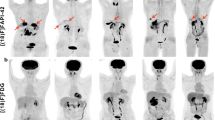
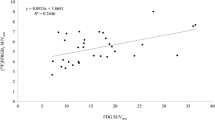
Abstract
Purpose
[18F]FDG PET/CT to detect unknown primary lesions is essential for clinical management but still has limitations. [68Ga]Ga-FAPI is a tumor-stromal imaging agent that provides a promising alternative to [18F]FDG for the assessment of malignancies. We aimed to investigate whether [68Ga]Ga-FAPI PET/CT has an additional role in identifying unknown primary lesions with negative or equivocal [18F] FDG PET/CT results.
Methods
This single-center prospective clinical study was conducted between March 2020 and March 2022 at Southwest Medical University Hospital. Patients underwent [18F]FDG PET/CT for the identification of unknown primary lesions. They underwent repeat [68Ga]Ga-FAPI PET/CT when [18F]FDG PET/CT results were negative or equivocal. Histopathological examination, surgery, or clinical follow-up (at least 3 months) for FAPI-positive lesions. The diagnostic efficacy of [68Ga]Ga-FAPI in identifying unknown primary lesions was evaluated.
Results
A total of 44 participants (median age, 57 ± 12 [SD]; 22 [50%] men) were evaluated. Thirteen of the 44 patients had equivocal [18F]FDG PET/CT findings, while the diagnosis was clear on [68Ga]Ga-FAPI PET/CT. [68Ga]Ga-FAPI PET/CT also revealed primary lesions in additional 17 patients with negative [18F]FDG PET/CT findings. In fourteen of 44 patients, no primary lesion was detected by either tracer. On this basis, we analyzed 94 lymph node metastatic lesions. The mean SUVmax of lymph node metastases on [68Ga] Ga-FAPI PET/CT and [18F]FDG PET/CT were 9.2 ± 5.1, 7.9 ± 4.8 (p = 0.03) and the mean TBR were 9.1 ± 5.2, 4.9 ± 3.1 (p < 0.01), respectively.
Conclusion
[68Ga]Ga-FAPI PET/CT showed great potential for identifying unknown primary lesions and has the potential to improve the detection rate of unknown primary lesions with negative or equivocal for [18F]FDG findings.
Trial registration
ClinicalTrial.gov. Identifier: ChiCTR2100044131





Similar content being viewed by others
References
Tomuleasa C, Zaharie F, Muresan MS, Pop L, Fekete Z, Dima D, et al. How to diagnose and treat a cancer of unknown primary site. J Gastrointestin Liver Dis. 2017;26(69-79) https://doi.org/10.15403/jgld.2014.1121.261.haz.
Hamson EJ, Keane FM, Tholen S, Schilling O, Gorrell MD. Understanding fibroblast activation protein (FAP): substrates, activities, expression and targeting for cancer therapy. Proteomics Clin Appl. 2014;8:454–63. https://doi.org/10.1002/prca.201300095.
Terry SY, Koenders MI, Franssen GM, Nayak TK, Freimoser-Grundschober A, Klein C, et al. Monitoring therapy response of experimental arthritis with radiolabeled tracers targeting fibroblasts, macrophages, or integrin alphavbeta3. J Nucl Med. 2016;57:467–72. https://doi.org/10.2967/jnumed.115.162628.
Waumans Y, Baerts L, Kehoe K, Lambeir AM, De Meester I. The dipeptidyl peptidase family, prolyl oligopeptidase, and prolyl carboxypeptidase in the immune system and inflammatory disease, including atherosclerosis. Front Immunol. 2015;6:387. https://doi.org/10.3389/fimmu.2015.00387.
Kalluri R. The biology and function of fibroblasts in cancer. Nat Rev Cancer. 2016;16:582–98. https://doi.org/10.1038/nrc.2016.73.
Chen WT, Kelly T. Seprase complexes in cellular invasiveness. Cancer metastasis reviews. 2003;22:259–69. https://doi.org/10.1023/a:1023055600919.
