Abstract
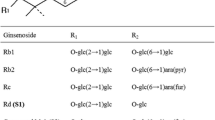
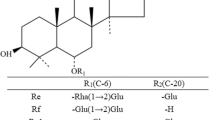
This study focused on the cloning, expression, and characterization of ginsenoside-transforming recombinant β-glucosidase from Actinosynnema mirum KACC 20028T in order to biotransform ginsenosides efficiently. The gene, termed as bglAm, encoding a β-glucosidase (BglAm) belonging to the glycoside hydrolase family 3 was cloned. bglAm consisted of 1,830 bp (609 amino acid residues) with a predicted molecular mass of 65,277 Da. This enzyme was overexpressed in Escherichia coli BL21(DE3) using a GST-fused pGEX 4T-1 vector system. The recombinant BglAm was purified with a GST·bind agarose resin and characterized. The optimum conditions of the recombinant BglAm were pH 7.0 and 37 °C. BglAm could hydrolyze the outer and inner glucose moieties at the C3 and C20 of the protopanaxadiol-type ginsenosides (i.e., Rb1 and Rd, gypenoside XVII) to produce protopanaxadiol via gypenoside LXXV, F2, and Rh2(S) with various pathways. BglAm can effectively transform the ginsenoside Rb1 to gypenoside XVII and Rd to F2; the K m values of Rb1 and Rd were 0.69 ± 0.06 and 0.45 ± 0.02 mM, respectively, and the V max values were 16.13 ± 0.29 and 51.56 ± 1.35 μmol min−1 mg−1 of protein, respectively. Furthermore, BglAm could convert the protopanaxatriol-type ginsenoside Re and Rg1 into Rg2(S) and Rh1(S) hydrolyzing the attached glucose moiety at the C6 and C20 positions, respectively. These various ginsenoside-hydrolyzing pathways of BglAm may assist in producing the minor ginsenosides from abundant major ginsenosides.





Similar content being viewed by others
References
An DS, Cui CH, Sung BH, Yang HC, Kim SC, Lee ST, Im WT, Kim SG (2010) Identification and characterization of a novel Terrabacter ginsenosidimutans sp. nov. β-glucosidase that transforms ginsenoside Rb1 into the rare gypenoside XVII and LXXV. Appl Environ Microbiol 76:5827–5836
An DS, Cui CH, Sung BH, Yang HC, Kim SC, Lee ST, Im WT, Kim SG (2012) Characterization of a novel ginsenoside-hydrolyzing alpha-l-arabinofuranosidase, AbfA, from Rhodanobacter ginsenosidimutans Gsoil 3054T. Appl Microbiol Biotechnol 94:673–682
Attele AS, Wu JA, Yuan CS (1999) Ginseng pharmacology: multiple constituents and multiple actions. Biochem Pharmacol 58:1685–1693
Bae EA, Han MJ, Kim EJ, Kim DH (2004) Transformation of ginseng saponins to ginsenoside Rh2 by acids and human intestinal bacteria and biological activities of their transformants. Arch Pharm Res 27:61–67
Cheng LQ, Na JR, Bang MH, Kim MK, Yang DC (2008) Conversion of major ginsenoside Rb1 to 20(S)-ginsenoside Rg3 by Microbacterium sp. GS514. Phytochemistry 69:218–224
Choi JR, Hong SW, Kim Y, Jang SE, Kim NJ, Han MJ, Kim DH (2011) Metabolic activities of ginseng and its constituents, ginsenoside Rb1 and Rg1, by human intestinal microflora. J Ginseng Res 35:301–307
Choi S, Kim TW, Singh SV (2009) Ginsenoside Rh2-mediated G1 phase cell cycle arrest in human breast cancer cells is caused by p15 Ink4B and p27 Kip1-dependent inhibition of cyclin-dependent kinases. Pharmaceutical Res 26:2280–2288
Christensen LP (2009) Ginsenosides chemistry, biosynthesis, analysis, and potential health effects. Adv Food Nutr Res 55:1–99
Chuankhayan P, Hua Y, Svasti J, Sakdarat S, Sullivan PA, Ketudat Cairns JR (2005) Purification of an isoflavonoid 7-O-beta-apiosyl-glucoside beta-glycosidase and its substrates from Dalbergia nigrescens Kurz. Phytochemistry 66:1880–1889
Cleland WW (1979) Statistical analysis of enzyme kinetic data. Methods Enzymol 63:103–138
Jia L, Zhao Y (2009) Current evaluation of the millennium phytomedicine—ginseng (I): etymology, pharmacognosy, phytochemistry, market and regulations. Curr Med Chem 16:2475–2484
