Abstract
Purpose
PET has been important for monitoring recurrence and metastasis of Gastrointestinal Stromal Tumors (GISTs) and the selection of therapeutic strategies. A significant portion of GISTs lesions show negative FDG uptake and therefore calls for more tumor-specific imaging biomarkers. This study compared the imaging performance of [18F]FAPI-42 PET/CT and [18F]FDG PET/CT in recurrent or metastatic gastrointestinal stromal tumors (R/M GISTs).
Methods
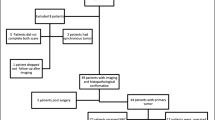
This study retrospectively included 35 patients with R/M GISTs who underwent both FAPI PET/CT and FDG PET/CT. The definite diagnosis was confirmed by pathology or follow-up drug treatment effects. The differences in detection rates and tumor-to-background SUVmax ratio (SUVTBR) of different locations between dual-tracer PET/CT were compared. Factors including tumor size, degree of enhancement, type of gene mutation, and targeted treatment potentially influencing the uptake of both tracers were assessed. The excised lesions (n = 3) underwent immunohistochemical staining to verify FAP expression in the tissue.
Results
A total of 106 lesions in 35 patients were identified, out of which 38/106 (35.8%) lesions (FAPI + /FDG −) were additionally detected by FAPI PET/CT as compared to that by FDG, including 26 liver metastases, ten peritoneal metastases, one gastrointestinal recurrence, and one bone metastasis. The positive detection rate of FAPI PET/CT for recurrent or metastatic GISTs was higher than that of FDG (80.2% vs. 53.8%, P< 0.001), especially in liver metastases (87.5% vs. 33.3%, P< 0.001). Moreover, the SUVTBR of liver metastases of GISTs in FAPI PET/CT was higher than that in FDG [2.4 (0.3 to 11.2) vs. 0.9 (0.3 to 6.5), P< 0.001]. The longest diameter of tumors in the FDG-positive group was higher than that of the FDG-negative group (P= 0.005); still, it did not differ between the FAPI-positive group and the FAPI-negative group. No difference in the degree of enhancement was observed between both tracers’ positive and negative groups. Besides, the SUVTBR of FDG but not FAPI differed significantly among various gene mutations (P< 0.001) as well as the targeted therapy and no targeted therapy groups (P= 0.001). FAP was expressed in R/M GISTs, and the uptake of FAPI corresponded to the level of FAP expression.
Conclusion
In conclusion, FAPI for imaging of R/M GISTs could be superior to FDG, specifically for liver metastases. The uptake of FAPI could reflect the level of FAP expression, and it was independent of tumor size, degree of enhancement, type of gene mutation, and targeted therapy as compared to FDG.





Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Change history
16 September 2022
A Correction to this paper has been published: https://doi.org/10.1007/s00259-022-05965-9
References
Blay JY, Kang YK, Nishida T, von Mehren M. Gastrointestinal stromal tumours. Nat Rev Dis Primers. 2021;7:22. https://doi.org/10.1038/s41572-021-00254-5.
Vallilas C, Sarantis P, Kyriazoglou A, Koustas E, Theocharis S, Papavassiliou AG et al. Gastrointestinal stromal tumors (GISTs): novel therapeutic strategies with immunotherapy and small molecules. Int J Mol Sci. 2021; 22. https://doi.org/10.3390/ijms22020493.
Ma GL, Murphy JD, Martinez ME, Sicklick JK. Epidemiology of gastrointestinal stromal tumors in the era of histology codes: results of a population-based study. Cancer Epidemiol Biomarkers Prev. 2015;24:298–302. https://doi.org/10.1158/1055-9965.EPI-14-1002.
Kelly CM, Gutierrez Sainz L, Chi P. The management of metastatic GIST: current standard and investigational therapeutics. J Hematol Oncol. 2021;14:2. https://doi.org/10.1186/s13045-020-01026-6.
Soreide K, Sandvik OM, Soreide JA, Giljaca V, Jureckova A, Bulusu VR. Global epidemiology of gastrointestinal stromal tumours (GIST): a systematic review of population-based cohort studies. Cancer Epidemiol. 2016;40:39–46. https://doi.org/10.1016/j.canep.2015.10.031.
Mantese G. Gastrointestinal stromal tumor: epidemiology, diagnosis, and treatment. Curr Opin Gastroenterol. 2019;35:555–9. https://doi.org/10.1097/MOG.0000000000000584.
Iwatsuki M, Harada K, Iwagami S, Eto K, Ishimoto T, Baba Y, et al. Neoadjuvant and adjuvant therapy for gastrointestinal stromal tumors. Ann Gastroenterol Surg. 2019;3:43–9. https://doi.org/10.1002/ags3.12211.
