Abstract
Purpose
4-18F-Fluoro-m-hydroxyphenethylguanidine (18F-4F-MHPG) and 3-18F-fluoro-p-hydroxyphenethylguanidine (18F-3F-PHPG) were developed for quantifying regional cardiac sympathetic nerve density using tracer kinetic analysis. The aim of this study was to evaluate their performance in cardiomyopathy patients.
Methods
Eight cardiomyopathy patients were scanned with 18F-4F-MHPG and 18F-3F-PHPG. Also, regional resting perfusion was assessed with 13N-ammonia. 18F-4F-MHPG and 18F-3F-PHPG kinetics were analyzed using the Patlak graphical method to obtain Patlak slopes Kp (mL/min/g) as measures of regional nerve density. Patlak slope polar maps were used to evaluate the pattern and extent of cardiac denervation. For comparison, “retention index” (RI) values (mL blood/min/mL tissue) were also calculated and used to assess denervation. Perfusion polar maps were used to estimate the extent of hypoperfusion.
Results
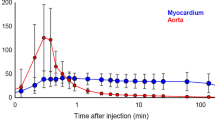
Patlak analysis of 18F-4F-MHPG and 18F-3F-PHPG kinetics was successful in all subjects, demonstrating the robustness of this approach in cardiomyopathy patients. Substantial regional denervation was observed in all subjects, ranging from 25 to 74% of the left ventricle. Denervation zones were equal to or larger than the size of corresponding areas of hypoperfusion. The two tracers provided comparable metrics of regional nerve density and the extent of left ventricular denervation. 18F-4F-MHPG exhibited faster liver clearance than 18F-3F-PHPG, reducing spillover from the liver into the inferior wall. 18F-4F-MHPG was also metabolized more consistently in plasma, which may allow application of population-averaged metabolite corrections.
Conclusion
The advantages of 18F-4F-MHPG (more rapid liver clearance, more consistent metabolism in plasma) make it the better imaging agent to carry forward into future clinical studies in patients with cardiomyopathy.
Trial registration: Registered at the ClinicalTrials.gov website (NCT02669563).








Similar content being viewed by others
References
Adabag AS, Luepker RV, Roger VL, Gersh BJ. Sudden cardiac death: epidemiology and risk factors. Nat Rev Cardiol. 2010;7:216–25.
Saour B, Smith B, Yancy CW. Heart failure and sudden cardiac death. Card Electrophysiol Clin. 2017;9:709–23.
Buxton AE, Waks JW, Shen C, Chen PS. Risk stratification for sudden cardiac death in North America – current perspectives. J Electrocard. 2016;49:817–23.
Passman R, Goldberger JJ. Predicting the future: risk stratification for sudden cardiac death in patients with left ventricular dysfunction. Circulation. 2012;125:3013–37.
Koneru JN, Swerdlow CD, Wood MA, Ellenbogen KA. Minimizing inappropriate or “unnecessary” implantable cardioverter-defibrillator shocks: appropriate programming. Circ Arrhythm Electrophysiol. 2011;4:778–90.
Srinivasa NT, Schilling RJ. Sudden cardiac death and arrhythmias. Arrhythm Electrophysiol Rev. 2018;7:111–7.
Allman KC, Wieland DM, Muzik O, Degrado TR, Wolfe ER, Schwaiger M. Carbon-11 hydroxyephedrine with positron emission tomography for serial assessment of cardiac adrenergic neuronal function after acute myocardial infarction in humans. J Am Coll Cardiol. 1993;22:368–75.
Gardner RT, Ripplinger CM, Myles RC, Habecker BA. Molecular mechanisms of sympathetic remodeling and arrhythmias. Circ Arrhythm Electrophysiol. 2016;9:e001359.
Jacobson AF, Senior R, Cerqueira MD, Wong ND, Thomas GS, Lopez VA, et al. Myocardial iodine-123 meta-iodobenzylguanidine imaging and cardiac events in heart failure: results of the prospective ADMIRE-HF (AdreView Myocardial Imaging for Risk Evaluation in Heart Failure) study. J Am Coll Cardiol. 2010;55:2212–21.
Fallavollita JA, Heavey BM, Luisi AJ, Michalek SM, Baldwa S, Mashtare TL, et al. Regional myocardial sympathetic denervation predicts the risk of sudden cardiac arrest in ischemic cardiomyopathy. J Am Coll Cardiol. 2014;63:141–9.
Cha YM, Chareonthaitawee P, Dong YX, Kemp BJ, Oh JK, Miyazak IC, et al. Cardiac sympathetic reserve and response to cardiac resynchronization therapy. Circ Heart Fail. 2011;4:339–44.
Martignani C, Diemberger I, Nanni C, Biffi M, Ziacchi M, Boschi S, et al. Cardiac resynchronization therapy and cardiac sympathetic function. Eur J Clin Invest. 2015;45:792–9.
Verschure DO, Poel E, De Vincentis G, Frantellizzi V, Nakajima K, Gheysens O, et al. The relation between cardiac 123I-mIBG scintigraphy and functional response 1 year after CRT implantation. Eur Heart J Cardiovasc Imaging. 2021;22:49–57.
