Abstract
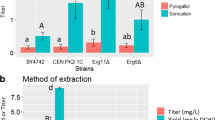
An efficient, time-saving, and cost-effective method for isolation of the polyamide cyanophycin from recombinant Saccharomyces cerevisiae was established. Due to its simple procedure, this isolation method may be also applicable at industrial scale and also to other intracellular compounds in this yeast. Production of cyanophycin gained preferential interest in the past, as degradation products thereof are of pharmaceutical and technical interest. Recently, it was shown that Saccharomyces cerevisiae represents a putative candidate for cyanophycin synthesis at industrial scale. For identification of optimal isolation procedures, several parameters such as heat stress, freeze drying, and freeze/thaw cycles of transgenic yeast cells were compared for their effectiveness of cyanophycin isolation. Additionally, optimal resuspension solutions for the applied cells and minimal required materials or chemicals were determined to make the process most environmentally and economically friendly. Maximal cyanophycin granule polypeptide yields of 21% (w/w) were obtained after incubation of dry cells at 70 °C or 80 °C and precipitation of the polymer with two volumes of ethanol.



Similar content being viewed by others
References
Aboulmagd E, Oppermann-Sanio FB, Steinbüchel A (2000) Molecular characterization of the cyanophycin synthetase from Synechocystis sp. strain PCC 6308. Arch Microbiol 174:297–306
Aboulmagd E, Voss I, Oppermann-Sanio FB, Steinbüchel A (2001) Heterologous expression of cyanophycin synthetase and cyanophycin synthesis in the industrial relevant bacteria Corynebacterium glutamicum and Ralstonia eutropha and in Pseudomonas putida. Biomacromolecules 2:1338–1342
Agbogbo FK, Coward-Kelly G (2008) Cellulosic ethanol production using the naturally occurring xylose-fermenting yeast, Pichia stipitis. Biotechnol Lett 30:1515–1524
Brachmann CB, Davies A, Cost GJ, Caputo E, Li J, Hieter P, Boeke JD (1998) Designer deletion strains derived from Saccharomyces cerevisiae S288C: a useful set of strains and plasmids for PCR-mediated gene disruption and other applications. Yeast 14:115–132
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cakar ZP, Seker UOS, Tamerler C, Sonderegger M, Sauer U (2005) Evolutionary engineering of multiple-stress resistant Saccharomyces cerevisiae. FEMS Yeast Res 5:569–578
Elbahloul Y, Frey K, Sanders J, Steinbüchel A (2005) Protamylasse, a residual compound of industrial starch production, provides a suitable medium for large-scale cyanophycin production. Appl Environ Microbiol 71:7759–7767
Frey KM, Oppermann-Sanio FB, Schmidt H, Steinbüchel A (2002) Technical-scale production of cyanophycin with recombinant strains of Escherichia coli. Appl Environ Microbiol 68:3377–3384
Füser G, Steinbüchel A (2005) Investigations on the solubility behavior of cyanophycin. Solubility of cyanophycin in solutions of simple inorganic salts. Biomacromolecules 6:1367–1374
Gellissen G, Kunze G, Gaillardin C, Cregg JM, Berardi E, Veenhuis M, van der Klei I (2005) New yeast expression platforms based on methylotrophic Hansenula polymorpha and Pichia pastoris and on dimorphic Arxula adeninivorans and Yarrowia lipolytica—a comparison. FEMS Yeast Res 5:1079–1096
Henes B, Sonnleitner B (2007) Controlled fed-batch by tracking the maximal culture capacity. J Bacteriol 132:118–126
Hühns M, Neumann K, Hausmann T, Ziegler K, Klemke F, Kahmann U, Staiger D, Lockau W, Pistorius EK, Broer I (2008) Plastid targeting strategies for cyanophycin synthetase to achieve high-level polymer accumulation in Nicotiana tabacum. Plant Biotechnol J 6:321–336