Alessandrino F, Tirumani SH, Jagannathan JP, Ramaiya NH. Imaging surveillance of gastrointestinal stromal tumour: current recommendation by National Comprehensive Cancer Network and European Society of Medical Oncology-European Reference Network for rare adult solid cancers. Clin Radiol. 2019;74:746–55. https://doi.org/10.1016/j.crad.2019.06.015.
Revheim M-E, Hole KH, Mo T, Bruland ØS, Reitan E, Julsrud L et al. Multimodal functional imaging for early response assessment in patients with gastrointestinal stromal tumor treated with tyrosine kinase inhibitors. Acta Radiologica (Stockholm, Sweden: 1987). 2022; 63. https://doi.org/10.1177/02841851211027389.
Hahn S, Bauer S, Heusner TA, Ebeling P, Hamami ME, Stahl A, et al. Postoperative FDG-PET/CT staging in GIST: is there a benefit following R0 resection? Eur J Radiol. 2011;80:670–4. https://doi.org/10.1016/j.ejrad.2010.09.017.
Choi H, Charnsangavej C, de Castro FS, Tamm EP, Benjamin RS, Johnson MM, et al. CT evaluation of the response of gastrointestinal stromal tumors after imatinib mesylate treatment: a quantitative analysis correlated with FDG PET findings. AJR Am J Roentgenol. 2004;183:1619–28.
Kratochwil C, Flechsig P, Lindner T, Abderrahim L, Altmann A, Mier W, et al. (68)Ga-FAPI PET/CT: tracer uptake in 28 different kinds of cancer. J Nucl Med. 2019;60:801–5. https://doi.org/10.2967/jnumed.119.227967.
Gu B, Liu X, Wang S, Xu X, Liu X, Hu S, et al. Head-to-head evaluation of [18F]FDG and [68 Ga]Ga-DOTA-FAPI-04 PET/CT in recurrent soft tissue sarcoma. Eur J Nucl Med Mol. 2022;I(49):2889–901. https://doi.org/10.1007/s00259-022-05700-4.
Lan L, Liu H, Wang Y, Deng J, Peng D, Feng Y, et al. The potential utility of [(68) Ga]Ga-DOTA-FAPI-04 as a novel broad-spectrum oncological and non-oncological imaging agent-comparison with [(18)F]FDG. Eur J Nucl Med Mol Imaging. 2022;49:963–79. https://doi.org/10.1007/s00259-021-05522-w.
Hamson EJ, Keane FM, Tholen S, Schilling O, Gorrell MD. Understanding fibroblast activation protein (FAP): substrates, activities, expression and targeting for cancer therapy. Proteomics Clin Appl. 2014;8:454–63. https://doi.org/10.1002/prca.201300095.
Yoon H, Tang CM, Banerjee S, Yebra M, Noh S, Burgoyne AM, et al. Cancer-associated fibroblast secretion of PDGFC promotes gastrointestinal stromal tumor growth and metastasis. Oncogene. 2021;40:1957–73. https://doi.org/10.1038/s41388-021-01685-w.
Yoon H, Tang CM, Banerjee S, Delgado AL, Yebra M, Davis J, et al. TGF-β1-mediated transition of resident fibroblasts to cancer-associated fibroblasts promotes cancer metastasis in gastrointestinal stromal tumor. Oncogenesis. 2021;10:13. https://doi.org/10.1038/s41389-021-00302-5.
Tang SM, Shen CY, Yin Y, Yin XN, Cai ZL, Chen ZX, et al. FAP expression and its association with the prognosis of gastric stromal tumors. Sichuan Da Xue Xue Bao Yi Xue Ban. 2017;48(2):234–8 (Chinese).
Qiu L, Lan L, Liu H, Deng J, Chen Y. 68Ga-FAPI PET/CT detected non-FDG-avid gastric stromal tumor. Clin Nucl Med. 2022;47:226–7. https://doi.org/10.1097/RLU.0000000000003856.
Giesel FL, Kratochwil C, Lindner T, Marschalek MM, Loktev A, Lehnert W, et al. (68)Ga-FAPI PET/CT: biodistribution and preliminary dosimetry estimate of 2 DOTA-containing FAP-targeting agents in patients with various cancers. J Nucl Med. 2019;60:386–92. https://doi.org/10.2967/jnumed.118.215913.
Pang Y, Zhao L, Luo Z, Hao B, Wu H, Lin Q, et al. Comparison of (68)Ga-FAPI and (18)F-FDG uptake in gastric, duodenal, and colorectal cancers. Radiology. 2021;298:393–402. https://doi.org/10.1148/radiol.2020203275.
