Abstract
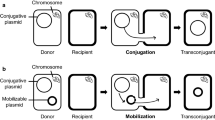
Because engineering of the 101.016-bp megaplasmid pKB1 of Gordonia westfalica Kb1 failed due to the absence of an effective transfer system, pKB1 was transferred by conjugation from G. westfalica Kb1 to a kanamycin-resistant mutant of Rhodococcus opacus PD630 at a frequency of about 6.2 × 10−8 events per recipient cell. Furthermore, pKB1 was transferred to G. polyisoprenivorans strains VH2 and Y2K and to Mycobacterium smegmatis by electroporation at frequencies of 5.5 × 103, 1.9 × 103, and 8.3 × 102 transformants per microgram plasmid DNA. The pKB1-encoded cadmium resistance gene cadA was used for selection in these experiments. Recombinant pKB1-containing G. polyisoprenivorans VH2 and M. smegmatis were then used to engineer pKB1. A kanamycin resistance cassette was inserted into the pKB1-encoded cadA gene, ligated to suicide plasmid pBBR1MCS-5, and the resulting plasmid was electroporated into plasmid-harboring strains. Homologous recombination between cadA on suicide plasmid and the respective sequence in pKB1 led to its integration into pKB1. Thus, two selection markers were accommodated in pKB1 to monitor plasmid transfer into Gordonia and related taxa for analysis of genes essential for rubber degradation and others. In this study, two transfer methods for large plasmids and strategies for engineering of pKB1 were successfully applied, thereby, extending the tool box for Gordonia.


Similar content being viewed by others
References
Arenskötter M, Baumeister D, Berekaa MM, Pötter G, Kroppenstedt RM, Linos A, Steinbüchel A (2001) Taxonomic characterization of two rubber-degrading bacteria belonging to the species Gordonia polyisoprenivorans and analysis of hypervariable regions of 16S rDNA sequences. FEMS Microbiol Lett 205:277–282
Arenskötter M, Baumeister D, Kalscheuer R, Steinbüchel A (2003) Identification and application of plasmids suitable for transfer of foreign DNA to members of the genus Gordonia. Appl Environ Microbiol 69:4971–4974
Arenskötter M, Bröker D, Steinbüchel A (2004) Biology of the metabolically diverse genus Gordonia. Appl Environ Microbiol 70:3195–3204
Arenskötter M, Linos A, Schumann P, Kroppenstedt RM, Steinbüchel A (2005) Gordonia nitida Yoon et al. 2001 is a later synonym of Gordonia alkanivorans Kummer et al. 1999. Int J Syst Evol Microbiol 55:695–697
Banh Q, Arenskötter M, Steinbüchel A (2005) Establishment of Tn5096-based transposon mutagenesis in Gordonia polyisoprenivorans. Appl Environ Microbiol 71:5077–5084
Birnboim HC, Doly J (1979) A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res 7:1513–1523
Bröker D, Arenskötter M, Legatzki A, Nies DH, Steinbüchel A (2004) Characterization of the 101.016-kbp megaplasmid pKB1 isolated from the rubber degrading bacterium Gordonia westfalica Kb1. J Bacteriol 186:212–225
Bullock WO, Fernandez JM, Stuart JM (1987) XL1-Blue: a high efficiency plasmid transforming recA Escherichia coli strain with β-galactosidase selection. Bio Techniques 5:376–379
Byrd DR, Matson SW (1997) Nicking by transesterification: the reaction catalysed by a relaxase. Mol Microbiol 25:1011–1022
Desomer J, Dhaese P, van Montagu M (1988) Conjugative transfer of cadmium resistance plasmids in Rhodococcus fascians strains. J Bacteriol 170:2401–2405
Dogan I, Pagilla KR, Webster DA, Stark BC (2006) Expression of Vitreoscilla hemoglobin in Gordonia amarae enhances biosurfactant production. J Ind Microbiol Biotechnol 33:693–700
Friedrich B, Hogrefe C, Schlegel HG (1981) Naturally occurring genetic transfer of hydrogen-oxidizing ability between strains of Alcaligenes eutrophus. J Bacteriol 147:198–205
Galamba A, Soetaert K, Wang X-M, De Bruyn J, Jacobs P, Content J (2001) Disruption of adhC reveals a large duplication in the Mycobacterium smegmatis mc2155 genome. Microbiology 147:3281–3294
Gilbert SC, Morton J, Buchanan S, Oldfield C, McRoberts A (1998) Isolation of a unique benzothiophene-desulphurizing bacterium, Gordona sp. strain 213E (NCIMB 40816), and characterization of desulphurization pathway. Microbiology (SGM) 144:2545–2553
Grohmann E, Muth G, Espinosa M (2003) Conjugative plasmid transfer in Gram-positive bacteria. Microbiol Mol Biol Rev 67:277–301
Guilhot C, Otal I, van Rompaey I, Martìn C, Gicquel B (1994) Efficient transposition in mycobacteria: construction of Mycobacterium smegmatis insertional mutant libraries. J Bacteriol 176:535–539
Hernandez-Perez G, Fayolle F, Vandecasteele J-P (2001) Biodegradation of ethyl t-butyl ether (ETBE), methyl t-butyl ether (MTBE) and t-amyl methyl ether (TAME) by Gordonia terrae. Appl Microbiol Biotechnol 55:117–121
Hinds J, Mahenthiralingam E, Kempsell KE, Duncan K, Stokes RW, Parish T, Stoker NG (1999) Enhanced gene replacement in mycobacteria. Microbiology 145:519–527