Kelly T. Fibroblast activation protein-alpha and dipeptidyl peptidase IV (CD26): cell-surface proteases that activate cell signaling and are potential targets for cancer therapy. Drug Resist Updat. 2005;8:51–8. https://doi.org/10.1016/j.drup.2005.03.002.
Mueller SC, Ghersi G, Akiyama SK, Sang QX, Howard L, Pineiro-Sanchez M, et al. A novel protease-docking function of integrin at invadopodia. J Biol Chem. 1999;274:24947–52. https://doi.org/10.1074/jbc.274.35.24947.
Loktev A, Lindner T, Mier W, Debus J, Altmann A, Jager D, et al. A tumor-imaging method targeting cancer-associated fibroblasts. J Nucl Med. 2018;59:1423-1429. https://doi.org/10.2967/jnumed.118.210435.
Lindner T, Loktev A, Altmann A, Giesel F, Kratochwil C, Debus J, et al. Development of quinoline-based theranostic ligands for the targeting of fibroblast activation protein. J Nucl Med. 2018;59:1415–22. https://doi.org/10.2967/jnumed.118.210443.
Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, et al. (68)Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med. 2019;60:386–92. https://doi.org/10.2967/jnumed.118.215913.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, et al. (68)Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med. 2019;60:801–5. https://doi.org/10.2967/jnumed.119.227967.
Chen H, Pang Y, Wu J, Zhao L, Hao B, Wu J, et al. Comparison of [(68)Ga]Ga-DOTA-FAPI-04 and [(18)F] FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur J Nucl Med Mol Imaging. 2020;47:1820–32. https://doi.org/10.1007/s00259-020-04769-z.
Zhao L, Chen S, Lin L, Sun L, Wu H, Lin Q, et al. [(68)Ga]Ga-DOTA-FAPI-04 improves tumor staging and monitors early response to chemoradiotherapy in a patient with esophageal cancer. Eur J Nucl Med Mol Imaging. 2020;47:3188–9. https://doi.org/10.1007/s00259-020-04818-7.
Karni RJ, Rich JT, Sinha P, Haughey BH. Transoral laser microsurgery: a new approach for unknown primaries of the head and neck. Laryngoscope. 2011;121:1194–201. https://doi.org/10.1002/lary.21743.
Haas I, Hoffmann TK, Engers R, Ganzer U. Diagnostic strategies in cervical carcinoma of an unknown primary (CUP). Eur Arch Otorhinolaryngol. 2002;259:325–33. https://doi.org/10.1007/s00405-002-0470-1.
Davis KS, Byrd JK, Mehta V, Chiosea SI, Kim S, Ferris RL, et al. Occult primary head and neck squamous cell carcinoma: utility of discovering primary lesions. Otolaryngol Head Neck Surg. 2014;151:272–8. https://doi.org/10.1177/0194599814533494.
Acquarelli MJ, Matsunaga RS, Cruze K. Metastatic carcinoma of the neck of unknown primary origin. Laryngoscope. 1961;71:962–74. https://doi.org/10.1288/00005537-196108000-00006.
Muraki AS, Mancuso AA, Harnsberger HR. Metastatic cervical adenopathy from tumors of unknown origin: the role of CT. Radiology. 1984;152:749–53. https://doi.org/10.1148/radiology.152.3.6463256.
Han A, Xue J, Hu M, Zheng J, Wang X. Clinical value of 18F-FDG PET-CT in detecting primary tumor for patients with carcinoma of unknown primary. Cancer Epidemiol. 2012;36:470–5. https://doi.org/10.1016/j.canep.2012.03.002.
Nikolova PN, Hadzhiyska VH, Mladenov KB, Ilcheva MG, Veneva S, Grudeva VV, et al. The impact of 18F-FDG PET/CT in the clinical management of patients with lymph node metastasis of unknown primary origin. Neoplasma. 2021;68:180–9. https://doi.org/10.4149/neo_2020_200315N263.
Cengiz A, Goksel S, Yurekli Y. Diagnostic Value of (18)F-FDG PET/CT in patients with carcinoma of unknown primary. Mol Imaging Radionucl Ther. 2018;27:126–32. https://doi.org/10.4274/mirt.64426.
