Abstract
Purpose
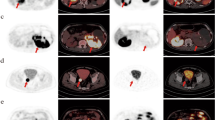
This pilot study was prospectively designed to evaluate and compare the diagnostic value of PET/CT using a PSMA-specific tracer [68Ga]Ga-P16-093 and a glucose metabolism probe 2-[18F]FDG in clear cell renal cell carcinoma (ccRCC) patients.
Methods
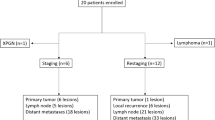
Forty-two pathologically confirmed ccRCC patients were included. Within 1 week, each patient underwent [68Ga]Ga-P16-093 and 2-[18F]FDG PET/CT. In addition to visual analysis of tumor number, the standardized uptake value (SUV) was measured for semiquantitative comparison and correlation analysis.
Results
For primary ccRCC patients, [68Ga]Ga-P16-093 PET/CT demonstrated a significantly higher detection rate (19/22 vs. 13/22, P = 0.031) and higher tumor uptake (15.7 ± 9.0 vs. 5.1 ± 3.4, P < 0.001) than 2-[18F]FDG PET/CT. In addition, the SUVmax of the primary tumor on [68Ga]Ga-P16-093 and 2-[18F]FDG PET/CT was significantly correlated with pT stage (for [68Ga]Ga-P16-093, r = 0.550, P = 0.008; for 2-[18F]FDG, r = 0.514, P = 0.014) and WHO/ISUP grade (for [68Ga]Ga-P16-093, r = 0.566, P = 0.006; for 2-[18F]FDG, r = 0.492, P = 0.020), respectively. For metastatic ccRCC patients, [68Ga]Ga-P16-093 PET/CT also demonstrated a better detection rate (21/22 vs. 14/22, P = 0.008) and higher tumor uptake (11.0 ± 6.4 vs. 4.4 ± 2.7, P < 0.001) than 2-[18F]FDG PET/CT. The SUVmax on [68Ga]Ga-P16-093 PET/CT had a significant association with PSMA expression in primary ccRCC (r = 0.776, P < 0.001) and metastatic ccRCC (r = 0.626, P = 0.029).
Conclusions
[68Ga]Ga-P16-093 PET/CT demonstrates significantly better tumor detectability than 2-[18F]FDG PET/CT for ccRCC patients.
Trial registration
68Ga-P16-093 and 18F-FDG PET/CT Imaging in the Same Group of Clear Cell Renal Cell Carcinoma Patients (NCT05432947, Registered 27 June 2021, retrospectively registered)
URL of registry






Similar content being viewed by others
Data availability
Not applicable
References
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72:7–33. https://doi.org/10.3322/caac.21708.
Joosten SC, Smits KM, Aarts MJ, Melotte V, Koch A, Tjan-Heijnen VC, et al. Epigenetics in renal cell cancer: mechanisms and clinical applications. Nat Rev Urol. 2018;15:430–51. https://doi.org/10.1038/s41585-018-0023-z.
Kelly-Morland C, Rudman S, Nathan P, Mallett S, Montana G, Cook G, et al. Evaluation of treatment response and resistance in metastatic renal cell cancer (mRCC) using integrated 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography/magnetic resonance imaging (PET/MRI). REMAP Study BMC Cancer. 2017;17:392. https://doi.org/10.1186/s12885-017-3371-9.
Romeo A, Garcia Marchinena P, Jurado AM, Gueglio G. Renal fossa recurrence after radical nephrectomy: current management, and oncological outcomes. Urol Oncol. 2020;38(42):e7–12. https://doi.org/10.1016/j.urolonc.2019.10.004.
Zhao Y, Wu C, Li W, Chen X, Li Z, Liao X, et al. 2-[18F]FDG PET/CT parameters associated with WHO/ISUP grade in clear cell renal cell carcinoma. Eur J Nucl Med Mol Imaging. 2021;48:570–9. https://doi.org/10.1007/s00259-020-04996-4.