Krehenbrink M, Oppermann-Sanio FB, Steinbüchel A (2002) Evaluation of non-cyanobacterial genome sequences for occurrence of genes encoding proteins homologous to cyanophycin synthetase and cloning of an active cyanophycin synthetase from Acinetobacter sp. strain DSM 587. Arch Microbiol 177:371–380
Kroll J, Steinle A, Reichelt R, Ewering C, Steinbüchel A (2009) Establishment of a novel anabolism-based addiction system with an artificially introduced mevalonate pathway: complete stabilization of plasmids as universal application in white biotechnology. Metabol Eng 11:168–177
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Neumann K, Stephan DP, Ziegler K, Hühns M, Broer I, Lockau W, Pistorius EK (2005) Production of cyanophycin, a suitable source for the biodegradable polymer polyaspartate, in transgenic plants. Plant Biotechnol J 3:249–258
Park J-I, Grant CM, Attfield PV, Dawes IW (1997) The freeze-thaw stress response of the yeast Saccharomyces cerevisiae is growth phase specific and is controlled by nutritional state via the RAS-cyclic AMP signal transduction pathway. Appl Environ Microbiol 63:3818–3824
Sallam A, Steinbüchel A (2009) Cyanophycin-degrading bacteria in digestive tracts of mammals, birds and fish, and consequences for possible applications of cyanophycin and its dipeptides in nutrition and therapy. J Appl Microbiol 107:474–484
Sallam A, Steinle A, Steinbüchel A (2009a) Cyanophycin: biosynthesis and applications. In: Rehm BHA (ed) Microbial production of biopolymers and polymer precursors. Caister Academic Press, Norfolk, UK
Sallam A, Kast A, Przybilla S, Meiswinkel T, Steinbüchel A (2009b) Process for biotechnological production of β-dipeptides from cyanophycin at technical scale and its optimization. Appl Environ Microbiol 75:29–38
Steinle A, Oppermann-Sanio FB, Reichelt R, Steinbüchel A (2008) Synthesis and accumulation of cyanophycin in transgenic strains of Saccharomyces cerevisiae. Appl Environ Microbiol 74:3410–3418
Steinle A, Bergander K, Steinbüchel A (2009) Metabolic engineering of Saccharomyces cerevisiae for production of novel cyanophycins with an extended range of constituent amino acids. Appl Environ Microbiol 75:3437–3446
Teunissen A, Dumortier F, Gorwa M-F, Bauer J, Tanghe A, Loiez A, Smet P, Van Dijck P, Thevelein JM (2002) Isolation and characterization of freeze-tolerant diploid derivative of an industrial baker’s yeast strain and its use in frozen doughs. Appl Environ Microbiol 68:4780–4787
Toscani S, Céolin R, Vincent J (1999) Physicochemical differences in dehydrated Saccharomyces boulardii yeast as a function of the dehydration process. J Therm Anal Calorim 57:293–301
Voss I, Steinbüchel A (2001) High cell density cultivation of Rhodococcus opacus for lipid production at a pilot-plant scale. Appl Microbiol Biotechnol 55:547–555
Wei P, Li Z, Lin Y, He P, Jiang N (2007) Improvement of the multiple-stress tolerance of an ethanologenic Saccharomyces cerevisiae strain by freeze-thaw treatment. Biotechnol Lett 29:1501–1508
Ziegler K, Deutzmann R, Lockau W (2002) Cyanophycin synthetase-like enzymes of non-cyanobacterial eubacteria: characterization of the polymer produced by a recombinant synthetase of Desulfitobacterium hafniense. Z Naturforsch 57:522–529
Acknowledgments
This project was supported by a grant (EOSLT02034) provided by SenterNovem (Utrecht, the Netherlands).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Steinle, A., Steinbüchel, A. Establishment of a simple and effective isolation method for cyanophycin from recombinant Saccharomyces cerevisiae . Appl Microbiol Biotechnol 85, 1393–1399 (2010). https://doi.org/10.1007/s00253-009-2213-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2213-3