Jang KS, Jung YW, Gu G, Koeppe RA, Sherman PS, Quesada CA, et al. 4-[18F]fluoro-m-hydroxyphenethylguanidine: a radiopharmaceutical for quantifying regional cardiac sympathetic nerve density with positron emission tomography. J Med Chem. 2013;56:7312–23.
Raffel DM, Jung YW, Gildersleeve DL, Sherman PS, Moskwa JJ, Tluczek LJ, et al. Radiolabeled phenethylguanidines: novel imaging agents for cardiac sympathetic neurons and adrenergic tumors. J Med Chem. 2007;50:2078–88.
Patlak CS, Blasberg RG. Graphical evaluation of blood-to-brain transfer constants from multiple-time uptake data. Generalizations J Cereb Blood Flow. 1985;5:584–90.
Raffel DM, Jung YW, Koeppe RA, Jang KS, Gu G, Scott PJH, et al. First-in-human studies of [18F]fluorohydroxyphenethylguanidines: positron emission tomography radiotracers for quantifying regional cardiac sympathetic nerve density. Circ Cardiovasc Imaging. 2018;11:e007965.
Raffel DM, Koeppe RA, Jung YW, Gu G, Jang KS, Sherman PS, et al. Quantification of cardiac sympathetic nerve density with N-11C-guanyl-meta-octopamine and tracer kinetic analysis. J Nucl Med. 2013;54:1645–52.
DeGrado TR, Hanson MW, Turkington TG, Delong DM, Brezinski DA, Vallée JP, et al. Estimation of myocardial blood flow for longitudinal studies with 13N-labeled ammonia and positron emission tomography. J Nucl Cardiol. 1996;3:494–507.
Kurz T, Richardt G, Hagl S, Seyfarth M, Schömig A. Two different mechanisms of noradrenaline release during normoxia and simulated ischemia in human cardiac tissue. J Mol Cell Cardiol. 1995;27:1161–72.
Lewandowski J, Symonides B, Gaciong Z, Sinski M. The effect of statins on sympathetic activity: a meta-analysis. Clin Auton Res. 2014;25:125–31.
Tatsumi M, Groshan K, Blakely RD, Richelson E. Pharmacological profile of antidepressants and related compounds at human monoamine transporters. Eur J Pharmacol. 1997;340:249–58.
Reis M, Aamo T, Ahlner J, Druid H. Reference concentrations of antidepressants. a compilation of postmortem and therapeutic levels. J Anal Toxicol. 2007;31:254–64.
Frishman WH, Saunders E. β-Adrenergic blockers. J Clin Hypertension. 2011;13:649–53.
Tank J, Diedrich A, Schroeder C, Stoffels M, Franke G, Sharma AM, et al. Limited effect of systemic β-blockade on sympathetic outflow. Hypertension. 2001;38:1377–81.
Cohen-Solal A, Jacobson AF, Piña IL. Beta blocker dose and markers of sympatheticactivation in heart failure patients: interrelationships and prognostic significance. ESC Heart Failure. 2017;4:499–506.
Krum H. Differentiation in the angiotensin II receptor 1 blocker class on autonomic function. Curr Hypertens Rep. 2001;3(Suppl I):S17–23.
Miller AJ, Arnold AC. The renin–angiotensin system in cardiovascular autonomic control: recent developments and clinical implications. Clin Auton Res. 2019;29:231–43.
Simões MV, Barthel P, Matsunari I, Nekolla SG, Schömig A, Schwaiger M, et al. Presence of sympathetically denervated but viable myocardium and its electrophysiologic correlates after early revascularised, acute myocardial infarction. Eur Heart J. 2004;25:551–7.
Bengel FM. Imaging of myocardial catecholamine uptake: toward robust absolute quantification [editorial]. Circ Cardiovasc Imaging. 2018;11:e008534.
Acknowledgements
We thank Dr. Peter Scott, Brian Hockley, Bradford Henderson, and Charles Schneider in the University of Michigan PET Radiopharmaceutical Production Program for their support in synthesizing the radiotracers. We also thank our clinical research coordinators Eric Puroll, James Pool, Mary Burton, and Tamara Harper for their many contributions.
Funding
This study was funded by a Mid-Stage Award (MTRAC-N019131) from the University of Michigan Fast Forward Medical Innovation (FFMI) Program and through a Services Award (RSA-000169) from the Science Moving Towards Research Translation and Therapy (SMARTT) Program of the National Heart, Lung and Blood Institute, National Institutes of Health, Bethesda, MD, USA.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The study was approved by the University of Michigan Institutional Review Board (HUM00105110). This article does not contain any studies with animals performed by any of the authors. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Cardiology.
Rights and permissions
About this article
Cite this article
Raffel, D.M., Crawford, T.C., Jung, YW. et al. Quantifying cardiac sympathetic denervation: first studies of 18F-fluorohydroxyphenethylguanidines in cardiomyopathy patients. Eur J Nucl Med Mol Imaging 49, 619–631 (2022). https://doi.org/10.1007/s00259-021-05517-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-021-05517-7