Wu C, Cui Y, Liu J, Ma L, Xiong Y, Gong Y, et al. Noninvasive evaluation of tumor immune microenvironment in patients with clear cell renal cell carcinoma using metabolic parameter from preoperative 2-[18F]FDG PET/CT. Eur J Nucl Med Mol Imaging. 2021;48:4054–66. https://doi.org/10.1007/s00259-021-05399-9.
Aide N, Cappele O, Bottet P, Bensadoun H, Regeasse A, Comoz F, et al. Efficiency of [(18)F]FDG PET in characterising renal cancer and detecting distant metastases: a comparison with CT. Eur J Nucl Med Mol Imaging. 2003;30:1236–45. https://doi.org/10.1007/s00259-003-1211-4.
Sweat SD, Pacelli A, Murphy GP, Bostwick DG. Prostate-specific membrane antigen expression is greatest in prostate adenocarcinoma and lymph node metastases. Urology. 1998;52:637–40. https://doi.org/10.1016/s0090-4295(98)00278-7.
Chang SS, Reuter VE, Heston WD, Bander NH, Grauer LS, Gaudin PB. Five different anti-prostate-specific membrane antigen (PSMA) antibodies confirm PSMA expression in tumor-associated neovasculature. Cancer Res. 1999;59:3192–8.
Carlucci G, Ippisch R, Slavik R, Mishoe A, Blecha J, Zhu S. 68Ga-PSMA-11 NDA approval: a novel and successful academic partnership. J Nucl Med. 2021;62:149–55. https://doi.org/10.2967/jnumed.120.260455.
Cytawa W, Seitz AK, Kircher S, Fukushima K, Tran-Gia J, Schirbel A, et al. 68Ga-PSMA I&T PET/CT for primary staging of prostate cancer. Eur J Nucl Med Mol Imaging. 2020;47:168–77. https://doi.org/10.1007/s00259-019-04524-z.
Afshar-Oromieh A, Hetzheim H, Kratochwil C, Benesova M, Eder M, Neels OC, et al. The theranostic PSMA ligand PSMA-617 in the diagnosis of prostate cancer by PET/CT: biodistribution in humans, radiation dosimetry, and first evaluation of tumor lesions. J Nucl Med. 2015;56:1697–705. https://doi.org/10.2967/jnumed.115.161299.
Golan S, Aviv T, Groshar D, Yakimov M, Zohar Y, Prokocimer Y, et al. Dynamic 68Ga-PSMA-11 PET/CT for the primary evaluation of localized renal mass: a prospective study. J Nucl Med. 2021;62:773–8. https://doi.org/10.2967/jnumed.120.251272.
Gao J, Xu Q, Fu Y, He K, Zhang C, Zhang Q, et al. Comprehensive evaluation of 68Ga-PSMA-11 PET/CT parameters for discriminating pathological characteristics in primary clear-cell renal cell carcinoma. Eur J Nucl Med Mol Imaging. 2021;48:561–9. https://doi.org/10.1007/s00259-020-04916-6.
Raveenthiran S, Esler R, Yaxley J, Kyle S. The use of 68Ga-PET/CT PSMA in the staging of primary and suspected recurrent renal cell carcinoma. Eur J Nucl Med Mol Imaging. 2019;46:2280–8. https://doi.org/10.1007/s00259-019-04432-2.
Doot RK, Young AJ, Daube-Witherspoon ME, Alexoff D, Labban KJ, Lee H, et al. Biodistribution, dosimetry, and temporal signal-to-noise ratio analyses of normal and cancer uptake of [68Ga]Ga-P15-041, a gallium-68 labeled bisphosphonate, from first-in-human studies. Nucl Med Biol. 2020;86–87:1–8. https://doi.org/10.1016/j.nucmedbio.2020.04.002.
Lee H, Scheuermann JS, Young AJ, Doot RK, Daube-Witherspoon ME, Schubert EK, et al. Preliminary evaluation of 68Ga-P16-093, a PET radiotracer targeting prostate-specific membrane antigen in prostate cancer. Mol Imaging Biol. 2022;24:710–20. https://doi.org/10.1007/s11307-022-01720-6.
