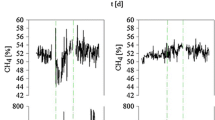
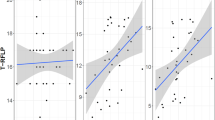
Abstract
This study provides a comprehensive, long-term microbiological study of a continuously operated, mesophilic, agricultural biogas plant fed with whole-crop silages of maize and rye, cattle manure and cattle slurry. The microbial community structure was accessed by high-throughput 16S rRNA gene amplicon sequencing. For the characterisation of the microbial dynamics, the community profiling method terminal restriction fragment length polymorphism (TRFLP) in combination with a cloning-sequencing approach as well as a LC-MS/MS approach for protein identification were applied. Our results revealed that the anaerobic digestion is a highly sensitive process: small variations in the process performance induce fluctuations in the microbial community composition and activity. In this context, it could be proven that certain microbial species were better adapted to changing process condition such as temperature (interspecies competition) and that there is a physiological compensation between different microorganisms so that the reactor efficiency was not adversely affected despite of structural and functional changes within the microbial community.





Similar content being viewed by others
References
Allison SD, Martiny JBH (2008) Resistance, resilience, and redundancy in microbial communities. PNAS 105:11512–11519
Angly FE, Dennis PG, Skarshewski A, Vanwonterghem I, Hugenholtz P, Tyson GW (2014) CopyRighter: a rapid tool for improving the accuracy of microbial community profiles through lineage-specific gene copy number correction. Microbiome 2:11–23
Appels L, Lauwers J, Degrève J, Helsen L, Lievens B, Willems K, Van Impe J, Dewil R (2011) Anaerobic digestion in global bio-energy production: potential and research challenges. Renew Sust Energ Rev 15:4295–4301
Benndorf D, Reichl U (2014) Proteomics in environmental and technical microbiology. Eng Life Sci 14:27–46
Blume F, Bergmann I, Nettmann E, Schelle H, Rehde G, Mundt K, Klocke M (2010) Methanogenic population dynamics during semi-continuous biogas fermentation and acidification by overloading. J Appl Microbiol 109:441–450
Caporaso JG, Lauber CL, Walters WA, Berg-Lyons D, Huntley J, Fierer N, Owens SM, Betley J, Fraser L, Bauer M, Gormley N, Gilbert JA, Smith G, Knight R (2012) Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J 6:1621–1624
Carballa M, Smit M, Etchebehere C, Boon N, Verstrate W (2011) Correlation between molecular and operational parameters in continuous lab-scale anaerobic digesters. Appl Microbiol Biotechnol 89:303–314
Chen Y, Cheng JJ, Creamer KS (2008) Inhibition of anaerobic digestion process: a review. Bioresour Technol 99:4044–4064
De Vrieze J, Hennebel T, Boon N, Verstraete W (2012) Methanosarcina: the rediscovered methanogen for heavy duty biomethanation. Bioresour Technol 112:1–9
Donoso-Bravo A, Retamal C, Carballa M, Ruiz-Filippi G, Chamy R (2009) Influence of temperature on the hydrolysis, acidogenesis and methanogenesis in mesophilic anaerobic digestion: parameter identification and modeling application. Water Sci Technol 60:9–17
Drosg B (2013) Process monitoring in biogas plants. IEA Bioenergy Task 37 – Energy from Biogas
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol Monogr 67:345–366
Edgar RC (2010) Search and clustering of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998
Fotidis IA, Karakashev D, Angelidaki I (2013) The dominant acetate degradation pathway/methanogenic composition in full-scale anaerobic digesters operating under different ammonia levels. Int J Environ Sci Technol. doi:10.1007/s13762-013-0407-9
