Abstract
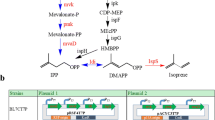
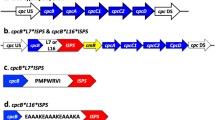
We constructed a biosynthetic pathway of isoprene production in Escherichia coli by introducing isoprene synthase (ispS) from Populus alba. 1-deoxy-d-xylulose 5-phosphate synthase (dxs), 1-deoxy-d-xylulose 5-phosphate reductoisomerase (dxr) and isopentenyl diphosphate (IPP) isomerase (idi) were overexpressed to enhance the isoprene production. The isoprene production was improved 0.65, 0.16, and 1.22 fold over the recombinant BL21 (pET-30a-ispS), respectively, and idi was found to be a key regulating point for isoprene production. In order to optimize the production of isoprene in E. coli, we attempted to construct polycistronic operons based on pET-30a with genes dxs, dxr, and idi in various orders. The highest isoprene production yield of 2.727 mg g−1 h−1 (per dry weight) was achieved by E. coli transformed with pET-30a-dxs/dxr/idi. Interestingly, the gene order was found to be consistent with that of the metabolic pathway. This indicates that order of genes is a significant concern in metabolic engineering and a sequential expression pattern can be optimized according to the biosynthetic pathway for efficient product synthesis.








Similar content being viewed by others
References
Ajikumar PK, Xiao WH, Tyo KEJ, Wang Y, Simeon F, Leonard E, Mucha O, Phon TH, Pfeifer B, Stephanopoulos G (2010) Isoprenoid pathway optimization for Taxol precursor overproduction in Escherichia coli. Science 330:70
Albrecht M, Misawa N, Sandmann G (1999) Metabolic engineering of the terpenoid biosynthetic pathway of Escherichia coli for production of the carotenoids β-carotene and zeaxanthin. Biotechnol Lett 21:791–795
Emerman M, Temin HM (1984) Genes with promoters in retrovirus vectors can be independently suppressed by an epigenetic mechanism. Cell 39:459–467
Ghattas IR, Sanes J, Majors J (1991) The encephalomyocarditis virus internal ribosome entry site allows efficient coexpression of two genes from a recombinant provirus in cultured cells and in embryos. Mol Cell Biol 11:5848–5859
Gräwert T, Kaiser J, Zepeck F, Laupitz R, Hecht S, Amslinger S, Schramek N, Schleicher E, Weber S, Haslbeck M (2004) IspH protein of Escherichia coli: studies on iron–sulfur cluster implementation and catalysis. J Am Chem Soc 126(40):12847–12855
Johnston K, Clements A, Venkataramani RN, Trievel RC, Marmorstein R (2000) Coexpression of proteins in bacteria using T7-based expression plasmids: expression of heteromeric cell-cycle and transcriptional regulatory complexes. Protein Expr Purif 20(3):435–443
Kajiwara S, Fraser PD, Kondo K, Misawa N (1997) Expression of an exogenous isopentenyl diphosphate isomerase gene enhances isoprenoid biosynthesis in Escherichia coli. Biochem J 324:421
Kesselmeier J, Staudt M (1999) Biogenic volatile organic compounds (VOC): an overview on emission, physiology and ecology. J Atmos Chem 33:23–88
Kim SW, Keasling J (2001) Metabolic engineering of the nonmevalonate isopentenyl diphosphate synthesis pathway in Escherichia coli enhances lycopene production. Biotechnol Bioeng 72:408–415
Kuzuyama T (2002) Mevalonate and nonmevalonate pathways for the biosynthesis of isoprene units. Biosci Biotechnol Biochem 66:1619–1627
Lehning A, Zimmer I, Steinbrecher R, Bruggemann N, Schnitzler JP (1999) Isoprene synthase activity and its relation to isoprene emission in Quercus robur L. leaves. Plant Cell Environ 22:495–504
Lichtenthaler HK (2000) Non-mevalonate isoprenoid biosynthesis: enzymes, genes and inhibitors. Biochem Soc Trans 28:785–789