Husson RN, James BE, Young RA (1990) Gene replacement and expression of foreign DNA in mycobacteria. J Bacteriol 172:519–524
Ibrahim EMA, Arenskötter M, Luftmann H, Steinbüchel A (2006) Identification of poly(cis-1,4-isoprene) degradation intermediates during growth of moderately thermophilic Actinomycetes on rubber and cloning of a functional lcp homologue from Nocardia farcinica strain E1. Appl Environ Microbiol 72:3375–3382
Kalscheuer R, Arenskötter M, Steinbüchel A (1999) Establishment of a gene transfer system for Rhodococcus opacus PD630 based on electroporation and its application for recombinant biosynthesis of poly(3-hydroxyalkanoic acids). Appl Microbiol Biotech 52:508–515
Kim SB, Brown R, Oldfield C, Gilbert SC, Goodfellow M (1999) Gordonia desulfuricans sp. nov., a benzothiophene-desulfurizing actinomycete. Int J Syst Bacteriol 49:1845–1851
Kim SB, Brown R, Oldfield C, Gilbert SC, Iliarionov S, Goodfellow M (2000) Gordonia amicalis sp. nov., a dibenzothiophene–desulfurizing actinomycete. Int J Syst Bacteriol 50:2031–2036
Kotani T, Yamamoto T, Yurimoto H, Sakai Y, Kato N (2003) Propane monooxygenase and NAD+-dependent secondary alcohol dehydrogenase in propane metabolism by Gordonia sp. strain TY-5. J Bacteriol 185:7120–7128
Kotani T, Yurimoto H, Kato N, Sakai Y (2007) Novel acetone metabolism in a propane-utilizing bacterium, Gordonia sp. strain TY-5. J Bacteriol 189:886–893
Kovach ME, Elzer PH, Hill DS, Robertson GT, Farris MA, Roop RM, Peterson KM (1995) Four new derivates of the broad host range cloning vector pBBR1MCS, carrying different antibiotic resistance cassettes. Gene 166:175–176
Kummer C, Schumann P, Stackebrandt E (1999) Gordonia alkanivorans sp. nov., isolated from tar-contaminated soil. Int J Syst Bacteriol 49:1513–1522
Linos A, Steinbüchel A (1998) Microbial degradation of natural and synthetic rubbers by novel bacteria belonging to the genus Gordona. Kautsch Gummi Kunstst 51:496–499
Linos A, Steinbüchel A, Spröer C, Kroppenstedt RM (1999) Gordonia polyisoprenivorans sp. nov., a rubber degrading actinomycete isolated from automobile tire. Int J Syst Bacteriol 49:1785–1791
Linos A, Berekaa MM, Steinbüchel A, Kim KK, Spröer C, Kroppenstedt RM (2002) Gordonia westfalica sp. nov., a novel rubber-degrading actinomycete. Int J Syst Evol Microbiol 52:1133–1139
Makarova KS, Grishin NV, Shabalina SA, Wolf YI, Koonin EV (2006) A putative RNA-interference-based immune system in prokaryotes: computational analysis of the predicted enzymatic machinery, functional analogies with eukaryotic RNAi, and hypothetical mechanisms of action. Biol Direct 1:7–32
Mozo T, Hooykaas PJJ (1991) Electroporation of megaplasmids into Agrobacterium. Plant Mol Biol 16:917–918
McFadden J (1996) Recombination in mycobacteria. Mol Microbiol 21:2056–211
Navarre WW, McClelland M, Libby SJ, Fang FC (2007) Silencing of xenogeneic DNA by H-NS—facilitation of lateral gene transfer in bacteria by a defense system that recognizes foreign DNA. Genes Dev 21:1456–1471
Pansegrau W, Lanka E (1996) Enzymology of DNA transfer by conjugative mechanisms. Prog Nucleic Acid Res Mol Biol 54:197–251
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY
Schlegel HG, Kaltwasser H, Gottschalk G (1961) Ein Submersverfahren zur Kultur wasserstoffoxidierender Bakterien: Wachstumsphysiologische Untersuchungen. Arch Mikrobiol 38:209–222
Sheng Y, Mancino V, Birren B (1995) Transformation of Escherichia coli with large DNA molecules by electroporation. Nucleic Acids Res 23:1990–1996
Snapper SB, Melton RE, Mustafa S, Kieser T, Jacobs WR Jr (1990) Isolation and characterization of efficient plasmid transformation mutants of Mycobacterium smegmatis. Mol Microbiol 4:1911–1919
Wilson GG, Murray NE (1991) Restriction and modification systems. Annu Rev Genet 25:585–627
Yang JC, Lessard PA, Sengupta N, Windsor SD, O’Brien XM, Bramucci M, Tomb J-F, Nagarajan V, Sinskey AJ (2007) TraA is required for megaplasmid conjugation in Rhodococcus erythropolis AN12. Plasmid 57:55–70
Veiga-Crespo P, Feijoo-Siota L, de Miguel T, Poza M, Villa TG (2006) Proposal of a method for the genetic transformation of Gordonia jacobae. J Appl Microbiol 100:608–614
Yoon J-H, Lee JJ, Kang SS, Takeuchi M, Shin YK, Lee ST, Kang KH, Park YH (2000) Gordonia nitida sp. nov., a bacterium that degrades 3-ethylpyridine and 3-methylpyridine. Int J Syst Evol Microbiol 50:1203–1210
Acknowledgment
Financial support by a grant of the Deutsche Forschungsgemeinschaft (STE 386/10–1) is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bröker, D., Arenskötter, M. & Steinbüchel, A. Transfer of megaplasmid pKB1 from the rubber-degrading bacterium Gordonia westfalica strain Kb1 to related bacteria and its modification. Appl Microbiol Biotechnol 77, 1317–1327 (2008). https://doi.org/10.1007/s00253-007-1262-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-007-1262-8