Franke-Whittle IH, Goberna M, Pfister V, Insam H (2009) Design and development of the ANAEROCHIP microarray for investigation of methanogenic communities in biogas reactors. J Microbiol Methods 79:279–288
Galagan JE, Nusbaum C, Roy A, Endrizzi MG, Macdonald P, FitzHugh W, Calvo S, Engels R, Smirnov S, Atnoor D, Brown A, Allen N, Naylor J, Stange-Thomann N, DeArellano K, Johnson R, Linton L, McEwan P, McKernan K, Talamas J, Tirrell A, Ye W, Zimmer A, Barber RD, Cann I, Graham DE, Grahame DA, Guss AM, Hedderich R, Ingram-Smith C, Kuettner HG, Krzycki JA, Leigh JA, Li W, Liu J, Mukhopadhyay B, Reeve JN, Smith K, Springer TA, Umayam LA, White O, White RH, Conway de Macario E, Ferry JG, Jarrell KF, Jing H, Macario AJL, Paulsen I, Pritchett M, Sowers KR, Swanson RV, Zinder SH, Lander E, Metcalf WW, Birren B (2002) The genome of Methanosarcina acetivorans reveals extensive metabolic and physiological diversity. Genome Res 12:532–542
Goberna M, Gademaier M, Garcia C, Wett B, Insam H (2010) Adaptation of methanogenic communities to the cofermentation of cattle excreta and olive mill wastes at 37 °C and 55 °C. Appl Environ Microbiol 76:6564–6571
Hanreich A, Schimpf U, Zakrzewski M, Schlüter A, Benndorf D, Heyer R, Rapp E, Pühler A, Reichl U, Klocke M (2013) Metagenome and metaproteome analyses of mesophilic biogas-producing anaerobic batch fermenters indicate concerted plant carbohydrate degradation. Syst Appl Microbiol 36:330–338
Hensens K, Martens L, Vandekerckhove J, Gevaert K (2007) MascotDatfile: An open-source library to fully parse and analyse MASCOT MS/MS search results. Proteomics 7:364–366
Heyer R, Kohrs F, Benndorf D, Rapp E, Kausmann R, Heiermann M, Klocke M, Reichl U (2013) Metaproteome analysis of the microbial communities in agricultural biogas plants. New Biotechnol 30:614–622
Heyer R, Kohrs F, Reichl U, Benndorf D (2015) Metaproteomics of complex microbial communities in biogas plants. Microb Biotechnol. doi:10.1111/1751-7915-12276
Jaenicke S, Ander C, Bekel T, Bisdorf R, Dröge M, Gartemann KH, Jünemann S, Kaiser O, Krause L, Tille F, Zakrzewski M, Pühler A, Schlüter A, Goesmann A (2011) Comparative and joint analysis of two metagenomic datasets from a biogas fermenter obtained by 454-pyrosequencing. PLoS One 6(1), e14519
Kim M, Morrison M, Yu Z (2011) Evaluation of different partial 16S rRNA gene sequence regions for phylogenetic analysis of microbiomes. J Microbiol Methods 84:81–87
Klang J, Theuerl S, Szewzyk U, Huth M, Tölle R, Klocke M (2015) Dynamic variation of the microbial community structure during the long-time mono-fermentation of maize and sugar beet silage. Microb Biotechnol. doi:10.1111/1751-7915.12263
Klindworth A, Pruesse E, Schweer T, Peplies J, Quast C, Horn M, Glöckner FO (2013) Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res 41: doi:10.1093/nar/gks808
Kohrs F, Heyer R, Magnussen A, Benndorf D, Muth T, Behne A, Rapp E, Kausmann R, Heiermann M, Klocke M, Reichl U (2014) Sample prefractionation with liquid isoelectric focusing enables in depth microbial metaproteome analysis of mesophilic and thermophilic biogas plants. Anaerobe 29:59–67
Krause L, Diaz NN, Edwards RA, Gartemann KH, Krömeke H, Neuweger H, Pühler A, Runte KJ, Schlüter A, Stoye J, Szczepanowski R, Tauch A, Goesmann A (2008) Taxonomic composition and gene content of a methane-producing microbial community isolated from a biogas reactor. J Biotechnol 136(1–2):91–101
Krieg NR, Ludwig W, Whitman WB, Hedlund BP, Paster BJ, Staley JT, Ward N, Brown D, Parte A (2010) Bergey’s manual of systematic bacteriology, volume 4: The Bacteroidetes, Spirochaetes, Tenericutes (Mollicutes), Acidobacteria, Fibrobacteres, Fusobacteria, Dictyoglomi, Gemmatimonadetes, Lentisphaerae, Verrucomicrobia, Chlamydiae, and Planctomycetes. Springer, pp. 25–102