Zha Z, Ploessl K, Choi SR, Wu Z, Zhu L, Kung HF. Synthesis and evaluation of a novel urea-based 68Ga-complex for imaging PSMA binding in tumor. Nucl Med Biol. 2018;59:36–47. https://doi.org/10.1016/j.nucmedbio.2017.12.007.
Green MA, Hutchins GD, Bahler CD, Tann M, Mathias CJ, Territo W, et al. [68Ga]Ga-P16-093 as a PSMA-targeted PET radiopharmaceutical for detection of cancer: initial evaluation and comparison with [68Ga]Ga-PSMA-11 in prostate cancer patients presenting with biochemical recurrence. Mol Imaging Biol. 2020;22:752–63. https://doi.org/10.1007/s11307-019-01421-7.
Wang G, Hong H, Zang J, Liu Q, Jiang Y, Fan X, et al. Head-to-head comparison of [68 Ga]Ga-P16-093 and [68 Ga]Ga-PSMA-617 in dynamic PET/CT evaluation of the same group of recurrent prostate cancer patients. Eur J Nucl Med Mol Imaging. 2022;49:1052–62. https://doi.org/10.1007/s00259-021-05539-1.
Verhoeff SR, van Es SC, Boon E, van Helden E, Angus L, Elias SG, et al. Lesion detection by [89Zr]Zr-DFO-girentuximab and [18F]FDG-PET/CT in patients with newly diagnosed metastatic renal cell carcinoma. Eur J Nucl Med Mol Imaging. 2019;46:1931–9. https://doi.org/10.1007/s00259-019-04358-9.
Park S, Lee HY, Lee S. Role of F-18 FDG PET/CT in the follow-up of asymptomatic renal cell carcinoma patients for postoperative surveillance: based on conditional survival analysis. J Cancer Res Clin Oncol. 2022;148:215–24. https://doi.org/10.1007/s00432-021-03688-2.
Meyer AR, Carducci MA, Denmeade SR, Markowski MC, Pomper MG, Pierorazio PM, et al. Improved identification of patients with oligometastatic clear cell renal cell carcinoma with PSMA-targeted 18F-DCFPyL PET/CT. Ann Nucl Med. 2019;33:617–23. https://doi.org/10.1007/s12149-019-01371-8.
Rhee H, Blazak J, Tham CM, Ng KL, Shepherd B, Lawson M, et al. Pilot study: use of gallium-68 PSMA PET for detection of metastatic lesions in patients with renal tumour. EJNMMI Res. 2016;6:76. https://doi.org/10.1186/s13550-016-0231-6.
Zhang X, Zhang G, Xu L, Bai X, Lu X, Yu S, et al. Utilisation of virtual non-contrast images and virtual mono-energetic images acquired from dual-layer spectral CT for renal cell carcinoma: image quality and radiation dose. Insights Imaging. 2022;13:12. https://doi.org/10.1186/s13244-021-01146-8.
Zhang GM, Shi B, Xue HD, Ganeshan B, Sun H, Jin ZY. Can quantitative CT texture analysis be used to differentiate subtypes of renal cell carcinoma? Clin Radiol. 2019;74:287–94. https://doi.org/10.1016/j.crad.2018.11.009.
Jiang C, Kong Z, Liu S, Feng S, Zhang Y, Zhu R, et al. Fusion radiomics features from conventional MRI predict MGMT promoter methylation status in lower grade gliomas. Eur J Radiol. 2019;121:108714. https://doi.org/10.1016/j.ejrad.2019.108714.
Woythal N, Arsenic R, Kempkensteffen C, Miller K, Janssen JC, Huang K, et al. Immunohistochemical validation of PSMA expression measured by 68Ga-PSMA PET/CT in primary prostate cancer. J Nucl Med. 2018;59:238–43. https://doi.org/10.2967/jnumed.117.195172.
Spatz S, Tolkach Y, Jung K, Stephan C, Busch J, Ralla B, et al. Comprehensive evaluation of prostate specific membrane antigen expression in the vasculature of renal tumors: implications for imaging studies and prognostic role. J Urol. 2018;199:370–7. https://doi.org/10.1016/j.juro.2017.08.079.
