Abstract
Purpose
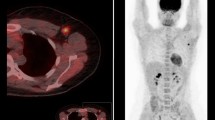
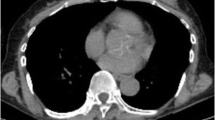
The objective of this study was to evaluate the role of 18F-FDG PET/CT in predicting overall survival in inflammatory breast cancer patients undergoing neoadjuvant chemotherapy.
Methods
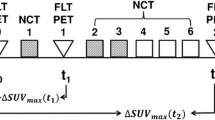
Included in this retrospective study were 53 patients with inflammatory breast cancer who had at least two PET/CT studies including a baseline study before the start of neoadjuvant chemotherapy. Univariate and multivariate analyses were performed to assess the effects on survival of the following factors: tumor maximum standardized uptake value (SUVmax) at baseline, preoperatively and at follow-up, decrease in tumor SUVmax, histological tumor type, grade, estrogen, progesterone, HER2/neu receptor status, and extent of disease at presentation including axillary nodal and distant metastases.
Results
By univariate analysis, survival was significantly associated with decrease in tumor SUVmax and tumor receptor status. Patients with decrease in tumor SUVmax had better survival (P = 0.02). Patients with a triple-negative tumor (P = 0.0006), a Her2/neu-negative tumor (P = 0.038) or an ER-negative tumor (P = 0.039) had worse survival. Multivariate analysis confirmed decrease in tumor SUVmax and triple-negative receptor status as significant predictors of survival. Every 10 % decrease in tumor SUVmax from baseline translated to a 15 % lower probability of death, and complete resolution of tumor FDG uptake translated to 80 % lower probability of death (P = 0.014). Patients with a triple-negative tumor had 4.11 times higher probability of death (P = 0.004).
Conclusion
Decrease in tumor SUVmax is an independent predictor of survival in patients with inflammatory breast cancer undergoing neoadjuvant chemotherapy. Further investigation with prospective studies is warranted to clarify its role in assessing response and altering therapy.



Similar content being viewed by others
References
Giordano SH, Hortobagyi GN. Inflammatory breast cancer. Clinical progress and the main problems that must be addressed. Breast Cancer Res. 2003;5:284–8.
Robertson F, Bondy M, Yang W, Yamauchi H, Wiggins S, Kamrudin S, et al. Inflammatory breast cancer: the disease, the biology, the treatment. CA Cancer J Clin. 2010;60:351–75.
Ueno NT, Buzdar AU, Singletary SE, Ames FC, McNeese MD, Holmes FA, et al. Combined-modality treatment of inflammatory breast carcinoma: twenty years of experience at M.D. Anderson Cancer Center. Cancer Chemother Pharmacol. 1997;40:321–9.
Fleming RY, Asmar L, Buzdar AU, McNeese MD, Ames FC, Ross MI, et al. Effectiveness of mastectomy by response to induction chemotherapy for control in inflammatory breast carcinoma. Ann Surg Oncol. 1997;4:452–61.
Mankoff DA, Dunnwald LK, Gralow JR, Ellis GK, Schubert EK, Tseng J, et al. Changes in blood flow and metabolism in locally advanced breast cancer treated with neoadjuvant chemotherapy. J Nucl Med. 2003;44:1806–14.
Schelling M, Avril N, Nahrig J, Kuhn W, Römer W, Sattler D, et al. Positron emission tomography using [18F]fluorodeoxyglucose for monitoring primary chemotherapy in breast cancer. J Clin Oncol. 2000;18:1689–95.
Bassa P, Kim EE, Inoue T, Wong FC, Korkmaz M, Yang DJ, et al. Evaluation of preoperative chemotherapy using PET with fluorine-18-fluorodeoxyglucose in breast cancer. J Nucl Med. 1996;37:931–8.
Martoni AA, Di Fabio F, Pinto C, Castellucci P, Pini S, Ceccarelli C, et al. Prospective study on the FDG-PET/CT predictive and prognostic values in patients treated with neoadjuvant chemoradiation therapy and radical surgery for locally advanced rectal cancer. Ann Oncol. 2011;22:650–6.
MacManus MP, Hicks RJ, Matthews JP, McKenzie A, Rischin D, Salminen EK, et al. Positron emission tomography is superior to computed tomography scanning for response-assessment after radical radiotherapy or chemoradiotherapy in patients with non-small-cell lung cancer. J Clin Oncol. 2003;21:1285–92.
Martoni AA, Zamagni C, Quercia S, Rosati M, Cacciari N, Bernardi A, et al. Early (18)F-2-fluoro-2-deoxy-d-glucose positron emission tomography may identify a subset of patients with estrogen receptor-positive breast cancer who will not respond optimally to preoperative chemotherapy. Cancer. 2010;116:805–13.
Costelloe CM, Macapinlac HA, Madewell JE, Fitzgerald NE, Mawlawi OR, Rohren EM, et al. 18F-FDG PET/CT as an indicator of progression-free and overall survival in osteosarcoma. J Nucl Med. 2009;50:340–7.
Johnson MS, Gonzales MN, Bizila S. Responsible conduct of radiology research. Part V. The Health Insurance Portability and Accountability Act and research. Radiology. 2005;237:757–64.
