Abstract
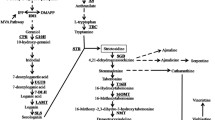
Fungal interspecific interactions enhance biosynthesis of phenylpropanoid metabolites (PM), and production of nitric oxide (NO) is known to be involved in this process. However, it remains unknown which signaling pathway(s) or regulator(s) mediate fungal PM biosynthesis. In this study, we cocultured two white-rot fungi, Inonotus obliquus and Phellinus morii, to examine NO production, expression of the genes involved in phenylpropanoid metabolism and accumulation of phenylpropanoid-derived polyphenols by I. obliquus. Coculture of the two fungi caused an enhanced NO biosynthesis followed by increased transcription of the genes encoding phenylalanine ammonia lyase (PAL) and 4-coumarate CoA ligase (4CL), as well as an upregulated biosynthesis of styrylpyrone polyphenols in I. obliquus. Addition of the NO synthase (NOS) selective inhibitor aminoguanidine (AG) inhibited NO production by more than 90 % followed by cease in transcription of PAL and 4Cl. Treatment of guanylyl cyclase inhibitor 1H-[1,2,4]oxadiazolo[4,3-a]quinoxalin-1-one did not affect NO production but suppressed transcription of PAL and 4CL and reduced accumulation of total phenolic constituents. Genome-wide analysis of I. obliquus revealed two genes encoding a constitutive and an inducible NOS-like protein, respectively (cNOSL and iNOSL). Coculture of the two fungi did not increase the expression of the cNOSL gene but triggered expression of the iNOSL gene. Cloned iNOSL from Escherichia coli shows higher activity in transferring l-arginine to NO, and this activity is lost upon AG addition. Thus, iNOSL is more responsible for NO production in I. obliquus and may act as an important regulator governing PM production during fungal interspecific interactions.





Similar content being viewed by others
References
Afri Y, Levasseur A, Record E (2013) Differential gene expression in Pycnoporus coccineus during interspecific mycelial interactions with different competitors. Appl Environ Microbiol 79:6626–6636
Arasimowicz-Jelonek M, Floryszak-Wieczork J (2014) Nitric oxide: an effective weapon of the plant or the pathogen? Mol Plant Pathol 15:406–416
Bryan NS, Bian K, Murad F (2009) Discovery of the nitric oxide signaling pathway and targets for drug development. Front Biosci 14:1–18
Cechova S, Pajewski TN (2004) The soluble guanylyl cyclase inhibitor ODQ, 1H-[1,2,4] oxadiazolo[4,3-a]quinoxalin-1-one, dose-dependently reduces the threshold for isoflurane anesthesia in rats. Anesth Analg 99:752–757
Cochrane FC, Davin LB, Lewis NG (2004) The Arabidopsis phenylalanine ammonia lyase gene family kinetic characterization of the four isoforms. Phytochem 65:1557–1564
de Pinto MC, Tommasi F, De Gara L (2002) Changes in the antioxidant systems as part of the signaling pathway responsible for the programmed cell death activated by nitric oxide and reactive oxygen species in tobacco bright-yellow 2 cells. Plant Physiol 130:698–708
Dempsey DA, Vlot AC, Wildermuth MC, Klessiq DF (2011) Salicylic acid biosynthesis and metabolism. Arabidopsis Book e0156. American Society of Plant Biologists, Washington. doi:10.1199/tab.0156
Feechan A, Kwon E, Yun BW, Wang Y, Pallas JA, Loake G (2005) A central role for S-nitrosothiols in plant disease resistance. PNAS 102:8054–8059
Fӧrstermann U, Mȕnzel T (2006) Endothelial nitric oxide synthase in vascular disease: from marvel to menace. Circulation 113:1708–1714
Frӧhlich A, Durner J (2011) The hunt for plant nitric oxide synthase (NOS): is one really needed? Plant Sci 181:401–404
Gas E, Flores-Pérez Ú, Sauret-Güeto S, Rodríguez-Concepción M (2009) Hunting for plant nitric oxide synthase provides new evidence of central role for plastids in nitric oxide metabolism. Plant Cell 21:18–23