Al-Ahmadie HA, Olgac S, Gregor PD, Tickoo SK, Fine SW, Kondagunta GV, et al. Expression of prostate-specific membrane antigen in renal cortical tumors. Mod Pathol. 2008;21:727–32. https://doi.org/10.1038/modpathol.2008.42.
Liu Y. The place of FDG PET/CT in renal cell carcinoma: value and limitations. Front Oncol. 2016;6:201. https://doi.org/10.3389/fonc.2016.00201.
Takahashi M, Kume H, Koyama K, Nakagawa T, Fujimura T, Morikawa T, et al. Preoperative evaluation of renal cell carcinoma by using 18F-FDG PET/CT. Clin Nucl Med. 2015;40:936–40. https://doi.org/10.1097/RLU.0000000000000875.
Miyakita H, Tokunaga M, Onda H, Usui Y, Kinoshita H, Kawamura N, et al. Significance of 18F-fluorodeoxyglucose positron emission tomography (FDG-PET) for detection of renal cell carcinoma and immunohistochemical glucose transporter 1 (GLUT-1) expression in the cancer. Int J Urol. 2002;9:15–8. https://doi.org/10.1046/j.1442-2042.2002.00416.x.
Sawicki LM, Buchbender C, Boos J, Giessing M, Ermert J, Antke C, et al. Diagnostic potential of PET/CT using a 68Ga-labelled prostate-specific membrane antigen ligand in whole-body staging of renal cell carcinoma: initial experience. Eur J Nucl Med Mol Imaging. 2017;44:102–7. https://doi.org/10.1007/s00259-016-3360-2.
Li Y, Zheng R, Zhang Y, Huang C, Tian L, Liu R, et al. Special issue “the advance of solid tumor research in China”: 68Ga-PSMA-11 PET/CT for evaluating primary and metastatic lesions in different histological subtypes of renal cell carcinoma. Int J Cancer. 2022. https://doi.org/10.1002/ijc.34189.
Liu Y, Wang G, Yu H, Wu Y, Lin M, Gao J, et al. Comparison of 18F-DCFPyL and 18F-FDG PET/computed tomography for the restaging of clear cell renal cell carcinoma: preliminary results of 15 patients. Nucl Med Commun. 2020;41:1299–305. https://doi.org/10.1097/MNM.0000000000001285.
Soret M, Bacharach SL, Buvat I. Partial-volume effect in PET tumor imaging. J Nucl Med. 2007;48:932–45. https://doi.org/10.2967/jnumed.106.035774.
Erdi YE, Nehmeh SA, Pan T, Pevsner A, Rosenzweig KE, Mageras G, et al. The CT motion quantitation of lung lesions and its impact on PET-measured SUVs. J Nucl Med. 2004;45:1287–92.
Funding
This study was supported by the Fundamental Research Funds for the Central Universities (3332022110), the National High Level Hospital Clinical Research Funding (2022-PUMCH-C-002), Chinese Academy of Medical Science Innovation Fund for Medical Sciences (2022-I2M-C&T-A-008, 2022-I2M-2–002, 2021-I2M-1–016), Beijing Natural Science Foundation (M22035), and the National Natural Science Foundation of China (82272046, 81871392).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
Ethical approval was obtained from the Institute Review Board of Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College (no. ZS-2532), and this study was conducted in accordance with the principles of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all participants included in the study.
Conflict of interest
Hank F. Kung is the founder of Five Eleven Pharma, which holds the patent rights for [68Ga]Ga-P16-093 and related technology. Other authors have no conflicts of interest or relevant financial activities to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - Genitourinary
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, G., Li, L., Wang, J. et al. Head-to-head comparison of [68Ga]Ga-P16-093 and 2-[18F]FDG PET/CT in patients with clear cell renal cell carcinoma: a pilot study. Eur J Nucl Med Mol Imaging 50, 1499–1509 (2023). https://doi.org/10.1007/s00259-022-06101-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-06101-3