Dawood S, Ueno NT, Valero V, Woodward WA, Buchholz TA, Hortobagyi GN, et al. Identifying factors that impact survival among women with inflammatory breast cancer. Ann Oncol. 2012;23:870–5.
Kuerer HM, Newman LA, Smith TL, Ames FC, Hunt KK, Dhingra K, et al. Clinical course of breast cancer patients with complete pathological primary tumour and axillary lymph node response to doxorubicin-based neoadjuvant chemotherapy. J Clin Oncol. 1999;17:460–9.
Verril MW, Ashley SE, Walsh GA, Ellis P, Sacks N, Gui G, et al. Pathological complete response (pCR) in patients treated with neoadjuvant chemotherapy for operable breast cancer. Breast Cancer Res Treat. 1998;50:328.
Harris EE, Schultz D, Bertsch H, Fox K, Glick J, Solin LJ. Ten year outcome after combined modality therapy for inflammatory breast cancer. Int J Radiat Oncol Biol Phys. 2003;55:1200–8.
Buchholz TA, Katz A, Strom EA, McNeese MD, Perkins GH, Hortobagyi GN, et al. Pathologic tumor size and lymph node status predict for different rates of locoregional recurrence after mastectomy for breast cancer patients treated with neoadjuvant versus adjuvant chemotherapy. Int J Radiat Oncol Biol Phys. 2002;53:880–8.
Honkoop AH, van Diest PJ, de Jong JS, Linn SC, Giaccone G, Hoekman K, et al. Prognostic role of clinical, pathological and biological characteristics in patients with locally advanced breast cancer. Br J Cancer. 1998;77:621–6.
Chen JH, Mehta RS, Nalcioglu O, Su MY. Inflammatory breast cancer after neoadjuvant chemotherapy: can magnetic resonance imaging precisely diagnose the final pathological response? Ann Surg Oncol. 2008;15:3609–13.
Dunnwald LK, Gralow JR, Ellis GK, Livingston RB, Linden HM, Specht JM, et al. Tumor metabolism and blood flow changes by positron emission tomography: relation to survival in patients treated with neoadjuvant chemotherapy for locally advanced breast cancer. J Clin Oncol. 2008;26:4449–57.
Cachin F, Prince HM, Hogg A, Ware RE, Hicks RJ. Powerful prognostic stratification by [18F]fluorodeoxyglucose positron emission tomography in patients with metastatic breast cancer treated with high dose chemotherapy. J Clin Oncol. 2006;24:3026–31.
Couturier O, Jerusalem G, N’Guyen JM, Hustinx R. Sequential positron emission tomography using [18F]fluorodeoxyglucose for monitoring response to chemotherapy in metastatic breast cancer. Clin Cancer Res. 2006;12:6437–43.
Emmering J, Krak NC, Van der Hoeven JJ, Spreeuwenberg MD, Twisk JW, Meijer S, et al. Preoperative [18F]FDG-PET after chemotherapy in locally advanced breast cancer: prognostic value as compared with histopathology. Ann Oncol. 2008;19:1573–7.
Alberini JL, Lerebours F, Wartski M, Fourme E, Le Stanc E, Gontier E, et al. 18F-fluorodeoxyglucose positron emission tomography/computed tomography (FDG-PET/CT) imaging in the staging and prognosis of inflammatory breast cancer. Cancer. 2009;115:5038–47.
Dawood S, Merajver SD, Viens P, Vermeulen PB, Swain SM, Buchholz TA, et al. International expert panel on inflammatory breast cancer: consensus statement for standardized diagnosis and treatment. Ann Oncol. 2011;22:515–23.
Kurosumi M. Significance of histopathological evaluation in primary therapy for breast cancer – recent trends in primary modality with pathological complete response (pCR) as end point. Breast Cancer. 2004;11:139–47.
Pons F, Duch J, Fuster D. Breast cancer therapy: the role of PET-CT in decision making. Q J Nucl Med Mol Imaging. 2009;53:210–23.
Lee Jean H, Rosen EL, Mankoff DA. The role of radiotracer imaging in the diagnosis and management of patients with breast cancer: part 1 – overview, detection, and staging. J Nucl Med. 2009;50:569–81.
Burcombe RJ, Makris A, Pittam M, Lowe J, Emmott J, Wong WL. Evaluation of good clinical response to neoadjuvant chemotherapy in primary breast cancer using [18F]-fluorodeoxyglucose positron emission tomography. Eur J Cancer. 2002;38:375–9.
Boellaard R. Standards for PET image acquisition and quantitative data analysis. J Nucl Med. 2009;50 Suppl 1:11S–20.
Young H, Baum R, Cremerius U, Herholz K, Hoekstra O, Lammertsma AA, et al. Measurement of clinical and subclinical tumour response using [18F]-fluorodeoxyglucose and positron emission tomography: review and 1999 EORTC recommendations. European Organization for Research and Treatment of Cancer (EORTC) PET Study Group. Eur J Cancer. 1999;35:1773–82.
Wahl RL, Jacene H, Kasamon Y, Lodge MA. From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med. 2009;45:228–47.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carkaci, S., Sherman, C.T., Ozkan, E. et al. 18F-FDG PET/CT predicts survival in patients with inflammatory breast cancer undergoing neoadjuvant chemotherapy. Eur J Nucl Med Mol Imaging 40, 1809–1816 (2013). https://doi.org/10.1007/s00259-013-2506-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-013-2506-8