Liu M, Yu H (2012) Cocktail production of an endo-β-xylanase and a β-glucosidase from Trichoderma reesei QM 9414 in Escherichia coli. Biochem Eng J 68:1–6
Lois LM, Rodríguez-Concepción M, Gallego F, Campos N, Boronat A (2000) Carotenoid biosynthesis during tomato fruit development: regulatory role of 1-deoxy-d-xylulose 5-phosphate synthase. Plant J 22:503–513
Matthews P, Wurtzel ET (2000) Metabolic engineering of carotenoid accumulation in Escherichia coli by modulation of the isoprenoid precursor pool with expression of deoxyxylulose phosphate synthase. Appl Microbiol Biotechnol
Miller B, Heuser T, Zimmer W (2000) Functional involvement of a deoxy–xylulose 5-phosphate reductoisomerase gene harboring locus of Synechococcus leopoliensis in isoprenoid biosynthesis. FEBS Lett 481:221–226
Miller B, Oschinski C, Zimmer W (2001) First isolation of an isoprene synthase gene from poplar and successful expression of the gene in Escherichia coli. Planta 213:483–487
Monson RK, Fall R (1989) Isoprene emission from aspen leaves—influence of environment and relation to photosynthesis and photorespiration. Plant Physiol 90:267–274
Nishizaki T, Tsuge K, Itaya M, Doi N, Yanagawa H (2007) Metabolic engineering of carotenoid biosynthesis in Escherichia coli by ordered gene assembly in Bacillus subtilis. Appl Microbiol Biotechnol 73:1355–1361
Rohdich F, Hecht S, Gärtner K, Adam P, Krieger C, Amslinger S, Arigoni D, Bacher A, Eisenreich W (2002) Studies on the nonmevalonate terpene biosynthetic pathway: metabolic role of IspH (LytB) protein. PNAS 99:1158
Sanadze J (1957) Emission of organic matters by leaves of Robinia pseudoacacia L. Soobshch Akad Nauk Gruz SSR 19:83–86
Sasaki K, Ohara K, Yazaki K (2005) Gene expression and characterization of isoprene synthase from Populus alba. FEBS Lett 579(11):2514–2518
Sharkey TD, Yeh S, Wiberley AE, Falbel TG, Gong DM, Fernandez DE (2005) Evolution of the isoprene biosynthetic pathway in Kudzu. Plant Physiol 137:700–712
Xiao Y, Zhao ZK, Liu P (2008) Mechanistic studies of IspH in the deoxyxylulose phosphate pathway: heterolytic CO bond cleavage at C4 position. J Am Chem Soc 130:2164–2165
Xue JF, Ahring BK (2011) Enhancing isoprene production by genetic modification of the 1-deoxy-d-xylulose-5-phosphate pathway in Bacillus subtilis. Appl Environ Microbiol 77:2399–2405
Yang W, Zhang L, Lu Z, Tao W, Zhai Z (2001) A new method for protein coexpression in Escherichia coli using two incompatible plasmids. Protein Expr Purif 22:472–478
Zaslaver A, Mayo AE, Rosenberg R, Bashkin P, Sberro H, Tsalyuk M, Surette MG, Alon U (2004) Just-in-time transcription program in metabolic pathways. Nat Genet 36:486–491
Zhao YR, Yang JM, Qin B, Li YH, Sun YZ, Su SZ, Xian M (2011) Biosynthesis of isoprene in Escherichia coli via methylerythritol phosphate (MEP) pathway. Appl Microbiol Biotechnol 90:1915–1922
Acknowledgments
This work was financially supported by the Natural Science Foundation of China (grant no. 21176215), Outstanding Young Scholar of Zhejiang Province (grant no. R4110092) and the Program for Zhejiang Leading Team of S&T Innovation (grant no.2011R50007). We are grateful for all editor and reviewers for their helpful advice.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lv, X., Xu, H. & Yu, H. Significantly enhanced production of isoprene by ordered coexpression of genes dxs, dxr, and idi in Escherichia coli . Appl Microbiol Biotechnol 97, 2357–2365 (2013). https://doi.org/10.1007/s00253-012-4485-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4485-2