Gong B, Miao L, Kong W, Bai JG, Wang X, Wei M, Shi Q (2014) Nitric oxide, as a downstream signal, plays vital role in auxin induced cucumber tolerance to sodic alkaline stress. Plant Physiol Biochem 83:258–266
Gorren ACF, Mayer B (2007) Nitric oxide synthase: a cytochrome P450 family foster child. Biochim Biophys Acta 1770:432–445
Guo YJ, Deng GF, Xu XR, Wu S, Li S, Xia EQ, Li F, Chen F, Ling WH, Li HB (2012) Antioxidant capacities, phenolic compounds and polysaccharide contents of 49 edible macro-fungi. Food Funct 3:1195–1205
Gupta KJ, Fernie AR, Kaise WM, van Dongen JT (2011) On the origins of nitric oxide. Trend Plant Sci 16:160–168
Hamberger B, Hahlbrock K (2003) The 4-coumarate:CoA ligase gene family in Arabidopsis thaliana comprises one rare, sinapate-activating and three commonly occurring isoenzymes. PNAS 101:2209–2214
Hiscox J, Baldrian P, Rogers HJ, Boddy L (2010) Changes in oxidative enzyme activity during interspecific mycelial interactions involving the white-rot fungus Trametes versicolor. Fungal Genet Biol 47:562–571
Huang WY, Cai YZ, Xing J, Corke H, Sun M (2007) A potential antioxidant resource: endophytic fungi from medicinal plants. Econ Bot 61:14–30
Huang HY, Chieh SY, Tso TK, Chien Y, Lin HT, Tsai YC (2011) Orally administered mycelial culture of Phellinus linteus exhibits antitumor effects in hepatoma cell-bearing mice. J Ethnopharmacol 133:460–466
Hyun MW, Yun YH, Kim JY, Kim SH (2011) Fungal and plant phenylalanine ammonia-lyase. Microbiol 39:257–265
Korkina LG (2007) Phenylpropanoids as naturally occurring antioxidants: from plant defense to human health. Cell Mol Biol 53:15–27
Lamattina L, Garci-Mata C, Graziano M, Pagnussat G (2003) Nitric oxide: the versatility of an intensive signal molecule. Plant Biol 54:109–136
Lattanzio V, Lattanzio VMT, Cardinali A (2006) Role of phenolics in the resistance mechanisms of plants against fungal pathogens and insects. Phytochem Adv Res: 23–67
Lee IK, Yun BS (2006) Hispidin analogs from the mushroom Inonotus xeranticus and their free radical scavenging activity. Bioorg Med Chem Lett 16:2376–2379
Lee IK, Yun BS (2011) Styrylpyrone-class compounds from medicinal fungi Phellinus and Inonotus spp., and their medicinal importance. J Antibiot 64:349–359
Lee D, Meyer K, Chappie C, Douglas CJ (1997) Antisense suppression of 4-coumarate: coenzyme A ligase activity in Arabidopsis leads to altered lignin subunit composition. Plant Cell 9:985–1998
Lewin A, Mayer M, Chusainow J, Jacob D, Appel B (2005) Viral promoters can initiate expression of toxin genes introduced into Eschericha coli. BMC Biotechnol 5:19–28
Nallamsetty S, Waugh DS (2007) A generic protocol for the expression and purification of recombinant proteins in Escherichi coli using a combinatorial His6-maltose binding protein fusion tag. Nat Protocol 2:383–391
Ninnemann H, Maier J (1996) Indications for the occurrence of nitric oxide synthases in fungi and plants and the involvement in photoconidiation. Photochem Photobiol 64:393–398
Park HS, Huh SH, Kim MS, Lee SH, Choi EJ (2000) Nitric oxide negatively regulates cJun N-terminal kinase/stress-activated protein kinase by means of S-nitrosylation. PNAS 97:14382–14387
Park IH, Chung SK, Lee KB, Yoo YC, Kim SK, Kim GS, Song KS (2004) An antioxidant hispidin from the mycelial cultures of Phellinus linteus. Arch Pharm Res 27:615–618
Park HS, Huh SH, Kim MS, Kim DY, Gwaq BJ, Cho SG, Choi EJ (2006) Neuronal nitric oxide synthase (nNOS) modulates the JNK1 activity through redox mechanism: a cGMP independent pathway. Biochem Biophys Res Commun 346:408–414
Prado FE, Rosa M, Prado C, Podazza G, Interdonato R, Gonzalez JA, Hilal M (2012) UV-B radiation, its effects and defense mechanisms in terrestrial plants. In: Ahmad P, Prasad MNC (eds) Environmental adaptations and stress tolerance of plants in the era of climate changes. Springer, Berlin, pp 57–83
