, Beanlands RS, Chen W, Dorbala S, Miller EJ, Murthy VL, et al. Joint SNMMI-ASNC Expert Consensus Document on the Role of (18)F-FDG PET/CT in Cardiac Sarcoid Detection and Therapy Monitoring. J Nucl Med. 2017;58:1341–53. https://doi.org/10.2967/jnumed.117.196287.
Dilsizian V, Budde RPJ, Chen W, Mankad SV, Lindner JR, Nieman K. Best practices for imaging cardiac device-related infections and endocarditis: a JACC: Cardiovascular Imaging Expert Panel Statement. JACC Cardiovasc Imaging. 2022;15:891–911. https://doi.org/10.1016/j.jcmg.2021.09.029.
Erba PA, Lancellotti P, Vilacosta I, Gaemperli O, Rouzet F, Hacker M, et al. Recommendations on nuclear and multimodality imaging in IE and CIED infections. Eur J Nucl Med Mol Imaging. 2018;45:1795–815. https://doi.org/10.1007/s00259-018-4025-0.
Farrell MB, Galt JR, Georgoulias P, Malhotra S, Pagnanelli R, Rischpler C, et al. SNMMI Procedure Standard/EANM Guideline for Gated Equilibrium Radionuclide Angiography. J Nucl Med Technol. 2020;48:126–35. https://doi.org/10.2967/jnmt.120.246405.
Kumita S, Yoshinaga K, Miyagawa M, Momose M, Kiso K, Kasai T, et al. Recommendations for (18)F-fluorodeoxyglucose positron emission tomography imaging for diagnosis of cardiac sarcoidosis-2018 update: Japanese Society of Nuclear Cardiology recommendations. J Nucl Cardiol. 2019;26:1414–33. https://doi.org/10.1007/s12350-019-01755-3.
Ruddy TD, Al-Mallah M, Arrighi JA, Bois JP, Bluemke DA, Di Carli MF, et al. SNMMI/ACR/ASNC/SCMR Joint Credentialing Statement for Cardiac PET/MRI: Endorsed by the American Heart Association. J Nucl Med. 2023;64:149–52. https://doi.org/10.2967/jnumed.122.264200.
Slart R, Chen W, Tubben A, Tingen HSA, Davies DR, Grogan M, et al. Emerging role of scintigraphy using bone-seeking tracers for diagnosis of cardiac amyloidosis: AJR expert panel narrative review. AJR Am J Roentgenol. 2023. https://doi.org/10.2214/AJR.23.29347.
Slart RHJA, Writing group; Reviewer group; Members of EANM, Members of EACVI, et al. FDG-PET/CT(A) imaging in large vessel vasculitis and polymyalgia rheumatica: joint procedural recommendation of the EANM, SNMMI, and the PET Interest Group (PIG), and endorsed by the ASNC. Eur J Nucl Med Mol Imaging. 2018;45:1250–69. https://doi.org/10.1007/s00259-018-3973-8.
Standardization Promotion Subcommittee of the Japanese Society of Nuclear Medicine (JSNMT), Aoki T, Tsuboi K, Miki K, Takenaka K, Tsushima H, et al. Guidelines for Standardization of Myocardial Perfusion SPECT Imaging 1.0. Ann Nucl Cardiol. 2022;8:91–102. https://doi.org/10.17996/anc.22-004.
Subramaniam RM, Janowitz WR, Johnson GB, Lodge MA, Parisi MT, Ferguson MR, et al. ACR-SPR-STR Practice Parameter for the Performance of Cardiac Positron Emission Tomography - Computed Tomography (PET/CT) Imaging. Clin Nucl Med. 2017;42:918–27. https://doi.org/10.1097/RLU.0000000000001827.
Tahara N, Lairez O, Endo J, Okada A, Ueda M, Ishii T, et al. (99m) Technetium-pyrophosphate scintigraphy: a practical guide for early diagnosis of transthyretin amyloid cardiomyopathy. ESC Heart Fail. 2022;9:251–62. https://doi.org/10.1002/ehf2.13693.
Tragardh E, Hesse B, Knuuti J, Flotats A, Kaufmann PA, Kitsiou A, et al. Reporting nuclear cardiology: a joint position paper by the European Association of Nuclear Medicine (EANM) and the European Association of Cardiovascular Imaging (EACVI). Eur Heart J Cardiovasc Imaging. 2015;16:272–9. https://doi.org/10.1093/ehjci/jeu304.
Verberne HJ, Acampa W, Anagnostopoulos C, Ballinger J, Bengel F, De Bondt P, et al. EANM procedural guidelines for radionuclide myocardial perfusion imaging with SPECT and SPECT/CT: 2015 revision. Eur J Nucl Med Mol Imaging. 2015;42:1929–40. https://doi.org/10.1007/s00259-015-3139-x.
Slart RHJA, Glaudemans AWJM, Lancellotti P, Hyafil F, Blankstein R, Schwartz RG, Jaber WA, Russell R, Gimelli A, Rouzet F, Hacker M, Gheysens O, Plein S, Miller EJ, Dorbala S, Donal E; Document Reading Group. A joint procedural position statement on imaging in cardiac sarcoidosis: from the Cardiovascular and Inflammation & Infection Committees of the European Association of Nuclear Medicine, the European Association of Cardiovascular Imaging, and the American Society of Nuclear Cardiology. Eur Heart J Cardiovasc Imaging. 2017;18:1073-89. https://doi.org/10.1093/ehjci/jex146.