Albertson M, Chandra S, Sayed Z, Johnson C. PET/CT evaluation of head and neck cancer of unknown primary. Semin Ultrasound CT MR. 2019;40:414–23. https://doi.org/10.1053/j.sult.2019.07.005.
Gu B, Xu X, Zhang J, Ou X, Xia Z, Guan Q, et al. The added value of 68Ga-FAPI PET/CT in patients with head and neck cancer of unknown primary with 18F-FDG–negative findings. Journal of Nuclear Medicine. 2022;63:875–81. https://doi.org/10.2967/jnumed.121.262790.
Khan MA, Combs CS, Brunt EM, Lowe VJ, Wolverson MK, Solomon H, et al. Positron emission tomography scanning in the evaluation of hepatocellular carcinoma. Journal of hepatology. 2000;32:792–7. https://doi.org/10.1016/s0168-8278(00)80248-2.
Trojan J, Schroeder O, Raedle J, Baum RP, Herrmann G, Jacobi V, et al. Fluorine-18 FDG positron emission tomography for imaging of hepatocellular carcinoma. The American journal of gastroenterology. 1999;94:3314–9. https://doi.org/10.1111/j.1572-0241.1999.01544.x.
Almuhaideb A, Papathanasiou N, Bomanji J. 18F-FDG PET/CT imaging in oncology. Ann Saudi Med. 2011;31:3–13. https://doi.org/10.4103/0256-4947.75771.
Akin EA, Qazi ZN, Osman M, Zeman RK. Clinical impact of FDG PET/CT in alimentary tract malignancies: an updated review. Abdom Radiol (NY). 2020;45:1018–35. https://doi.org/10.1007/s00261-020-02447-0.
Yun M. Imaging of gastric cancer metabolism using 18 F-FDG PET/CT. J Gastric Cancer. 2014;14:1–6. https://doi.org/10.5230/jgc.2014.14.1.1.
Pang Y, Zhao L, Luo Z, Hao B, Wu H, Lin Q, et al. Comparison of (68)Ga-FAPI and (18)F-FDG uptake in gastric, duodenal, and colorectal cancers. Radiology. 2021;298:393–402. https://doi.org/10.1148/radiol.2020203275.
Guo W, Pang Y, Yao L, Zhao L, Fan C, Ke J, et al. Imaging fibroblast activation protein in liver cancer: a single-center post hoc retrospective analysis to compare [(68)Ga]Ga-FAPI-04 PET/CT versus MRI and [(18)F]-FDG PET/CT. Eur J Nucl Med Mol Imaging. 2021;48:1604–17. https://doi.org/10.1007/s00259-020-05095-0.
Coccolini F, Gheza F, Lotti M, Virzi S, Iusco D, Ghermandi C, et al. Peritoneal carcinomatosis. World J Gastroenterol. 2013;19:6979–94. https://doi.org/10.3748/wjg.v19.i41.6979.
Stahl A, Ott K, Weber WA, Becker K, Link T, Siewert JR, et al. FDG PET imaging of locally advanced gastric carcinomas: correlation with endoscopic and histopathological findings. Eur J Nucl Med Mol Imaging. 2003;30:288–95. https://doi.org/10.1007/s00259-002-1029-5.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Qiaoqiao Shu, Maoxue Deng, Mei Hu, MengNa Liu, Xi Chen, Yue Chen, and Liang Cai. The first draft of the manuscript was written by Qiaoqiao Shu, Maoxue Deng, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent to participate
The patient gave informed consent to using clinical data for research and scientifc purposes.
Informed consent
Informed consent was obtained from the patient for the publication of his case/report and accompanying images.
Consent for publication
The patient gave signed consent to using clinical data for scientifc publication.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - General.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shu, Q., Deng, M., Hu, M. et al. The additional role of [68Ga]Ga-FAPI-04 PET/CT in patients with unknown primary lesion with a negative or equivocal [18F]FDG. Eur J Nucl Med Mol Imaging 50, 1442–1452 (2023). https://doi.org/10.1007/s00259-022-06095-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-06095-y