Lane DJ, Pace B, Olsen GJ, Stahl DA, Sogin ML, Pace NR (1985) Rapid determination of 16S ribosomal RNA sequences for phylogenetic analyses. Proc Natl Acad Sci U S A 82:6955–6959
Lebuhn M, Munk B, Effenberger M (2014) Agricultural biogas production in Germany—from practice to microbiology basics. Energy Sustain Soc 4:1–21
Lindorfer H, Waltenberger R, Köllner K, Braun R, Kirchmayr R (2008) New data on temperature optimum and temperature changes in energy crop digesters. Bioresour Technol 99:7011–7019
Liu Y, Whitman WB (2008) Metabolic, phylogenetic, and ecological diversity of the methanogenic Archaea. Ann N Y Acad Sci 1125:171–189
Liu FH, Wang SB, Zhang JS, Zhang J, Yan X, Zhou HK, Zhao GP, Zhou ZH (2009) The structure of the bacterial and archaeal community in a biogas digester as revealed by denaturing gradient gel electrophoresis and 16S rDNA sequencing analysis. J Appl Microbiol 106:952–966
Maeder DL, Anderson I, Brettin TS, Bruce DC, Gilna P, Han CS, Lapidus A, Metcalf WW, Saunders E, Tapia R, Sowers KR (2006) The Methanosarcina barkeri genome: comparative analysis with Methanosarcina acetivorans and Methanosarcina mazei reveals extensive rearrangement within methanosarcinal genomes. J Bacteriol 188:7922–7931
Mähnert P, Linke B (2009) Kinetic study of biogas production from energy crops and animal waste slurry: effect of organic loading rate and reactor size. Environ Technol 30:93–99
Marzorati M, Wittebolle L, Boon N, Daffonchio D, Verstraete W (2008) How to get more out of molecular fingerprints: practical tools for microbial ecology. Environ Microbiol 10:1571–1581
McCune B, Mefford M J (2011) PC-ORD. Multivariate analysis of ecological data. Version 6.08 MjM Software, Gleneden Beach, Oregon, U.S.A.
Muth T, Behne A, Heyer R, Kohrs F, Benndorf D, Hoffmann M, Lehtevä M, Reuchl U, Martens L, Rapp E (2015) The MetaProteomeAnalyzer: a powerful open-source software suite for metaproteomic data analysis and interpretation. J Proteome Res 14:1557–1565
Nelson MC, Morrison M, Zhongtang Y (2011) A meta-analysis of the microbial diversity observed in anaerobic digesters. Bioresour Technol 102:3730–3739
Nettmann E, Bergmann I, Pramschüfer S, Mundt K, Plogsties V, Herrmann C, Klocke M (2010) Polyphasic analyses of methanogenic archaeal communities in agricultural biogas plants. Appl Environ Microbiol 76: 2540–2548 + Erratum Appl Environ Microbiol 77: 394.
Ondov BD, Bergman NH, Phillippy AM (2011) Interactive metagenomic visualization in a web browser. BMC Bioinforma 12:385–393
Pycke BFG, Etchebehere C, van de Caveye P, Negroni A, Verstraete W, Boon N (2011) A time-course analysis of four full-scale anaerobic digesters in relation to the dynamics of change of their microbial communities. Water Sci Technol 63:769–775
Rademacher A, Nolte C, Schönberg M, Klocke M (2012) Temperature increases from 55 to 75 °C in a two-phase biogas reactor result in fundamental alterations within the bacterial and archaeal community structure. Appl Microbiol Biotechnol 96:565–576
Read S, Marzorati M, Guimarães BCM, Boon N (2011) Microbial resource management revisited: successful parameters and new concepts. Appl Microbiol Biotechnol 90:861–871
Regueiro L, Veiga P, Figueroa M, Alonso-Gutierrez J, Stams AJM, Lema JM, Carballa M (2012) Relationship between microbial activity and microbial community structure in six full-scale anaerobic digesters. Microbiol Res 167:581–589
Schink B, Stams AJM (2013) Syntrophism among prokaryotes. In: Rosenberg E, DeLong EF, Lory S, Stakebrandt E, Thompson F (eds) The prokaryotes, 4th edn. Springer, Berlin, Heidelberg, pp 471–493