Hill KD, Frush DP, Han BK, Abbott BG, Armstrong AK, DeKemp RA, et al. Radiation safety in children with congenital and acquired heart disease: a scientific position statement on multimodality dose optimization from the image gently alliance. JACC Cardiovasc Imaging. 2017;10:797–818. https://doi.org/10.1016/j.jcmg.2017.04.003.
Hirshfeld JW Jr, Ferrari VA, Bengel FM, Bergersen L, Chambers CE, Einstein AJ, et al. 2018 ACC/HRS/NASCI/SCAI/SCCT Expert Consensus Document on Optimal Use of Ionizing Radiation in Cardiovascular Imaging: Best Practices for Safety and Effectiveness: A Report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol. 2018;71:e283–351. https://doi.org/10.1016/j.jacc.2018.02.016.
Murthy VL, Bateman TM, Beanlands RS, Berman DS, Borges-Neto S, Chareonthaitawee P, et al. Clinical quantification of myocardial blood flow using PET: joint position paper of the SNMMI Cardiovascular Council and the ASNC. J Nucl Med. 2018;59:273–93. https://doi.org/10.2967/jnumed.117.201368.
Wang CC, Chang WT, Lin YH, Tzeng BH, Chao TH, Hung CL, et al. 2023 Expert Consensus of the Taiwan Society of Cardiology on the Diagnosis and Treatment of Cardiac Amyloidosis. Acta Cardiol Sin. 2023;39:511–43. https://doi.org/10.6515/ACS.202307_39(4).20230610A.
Writing Committee, Kittleson MM, Ruberg FL, Ambardekar AV, Brannagan TH, Cheng RK, et al. 2023 ACC Expert Consensus Decision Pathway on Comprehensive Multidisciplinary Care for the Patient With Cardiac Amyloidosis: A Report of the American College of Cardiology Solution Set Oversight Committee. J Am Coll Cardiol. 2023;81:1076–126. https://doi.org/10.1016/j.jacc.2022.11.022.
De Leo A, Bloxsome D, Bayes S. Approaches to clinical guideline development in healthcare: a scoping review and document analysis. BMC Health Serv Res. 2023;23:37. https://doi.org/10.1186/s12913-022-08975-3.
Carande-Kulis V, Elder RW, Koffman DM. Standards required for the development of CDC evidence-based guidelines. MMWR Suppl. 2022;71:1–6. https://doi.org/10.15585/mmwr.su7101a1.
Lunny C, Ramasubbu C, Puil L, Liu T, Gerrish S, Salzwedel DM, et al. Over half of clinical practice guidelines use non-systematic methods to inform recommendations: a methods study. PLoS ONE. 2021;16: e0250356. https://doi.org/10.1371/journal.pone.0250356.
Cooper C, Booth A, Varley-Campbell J, Britten N, Garside R. Defining the process to literature searching in systematic reviews: a literature review of guidance and supporting studies. BMC Med Res Methodol. 2018;18:85. https://doi.org/10.1186/s12874-018-0545-3.
Andrews J, Guyatt G, Oxman AD, Alderson P, Dahm P, Falck-Ytter Y, et al. GRADE guidelines: 14. Going from evidence to recommendations: the significance and presentation of recommendations. J Clin Epidemiol. 2013;66:719–25. https://doi.org/10.1016/j.jclinepi.2012.03.013.
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. 2008;336:924–6. https://doi.org/10.1136/bmj.39489.470347.AD.
Yao X, Xia J, Jin Y, Shen Q, Wang Q, Zhu Y, et al. Methodological approaches for developing, reporting, and assessing evidence-based clinical practice guidelines: a systematic survey. J Clin Epidemiol. 2022;146:77–85. https://doi.org/10.1016/j.jclinepi.2022.02.015.
Li Q, Hou W, Wu M, Li L, Su M, Ma B, et al. Quality and consistency of clinical practice guideline recommendations for PET/CT and PET: a systematic appraisal. Eur Radiol. 2023;33:7879–89. https://doi.org/10.1007/s00330-023-09786-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Professor Riemer H.J.A Slart, from Department of Nuclear Medicine, University Medical Center Groningen, Netherland.
Conflict of interest
The authors declare no competing interests.
Statistics and biometry
Some of the authors (GT, FLB) have significant statistical expertise.
Informed consent
Not applicable.
Ethical approval
This was a cross-sectional survey of published literatures that did not involve human subjects, and hence was exempt from institutional review board approval.
Study subjects or cohorts overlap
No study subject or cohort has been previously reported.
Methodology
• Retrospective
• Cross-sectional study
• Literature survey
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Besson, F.L., Treglia, G., Bucerius, J. et al. A systematic review for the evidence of recommendations and guidelines in hybrid nuclear cardiovascular imaging. Eur J Nucl Med Mol Imaging 51, 2247–2259 (2024). https://doi.org/10.1007/s00259-024-06597-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-024-06597-x