Abstract.
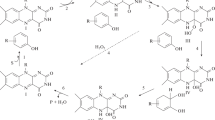
The dibenzothiophene (DBT) desulfurizing bacterium metabolizes DBT to form 2-hydroxybiphenyl without breaking the carbon skeleton. Of the DBT desulfurization enzymes, DszC and DszA catalyze monooxygenation reactions, both requiring flavin reductase. We searched for non-DBT-desulfurizing microorganisms producing a flavin reductase that couples more efficiently with DszC than that produced by the DBT desulfurizing bacterium Rhodococcus erythropolis D-1, and found Paenibacillus polymyxa A-1 to be a promising strain. The enzyme was purified to complete homogeneity. K m values for FMN and NADH were 2.1 µM and 0.57 mM, respectively. Flavin compounds were good substrates, some nitroaromatic compounds were also active, and regarding the electron donor, the activity for NADPH was about 1.5 times that for NADH. In the coupling assay with DszC, only FMN or riboflavin acted as the electron acceptor. The coupling reactions of P. polymyxa A-1 flavin reductase with DszC and DszA proceeded more efficiently (3.5- and 5-fold, respectively) than those of R. erythropolis D-1 flavin reductase when identical enzyme activities of each flavin reductase were added to the reaction mixture. The result of the coupling reaction suggested that, in the microbial DBT desulfurization, flavin reductase from the non-DBT-desulfurizing bacterium was superior to that from the DBT-desulfurizing bacterium.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ohshiro, T., Aoi, Y., Torii, K. et al. Flavin reductase coupling with two monooxygenases involved in dibenzothiophene desulfurization: purification and characterization from a non-desulfurizing bacterium, Paenibacillus polymyxa A-1. Appl Microbiol Biotechnol 59, 649–657 (2002). https://doi.org/10.1007/s00253-002-1082-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-002-1082-9