Jin XF, Yu HS, Wang DM, Liu TQ, Liu CY, An DS, Im WT, Kim SG, Jin FX (2012) Kinetics of a cloned special ginsenosidase hydrolyzing 3-O-glucoside of multi-protopanaxadiol-type ginsenosides, named ginsenosidase type III. J Microbiol Biotechnol 22:343–351
Jin Y, Hofseth AB, Cui X, Windust AJ, Poudyal D, Chumanevich AA, Matesic LE, Singh NP, Nagarkatti M, Nagarkatti PS, Hofseth LJ (2010) American ginseng suppresses colitis through p53-mediated apoptosis of inflammatory cells. Cancer Prev Res (Phila) 3:339–347
Kaya M, Ito J, Kotaka A, Matsumura K, Bando H, Sahara H, Ogino C, Shibasaki S, Kuroda K, Ueda M, Kondo A, Hata Y (2008) Isoflavone aglycones production from isoflavone glycosides by display of β-glucosidase from Aspergillus oryzae on yeast cell surface. Appl Microbiol Biotechnol 79:51–60
Kim BH, Lee SY, Cho HJ, You SN, Kim YJ, Park YM, Lee JK, Baik MY, Park CS, Ahn SC (2006) Biotransformation of Korean Panax ginseng by Pectinex. Biol Pharm Bull 29:2472–2478
Kim M, Ahn BY, Lee JS, Chung SS, Lim S, Park SG, Jung HS, Lee HK, Park KS (2009) The ginsenoside Rg3 has a stimulatory effect on insulin signaling in L6 myotubes. Biochem Biophys Res Commun 389:70–73
Kim SJ, Lee CM, Kim MY, Yeo YS, Yoon SH, Kang HC, Koo BS (2007) Screening and characterization of an enzyme with beta-glucosidase activity from environmental DNA. J Microbiol Biotechnol 17:905–912
Kim WY, Kim JM, Han SB, Lee SK, Kim ND, Park MK, Kim CK, Park JH (2000) Steaming of ginseng at high temperature enhances biological activity. J Nat Prod 63:1702–1704
Land M, Lapidus A, Mayilraj S, Chen F, Copeland A, Del Rio TG, Nolan M, Lucas S, Tice H, Cheng JF, Chertkov O, Bruce D, Goodwin L, Pitluck S, Rohde M, Goker M, Pati A, Ivanova N, Mavromatis K, Chen A, Palaniappan K, Hauser L, Chang YJ, Jeffries CC, Brettin T, Detter JC, Han C, Chain P, Tindall BJ, Bristow J, Eisen JA, Markowitz V, Hugenholtz P, Kyrpides NC, Klenk HP (2009) Complete genome sequence of Actinosynnema mirum type strain (101). Stand Genomic Sci 1:46–53
Lee JH, Hyun YJ, Kim DH (2011) Cloning and characterization of α-l-arabinofuranosidase and bifunctional α-l-arabinopyranosidase/beta-d-galactopyranosidase from Bifidobacterium longum H-1. J Appl Microbiol 111:1097–1107
Leung KW, Wong AS (2010) Pharmacology of ginsenosides: a literature review. Chinese Medicine 5:20
Li B, Wang CZ, He TC, Yuan CS, Du W (2010) Antioxidants potentiate American ginseng-induced killing of colorectal cancer cells. Cancer Lett 289:62–70
Liu L, Zhu XM, Wang QJ, Zhang DL, Fang ZM, Wang CY, Wang Z, Sun BS, Wu H, Sung CK (2010) Enzymatic preparation of 20(S, R)-protopanaxadiol by transformation of 20(S, R)-Rg3 from black ginseng. Phytochemistry 71:1514–1520
Nah SY, Kim DH, Rhim H (2007) Ginsenosides: are any of them candidates for drugs acting on the central nervous system? CNS Drug Rev 13:381–404
Noh KH, Oh DK (2009) Production of the rare ginsenosides compound K, compound Y, and compound Mc by a thermostable beta-glycosidase from Sulfolobus acidocaldarius. Biol Pharm Bull 32:1830–1835
Noh KH, Son JW, Kim HJ, Oh DK (2009) Ginsenoside compound K production from ginseng root extract by a thermostable beta-glycosidase from Sulfolobus solfataricus. Biosci Biotechnol Biochem 73:316–321
Opassiri R, Pomthong B, Onkoksoong T, Akiyama T, Esen A, Ketudat Cairns JR (2006) Analysis of rice glycosyl hydrolase family 1 and expression of Os4bglu12 β-glucosidase. BMC Plant Biol 6:33
Park CS, Yoo MH, Noh KH, Oh DK (2010) Biotransformation of ginsenosides by hydrolyzing the sugar moieties of ginsenosides using microbial glycosidases. Appl Microbiol Biotechnol 87:9–19
Park MW, Ha J, Chung SH (2008) 20(S)-ginsenoside Rg3 enhances glucose-stimulated insulin secretion and activates AMPK. Biological Pharm Bull 31:748–751