Scola D, Bahoura L, Copelan A, Shirkhoda A, Sokhandon F. Getting the GIST: a pictorial review of the various patterns of presentation of gastrointestinal stromal tumors on imaging. Abdom Radiol (NY). 2017;42:1350–64. https://doi.org/10.1007/s00261-016-1025-z.
Shankar S, vanSonnenberg E, Desai J, Dipiro PJ, Van Den Abbeele A, Demetri GD. Gastrointestinal stromal tumor: new nodule-within-a-mass pattern of recurrence after partial response to imatinib mesylate. Radiology. 2005;235:892–8. https://doi.org/10.1007/s00259-022-05700-4.
Choi H. Response evaluation of gastrointestinal stromal tumors. Oncologist. 2008;13(Suppl 2):4–7. https://doi.org/10.1634/theoncologist.13-S2-4.
Giesel FL, Adeberg S, Syed M, Lindner T, Jiménez-Franco LD, Mavriopoulou E, et al. FAPI-74 PET/CT Using Either (18)F-AlF or Cold-Kit (68)Ga labeling: biodistribution, radiation dosimetry, and tumor delineation in lung cancer patients. J Nucl Med. 2021;62:201–7. https://doi.org/10.2967/jnumed.120.245084.
Liegl B, Kepten I, Le C, Zhu M, Demetri GD, Heinrich MC, et al. Heterogeneity of kinase inhibitor resistance mechanisms in GIST. J Pathol. 2008;216:64–74. https://doi.org/10.1002/path.2382.
Albano D, Mattia B, Giubbini R, Bertagna F. Role of 18F-FDG PET/CT in restaging and follow-up of patients with GIST. Abdom Radiol (NY). 2020;45:644–51. https://doi.org/10.1007/s00261-019-02274-y.
Miyake KK, Nakamoto Y, Mikami Y, Tanaka S, Higashi T, Tadamura E, et al. The predictive value of preoperative (18)F-fluorodeoxyglucose PET for postoperative recurrence in patients with localized primary gastrointestinal stromal tumour. Eur Radiol. 2016;26:4664–74. https://doi.org/10.1007/s00330-016-4242-5.
Hassanzadeh-Rad A, Yousefifard M, Katal S, Asady H, Fard-Esfahani A, Moghadas Jafari A, et al. The value of (18) F-fluorodeoxyglucose positron emission tomography for prediction of treatment response in gastrointestinal stromal tumors: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2016;31:929–35. https://doi.org/10.1111/jgh.13247.
Goerres GW, Stupp R, Barghouth G, Hany TF, Pestalozzi B, Dizendorf E, et al. The value of PET, CT and in-line PET/CT in patients with gastrointestinal stromal tumours: long-term outcome of treatment with imatinib mesylate. Eur J Nucl Med Mol Imaging. 2005;32:153–62. https://doi.org/10.1007/s00259-004-1633-7.
Albano D, Bosio G, Tomasini D, Bonu M, Giubbini R, Bertagna F. Metabolic behavior and prognostic role of pretreatment 18F-FDG PET/CT in gist. Asia Pac J Clin Oncol. 2020;16:e207–15. https://doi.org/10.1111/ajco.13366.
McAuliffe JC, Hunt KK, Lazar AJ, Choi H, Qiao W, Thall P, et al. A randomized, phase II study of preoperative plus postoperative imatinib in GIST: evidence of rapid radiographic response and temporal induction of tumor cell apoptosis. Ann Surg Oncol. 2009;16:910–9. https://doi.org/10.1245/s10434-008-0177-7.
Dimitrakopoulou-Strauss A, Hohenberger P, Haberkorn U, Mäcke HR, Eisenhut M, Strauss LG. 68Ga-labeled bombesin studies in patients with gastrointestinal stromal tumors: comparison with 18F-FDG. J Nucl Med. 2007;48:1245–50. https://doi.org/10.2967/jnumed.106.038091.
Braat A, Goldschmeding R, Brosens LAA, Vriens MR, de Keizer B. Gastrointestinal stromal tumour detection with somatostatin receptor imaging, (68)Ga-HA-DOTATATE PET-CT. Lancet Oncol. 2017;18:e185. https://doi.org/10.1016/s1470-2045(17)30078-5.
Sasikumar A, Joy A, Pillai M, Bindu S, Sudin Sr. 68Ga-PSMA uptake in an incidentally detected gastrointestinal stromal tumor in a case of suspected carcinoma prostate. Clin Nucl Med. 2017;42:e447–8. https://doi.org/10.1097/rlu.0000000000001774.
Pretze M, Reffert L, Diehl S, Schönberg SO, Wängler C, Hohenberger P, et al. GMP-compliant production of [(68)Ga]Ga-NeoB for positron emission tomography imaging of patients with gastrointestinal stromal tumor. EJNMMI Radiopharm Chem. 2021;6:22. https://doi.org/10.1186/s41181-021-00137-w.