Pryor KD, Leithing B (1997) High-level expression of soluble protein in Escherichia coli using a His6-Tag and maltose-binding-protein double affinity fusion system. Protein Expr Purif 10:309–319
Romero-Puertsas MC, Perazzolli M, Zago ED, Delledonne M (2004) Nitric oxide signalling functions in plant-pathogen interactions. Cell Microbiol 6:795–803
Samalova M, Johnson J, IIIe M, Kelly S, Fricker M, Gurr S (2013) Nitric oxide generated by the rice blast fungus Magnaporthe oryzae drives plant infection. New Phytol 197:207–222
Turrion-Gomez JL, Benito EP (2011) Flux of nitric oxide between the necrotrophic pathogen Botrytis cinerea and the host plant. Mol Plant Pathol 12:606–616
Vannozzi A, Dry LB, Fasoli M, Zenoni S, Lucchin M (2012) Genome-wide analysis of the grapevine stilbene synthase multigenic family: genomic organization and expression profiles upon biotic and abiotic stresses. BMC Plant Biol 12:130–151
Velázquez-Cedeño M, Farnet AM, Billette C, Mata G, Savoie JM (2007) Interspecific interactions with Trichoderma longibrachiatum induce Pleurotus ostreatus defence reactions based on the production of laccase isozymes. Biotechnol Lett 29:1583–1590
Yoshioka H, Asai S, Yoshioka M, Kobayashi M (2009) Molecular mechanisms of generation for nitric oxide and reactive oxygen species, and role of the radical burst in plant immunity. Mol Cells 28:312–329
Yu M, Lamattina L, Spoel SH, Loake GJ (2014) Nitric oxide function in plant biology: a redox cue in deconvolution. New Phytol 202:1142–1156
Yun BW, Feechan A, Yin M, Saidi NB, Le Bihan T, Yu M, Moore JW, Kang JG, Kown E, Spoel SH, Pallas JA, Loake GJ (2011) S-nitrosylation of NADPH oxidase regulates cell death in plant immunity. Nature 478:264–268
Zheng W, Miao K, Zhao Y, Pan S, Zhang M, Jiang H (2009a) Nitric oxide mediates the fungal-elicitor enhanced biosynthesis of antioxidant polyphenols in submerged cultures of Inonotus obliquus. Microbiol 155:3440–3448
Zheng W, Zhang M, Zhao Y, Wang Y, Miao K, Wei Z (2009b) Accumulation of antioxidant phenolic constituents in submerged cultures of Inonotus obliquus. Bioresour Technol 100:1327–1335
Zheng W, Miao K, Liu Y, Zhao Y, Zhang M, Pan S, Dai Y (2010) Chemical diversity of biologically active metabolites in the sclerotia of Inonotus obliquus and submerged culture strategies for up-regulating their production. Appl Microbiol Biotechnol 87:1237–1254
Zheng W, Zhao Y, Zheng X, Liu Y, Pan S, Dai Y, Liu F (2011) Production of antioxidant and antitumor metabolites by submerged cultures of Inonotus obliquus cocultured with Phellinus punctatus. Appl Microbiol Biotechnol 89:157–167
Acknowledgments
This project is financially supported by grants from the Natural Science Foundation of China (31170063, 31470173) (for W. Zheng) and a grant from the key project of the Natural Science Foundation of Education Department of Jiangsu province (11KJA180002) (for W. Zheng), and a grant from the Key Laboratory for Biotechnology on Medicinal Plants of Jiangsu Province (KLBMP1301) (for Y. Zhao). The authors are very grateful to Dr. Yiqin Wang from Institute of Genetics and Developmental Biology, Chinese Academy of Sciences, for her generous help in the process of gene cloning and expression. Thanks should also be given to Dr. Wenbing Yin from the Institute of Microbiology, Chinese Academy of Sciences, for his precious advice during the process of I. obliquus genome annotation.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 1559 kb)
Rights and permissions
About this article
Cite this article
Zhao, Y., Xi, Q., Xu, Q. et al. Correlation of nitric oxide produced by an inducible nitric oxide synthase-like protein with enhanced expression of the phenylpropanoid pathway in Inonotus obliquus cocultured with Phellinus morii . Appl Microbiol Biotechnol 99, 4361–4372 (2015). https://doi.org/10.1007/s00253-014-6367-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-6367-2