Qi LW, Wang CZ, Yuan CS (2011) Ginsenosides from American ginseng: chemical and pharmacological diversity. Phytochemistry 72:689–699
Qu C, Bai Y, Jin X, Wnaga Y, Zhang K, You J, Zhang H (2009) Study on ginsenosides in different parts and ages of Panax quinquefolius L. Food Chem 115:340–346
Quan LH, Min JW, Yang DU, Kim YJ, Yang DC (2012a) Enzymatic biotransformation of ginsenoside Rb1 to 20(S)-Rg3 by recombinant β-glucosidase from Microbacterium esteraromaticum. Appl Microbiol Biotechnol 94:377–384. doi:10.1007/s00253-011-3861-7
Quan LH, Min JW, Sathiyamoorthy S, Yang DU, Kim YJ, Yang DC (2012b) Biotransformation of ginsenosides Re and Rg1 into ginsenosides Rg2 and Rh1 by recombinant β-glucosidase. Biotechnol Lett 34:913–917. doi:10.1007/s10529-012-0849-z
Ruan CC, Zhang H, Zhang LX, Liu Z, Sun GZ, Lei J, Qin YX, Zheng YN, Li X, Pan HY (2009) Biotransformation of ginsenoside Rf to Rh1 by recombinant β-glucosidase. Molecules 14:2043–2048
Sarry JE, Gunata Z (2004) Plant and microbial glycoside hydrolases: volatile release from glycosidic aroma precursors. Food Chem 87:509–521
Shi W, Wang Y, Li J, Zhang H, Ding L (2007) Investigation of ginsenosides in different parts and ages of Panax ginseng. Food Chem 102:664–668
Son JW, Kim HJ, Oh DK (2008) Ginsenoside Rd production from the major ginsenoside Rb1 by beta-glucosidase from Thermus caldophilus. Biotechnol Lett 30:713–716
Wang CZ, Yuan CS (2008) Potential role of ginseng in the treatment of colorectal cancer. Am J Chin Med 36:1019–1028
Wang DM, Yu HS, Song JG, Xu YF, Jin FX (2012) Enzyme kinetics of ginsenosidase type IV hydrolyzing 6-O-multi-glycosides of protopanaxatriol type ginsenosides. Process Biochem 47:133–138
Wang DM, Yu HS, Song JG, Xu YF, Liu CY, Jin FX (2011a) A novel ginsenosidase from an Aspergillus strain hydrolyzing 6-O-multi-glycosides of protopanaxatriol-type ginsenosides, named ginsenosidase type IV. J Microbiol Biotechnol 21:1057–1063
Wang L, Liu QM, Sung BH, An DS, Lee HG, Kim SG, Kim SC, Lee ST, Im WT (2011b) Bioconversion of ginsenosides Rb1, Rb2, Rc and Rd by novel β-glucosidase hydrolyzing outer 3-O glycoside from Sphingomonas sp. 2F2: cloning, expression, and enzyme characterization. J Biotechnol 156:125–133
Yoo MH, Yeom SJ, Park CS, Lee KW, Oh DK (2011) Production of aglycon protopanaxadiol via compound K by a thermostable β-glycosidase from Pyrococcus furiosus. Appl Microbiol Biotechnol 89:1019–1028
Yu HS, Liu QM, Zhang CZ, Lu MC, Fu YY, Im WT, Lee ST, Jin FX (2009) A new ginsenosidase from Aspergillus strain hydrolyzing 20-O-multi-glycoside of PPD ginsenoside. Process Biochem 44:772–775
Yu HS, Zhang CZ, Lu MC, Sun F, Fu YY, Jin FX (2007) Purification and characterization of new special ginsenosidase hydrolyzing multi-glycisides of protopanaxadiol ginsenosides, ginsenosidase type I. Chem Pharm Bull (Tokyo) 55:231–235
Zhou W, Chai H, Lin PH, Lumsden AB, Yao Q, Chen CJ (2004) Molecular mechanisms and clinical applications of ginseng root for cardiovascular disease. Med Sci Monit 10:RA187–RA192
Acknowledgments
This work was supported by a grant from the Next-Generation BioGreen 21 Program (no. PJ008193), Rural Development Administration, and by the Intelligent Synthetic Biology Center of Global Frontier Project funded by the Ministry of Education, Science and Technology (2011-0031967), Republic of Korea.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 311 kb)
Rights and permissions
About this article
Cite this article
Cui, CH., Kim, SC. & Im, WT. Characterization of the ginsenoside-transforming recombinant β-glucosidase from Actinosynnema mirum and bioconversion of major ginsenosides into minor ginsenosides. Appl Microbiol Biotechnol 97, 649–659 (2013). https://doi.org/10.1007/s00253-012-4324-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4324-5