Gruber L, Jiménez-Franco LD, Decristoforo C, Uprimny C, Glatting G, Hohenberger P, et al. MITIGATE-NeoBOMB1, a phase I/IIa study to evaluate safety, pharmacokinetics, and preliminary imaging of (68)Ga-NeoBOMB1, a gastrin-releasing peptide receptor antagonist, in GIST Patients. J Nucl Med. 2020;61:1749–55. https://doi.org/10.2967/jnumed.119.238808.
Raghavan K, Flavell RR, Westphalen AC, Behr SC. Gastrointestinal stromal tumor incidentally detected on 18F-fluciclovine PET/CT. Clin Nucl Med. 2021;46:345–7. https://doi.org/10.1097/rlu.0000000000003426.
Chen H, Pang Y, Wu J, Zhao L, Hao B, Wu J, et al. Comparison of [(68)Ga]Ga-DOTA-FAPI-04 and [(18)F] FDG PET/CT for the diagnosis of primary and metastatic lesions in patients with various types of cancer. Eur J Nucl Med Mol Imaging. 2020;47:1820–32. https://doi.org/10.1007/s00259-020-04769-z.
Qin C, Song Y, Liu X, Gai Y, Liu Q, Ruan W, et al. Increased uptake of (68)Ga-DOTA-FAPI-04 in bones and joints: metastases and beyond. Eur J Nucl Med Mol Imaging. 2022;49:709–20. https://doi.org/10.1007/s00259-021-05472-3.
Kosemehmetoglu K, Kaygusuz G, Fritchie K, Aydin O, Yapicier O, Coskun O, et al. Clinical and pathological characteristics of gastrointestinal stromal tumor (GIST) metastatic to bone. Virchows Arch. 2017;471:77–90. https://doi.org/10.1007/s00428-017-2138-7.
Park JW, Cho CH, Jeong DS, Chae HD. Role of F-fluoro-2-deoxyglucose positron emission tomography in gastric GIST: predicting malignant potential pre-operatively. J Gastric Cancer. 2011;11:173–9. https://doi.org/10.5230/jgc.2011.11.3.173.
Yoo J, Kim SH, Han JK. Multiparametric MRI and (18)F-FDG PET features for differentiating gastrointestinal stromal tumors from benign gastric subepithelial lesions. Eur Radiol. 2020;30:1634–43. https://doi.org/10.1007/s00330-019-06534-9.
Prior JO, Montemurro M, Orcurto MV, Michielin O, Luthi F, Benhattar J, et al. Early prediction of response to sunitinib after imatinib failure by 18F-fluorodeoxyglucose positron emission tomography in patients with gastrointestinal stromal tumor. J Clin Oncol. 2009;27:439–45. https://doi.org/10.1200/jco.2008.17.2742.
Kratochwil C, Giesel FL, Rathke H, Fink R, Dendl K, Debus J, et al. [(153)Sm]Samarium-labeled FAPI-46 radioligand therapy in a patient with lung metastases of a sarcoma. Eur J Nucl Med Mol Imaging. 2021;48:3011–3. https://doi.org/10.1007/s00259-021-05273-8.
Ferdinandus J, Costa PF, Kessler L, Weber M, Hirmas N, Kostbade K, et al. Initial clinical experience with (90)Y-FAPI-46 radioligand therapy for advanced-stage solid tumors: a case series of 9 patients. J Nucl Med. 2022;63:727–34. https://doi.org/10.2967/jnumed.121.262468.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the First Affiliated Hospital of Sun Yat-sen University. Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors Chunhui Wu, Xinhua Zhang, and Yu Zeng contributed equally as first authors.
This article is part of the Topical Collection on Oncology - Digestive tract
The original online version of this article was revised: The authors regret that some of the reference citations in the original article are incorrect. The sequence number incorrectly presented are as follows: [1, 2, 3] as [1], [3, 4, 5] as [3], [1, 5] as [1]; [9, 10, 11] as [9], [12, 13, 14] as [12],[16, 17] as [16], [18, 19] as [18], [20, 21] as [20], [27, 28, 29] as [27], [9, 30, 31] as [31], [36, 37] as [36], [20, 25] as [20].
The original article has been corrected.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, C., Zhang, X., Zeng, Y. et al. [18F]FAPI-42 PET/CT versus [18F]FDG PET/CT for imaging of recurrent or metastatic gastrointestinal stromal tumors. Eur J Nucl Med Mol Imaging 50, 194–204 (2022). https://doi.org/10.1007/s00259-022-05955-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05955-x