Schlüter A, Bekel T, Diaz NN, Dondrup M, Eichenlaub R, Gartemann KH, Krahn I, Krause L, Krömeke H, Kruse O, Mussgnug JH, Neuweger H, Niehaus K, Pühler A, Runte KJ, Szczepanowski R, Tauch A, Tilker A, Viehöver P, Goesmann A (2008) The metagenome of a biogas-producing microbial community of a production-scale biogas plant fermenter analysed by the 454-pyrosequencing technology. J Biotechnol 136(1–2):77–90
Schmidt A, Müller N, Schink B, Schleheck D (2013) A proteomic view at the biochemistry of syntrophic butyrate oxidation in Syntrophomonas wolfei. PLoS ONE 8, e56905. doi:10.1371/journal.pone.0056905
Sundberg C, Al-Soud WA, Larsson M, Alm E, Yekta SS, Svensson BH, Sørensen SJ, Karlsson A (2013) 454 pyrosequencing analyses of bacterial and archaeal richness in 21 full-scale biogas digesters. FEMS Microbiol Ecol 85:612–626
Talbot G, Topp E, Palin MF, Massé DI (2008) Evaluation of molecular methods used for establishing the interactions and functions of microorganisms in anaerobic reactors. Water Res 42:513–537
Tringe SG, Hugenholtz P (2008) A renaissance for the pioneering 16S rRNA gene. Curr Opin Microbiol 11:442–446
Vanwonterghem I, Jensen PD, Ho DP, Batstone DJ, Tyson GW (2014) Linking microbial community structure, interactions and function in anaerobic digesters using new molecular techniques. Curr Opin Biotechnol 27:55–64
VDLUFA - Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten: Methodenbuch – Band III (1997) Die chemische Untersuchung von Futtermitteln (The chemical analysis of feedstuffs). VDLUFA-Verlag, Speyer
VDLUFA - Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten: Methodenbuch – Band VII (2011) Umweltanalytik (environmental analysis). VDLUFA-Verlag, Speyer
Verstraete W, Wittebolle L, Heylen K, Vanparys B, de Vos P, van de Wiele T, Boon N (2007) Microbial resource management: the road to go for environmental biotechnology. Eng Life Sci 7:117–126
Vos P, Garrity G, Jones D, Krieg NR, Ludwig W, Rainey FA, Schleifer KH, Whitman WB (2011) Bergey’s manual of systematic bacteriology volume 3: The Firmicutes. Spriner
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
Wang H, Tolvanen K, Lehtomäki A, Puhakka J, Rintala J (2010) Microbial community structure in anaerobic co-digestion of grass silage and cow manure in a laboratory continuously stirred tank reactor. Biodegradation 21:135–146
Ward AJ, Hobbs PJ, Holliman PJ, Jones DL (2008) Optimisation of the anaerobic digestion of agricultural resources. Bioresource Technol 99:7928–7940
Weiland P (2010) Biogas production: current state and perspectives. Appl Microbiol Biotechnol 85:849–860
Wittebolle L, Marzorati M, Clement L, Balloi A, Daffonchio D, Heylen K, de Vos P, Verstraete W, Boon N (2009) Initial community evenness favours functionality under selective stress. Nature 458:623–626
Zakrzewski M, Goesmann A, Jaenicke S, Jünemann S, Eikmeyer F, Szczepanowski R, Al-Soud WA, Sørensen S, Pühler A, Schlüter A (2012) Profiling of the metabolically active community from a production-scale biogas plant by means of high-throughput metatranscriptome sequencing. J Biotechnol 158:248–58
Acknowledgments
S. Theuerl, F. Kohrs, I. Maus and R. Kausmann were supported by the German Federal Ministry of Food, Agriculture and Consumer Protection (BMELV), grant numbers 22010711, 22028711 and 22028811 (joint research project Biogas-Biocoenosis) and 22027707 (joint research project BiogasEnzyme).
The authors would like to thank Kerstin Mundt for her excellent technical support in the laboratory.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 163 kb)
Rights and permissions
About this article
Cite this article
Theuerl, S., Kohrs, F., Benndorf, D. et al. Community shifts in a well-operating agricultural biogas plant: how process variations are handled by the microbiome. Appl Microbiol Biotechnol 99, 7791–7803 (2015). https://doi.org/10.1007/s00253-015-6627-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6627-9